Impact of Coronavirus (Covid-19) Pandemic on the Clinical Diagnostics Industry
Since the onset of the world’s largest pandemic – Coronavirus (COVID-19), the overall clinical diagnostics industry is witnessing a sharp rise in demand for its products, especially in-vitro diagnostics. To control the outbreak, diagnostic device and consumable manufacturers have changed their strategies to address the diagnostics demand especially in the lower and middle income countries. There are more than 500 SARS-CoV-2 diagnostics products (the causative agent of COVID-19) in the form of immunoassays, molecular assays, sample collection kits and digital solutions which have been commercialized by respective regulatory bodies including US FDA, EMA, SFDA, and WHO EUL among others.
As diagnosis is the stepping stone to reduce incidence and mortality rate, regulatory bodies are focusing on initiatives to collaborate with diagnostic manufacturers to increase the production and supply of diagnostic products for Coronavirus in each country. On April 24, 2020, World Health Organization (WHO) launched the global collaboration act accelerator “Access to COVID-19 Tools Accelerator” to speed up the development and production of COVID-19 diagnostics and therapeutics products, globally. This will propel initiatives and activities within the clinical diagnostics industry worldwide.
The reports from Coherent Market Insights considers and evaluates the impact of the COVID-19, and anticipated change on the future outlook of this industry, by taking into the account the political, economic, social, and technological parameters.
Comparative Analysis - 1
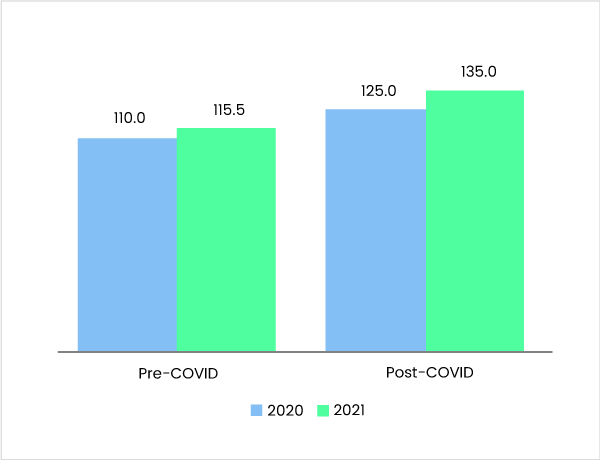
Pre-Covid-19 Vs Post Covid-19 - Market Estimates, 2020 – 2021 (US$ Bn)
Comparative Analysis - 2
Top Gainers Vs Top Losers
Marginal Growth
- Analyzers
- Other Assays
Top Gainers
- IVD
- Immunoassays
- Blood testing devices
Marginal Loss
- PCR
- Disease testing
Top Losers
- Chromatography

Senior Consultant Healthcare Research

Consultant Level 1 Healthcare Research
- Diagnostic industry experienced huge demand in terms of testing assay kits for COVID-19
- Diabetes testing, infectious disease testing, molecular diagnostics, NGS and other such testing devices is expected to face a downtrend during this pandemic
- Demand from diagnostic laboratories also fell during the pandemic due to lower patient volume






