Impact of Coronavirus (Covid-19) Pandemic on the Pharmaceutical Industry
The coronavirus pandemic represents a key challenge to the healthcare ecosystem comprising pharmaceutical and biotechnology industries. Having said that, it does represent a sizeable market opportunity for few of the pharmaceutical companies. Amid increasing number of cases of Covid-19, the need for effective therapies to treat Covid-19 has led to substantial upsurge in collaborative activities across the pharmaceutical sector. Standing out as one of the most prolific deal makers is Vir Biotechnology with its two antibodies, targeting the spike protein of the COVID-19 virus. The company signed deals with Wuxi, Alnylam, Xencor, the NIH, and Biogen, for development of antibodies, RNAi therapeutics, and vaccines for treatment of coronavirus. However, pharmaceutical businesses have been disrupted, and this would have financial implications on the Q2 and Q3 of 2020. In addition to this, many ongoing clinical research studies will be put on hold or cancelled, amid the social distancing norms implemented in most of the leading countries worldwide. Pharmaceutical drug makers such as AstraZeneca, Pfizer, and Eli Lilly have delayed the new and existing trials owing to reasons such as lockdown, difficulties in accessing hospitals and doctors, change in the priorities of labs, and hurdles in leveraging the logistic infrastructure.
The reports from Coherent Market Insights considers and evaluates the impact of the COVID-19, and anticipated change on the future outlook of this industry, by taking into the account the political, economic, social, and technological parameters.
Comparative Analysis - 1
Pre-Covid-19 Vs Post Covid-19 - Market Estimates, 2020 – 2021 (US$ Bn)
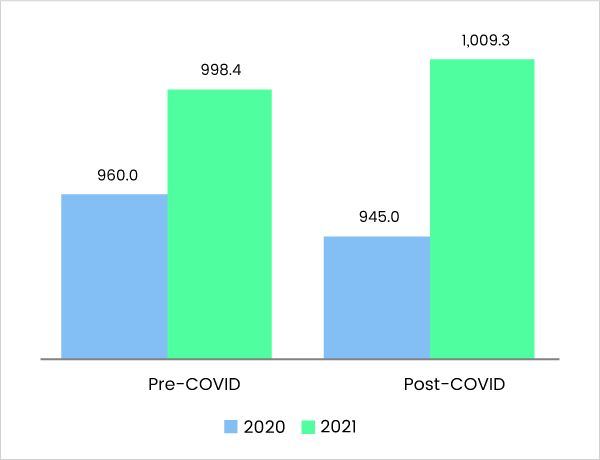
Comparative Analysis - 2
Top Gainers Vs Top Losers
Marginal Growth
- Neruo/CNS
- Respiratory
Top Gainers
- Cardiac
- Anti-diabetic
Marginal Loss
- Derma
- Hormones
- Gynaec
- GI
Top Losers
- Anti-infective
- Pain
- VMS
- Vaccines
- Ophthal

Principal Consultant Healthcare Research

Consultant Level 1 Healthcare Research
- The industry is impacted with the closing of manufacturing units across the globe. Some units have been working with minimal strength, leading to shortage or delay of product availability.
- Focus of the pharmaceutical industry has shifted to development of treatment for Corona
- Chronic diseases, demanding long term medications, are less impacted during the pandemic
- On account of reduced patient volume, sales of derma, ophthalmic, VMS, pain and other such therapies has reduced significantly






