Advanced Therapy Medicinal Products Market Size and Forecast – 2026 – 2033
The global Advanced Therapy Medicinal Products market is projected to grow at a CAGR of approximately 18% between 2026 and 2033. Valued near USD 30 billion in 2026, it is expected to exceed USD 100 billion by 2033, driven by rising approvals, expanding clinical pipelines, and increasing investment in gene and cell therapies worldwide.
Global Advanced Therapy Medicinal Products Market Overview
Advanced Therapy Medicinal Products are innovative medicines based on genes, cells, or tissue engineering, designed to treat or cure serious diseases. They include gene therapies, somatic cell therapies, tissue-engineered products, and combined ATMPs. In the European Union, ATMPs are regulated under a specific framework established by the European Medicines Agency to ensure quality, safety, and efficacy. These therapies target conditions such as cancer, genetic disorders, and degenerative diseases, offering personalized and regenerative treatment approaches. Although development costs and complex manufacturing pose challenges, ATMPs represent a rapidly growing segment of biotechnology with transformative potential for modern healthcare.
Key Takeaways
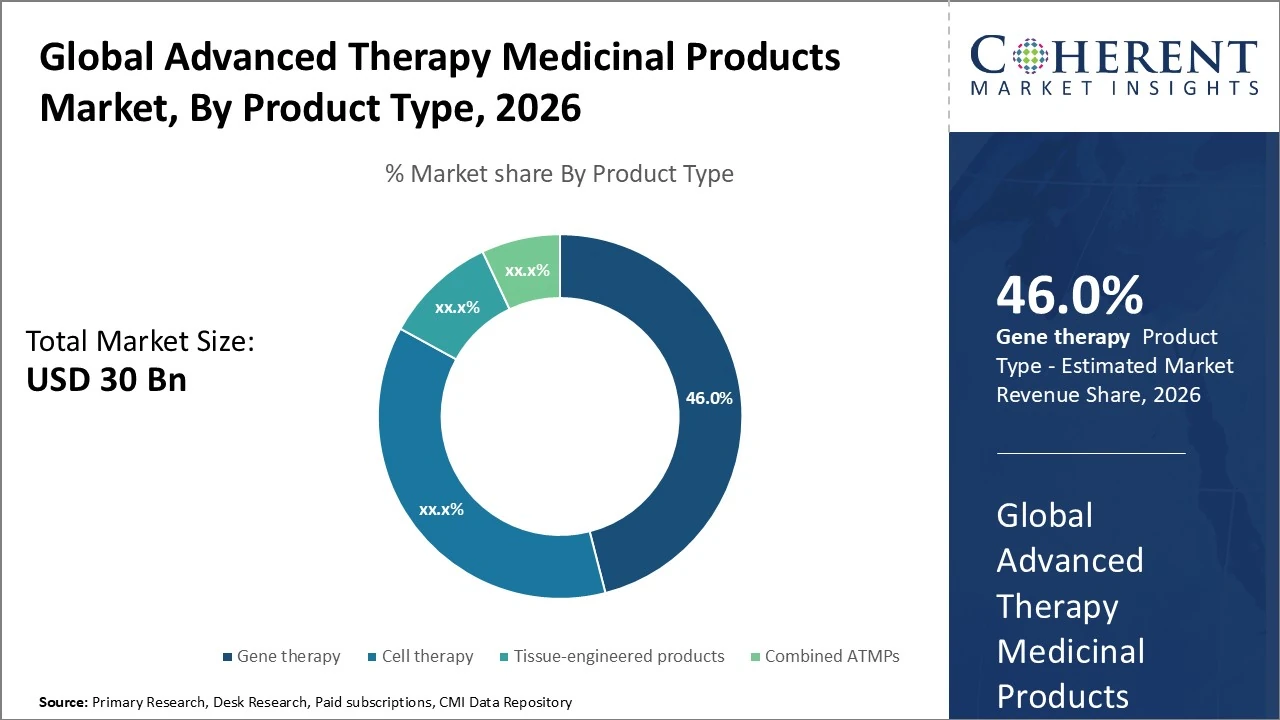
The Advanced Therapy Medicinal Products market, by product type, Gene therapy holds the largest share at approximately 46%.
The Advanced Therapy Medicinal Products market, by technology, Viral vector-based platforms dominate with approximately 50% share.
The Advanced Therapy Medicinal Products market, by end-user, Hospitals dominate with approximately 55% market share.
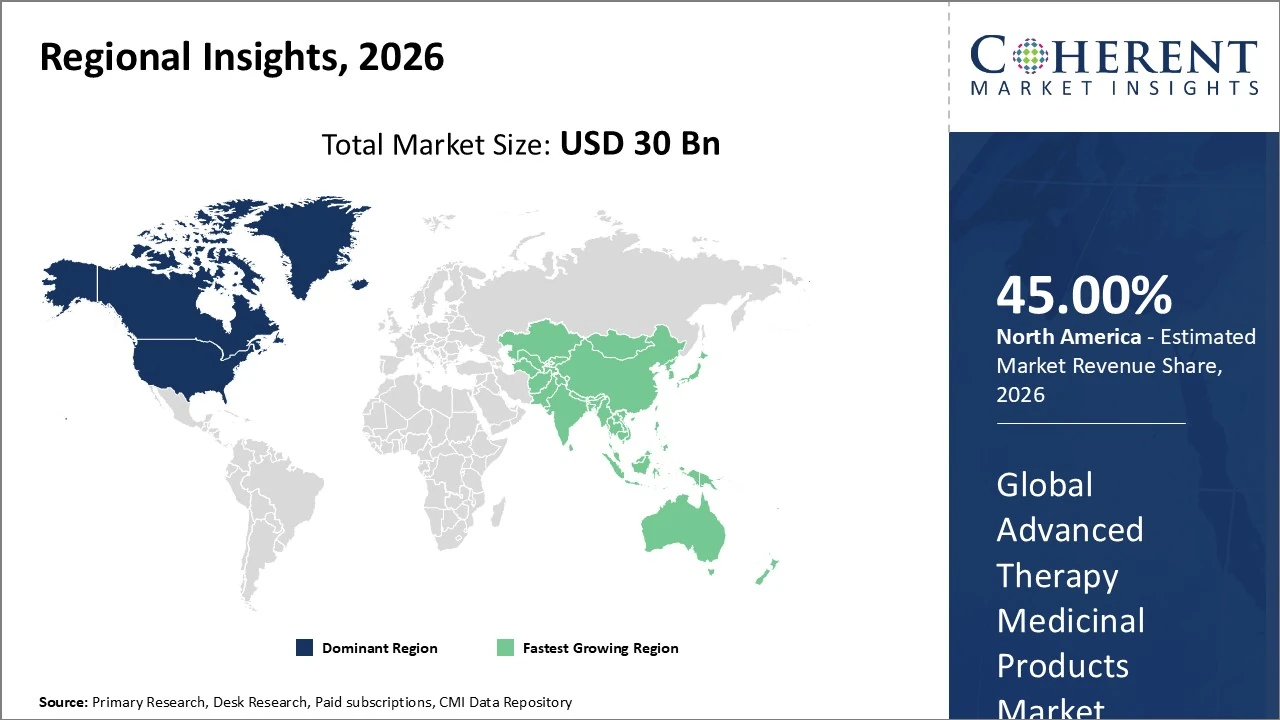
North America leads the Advanced Therapy Medicinal Products market with roughly 45% regional share.
The Asia Pacific Advanced Therapy Medicinal Products market is one of the fastest-growing regional segments.
Advanced Therapy Medicinal Products Market Segmentation Analysis
To learn more about this report, Request Free Sample
Advanced Therapy Medicinal Products Market Insights, By Product Type
Gene therapy holds the largest share at approximately 46%, driven by rising approvals and strong pipelines for rare genetic disorders and oncology. Cell therapy accounts for around 37%, supported by CAR-T innovations and expanding clinical applications. Tissue-engineered products contribute nearly 10%, mainly in wound care and orthopedics. Combined ATMPs represent about 7% of the market. Overall, gene and cell therapies dominate with over 80% share, reflecting higher investment, regulatory momentum, and commercialization potential.
Advanced Therapy Medicinal Products Market Insights, By Technology
Viral vector-based platforms dominate with approximately 50% share, widely used in gene and cell therapies. Cell processing and expansion technologies account for around 30%, driven by CAR-T and stem cell advancements. Gene editing technologies, including CRISPR-based systems, contribute nearly 10%, reflecting rapid innovation and clinical adoption. Non-viral delivery technologies hold about 10% share. Viral vectors remain central due to proven efficacy, while gene editing is projected to witness the fastest growth through 2033.
Advanced Therapy Medicinal Products Market Insights, By End-User
Hospitals dominate with approximately 55% market share, driven by advanced infrastructure, certified treatment centers, and administration of complex gene and cell therapies. Specialty clinics account for around 30%, particularly in oncology and regenerative medicine applications. Research and academic institutes contribute nearly 15%, supported by clinical trials and translational research activities. Hospitals remain the primary end-users due to regulatory requirements and specialized handling capabilities, while specialty centers are expected to witness steady growth through 2033.
Advanced Therapy Medicinal Products Market Trends
Increasing regulatory approvals of gene and CAR-T cell therapies are accelerating market growth, with gene therapies accounting for nearly 46% of the total ATMP market share.
Growing demand for precision treatments in oncology and rare diseases is driving investment, with over 60% of ATMP pipelines focused on personalized and targeted therapies.
Adoption of automated cell processing and CRISPR-based gene editing platforms is improving scalability and reducing production timelines, supporting an expected double-digit CAGR of 18% through 2033.
Advanced Therapy Medicinal Products Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Advanced Therapy Medicinal Products Market Analysis and Trends
North America leads the Advanced Therapy Medicinal Products market with roughly 45% regional share, driven by strong R&D investment, a robust biotechnology ecosystem, and a favorable regulatory environment including accelerated pathways from the U.S. FDA. The United States accounts for the majority of clinical trials and commercial launches, particularly in gene and cell therapies, fueling adoption in hospitals and specialty centers. Canada’s harmonized standards further support market growth. Increased funding, strategic collaborations, and advanced manufacturing capacity continue to bolster innovation, while payer and reimbursement complexities remain challenges.
Asia Pacific Advanced Therapy Medicinal Products Market Analysis and Trends
The Asia Pacific Advanced Therapy Medicinal Products market is one of the fastest-growing regional segments, driven by rising investment, expanding clinical activity, and supportive government initiatives across China, Japan, South Korea, and India. APAC held roughly 24% of global ATMP revenue by 2025 and is expected to grow at a 22% CAGR through 2033, outpacing many mature regions. Gene and cell therapies dominate, with China often leading growth rates, while CDMO and manufacturing capacities rapidly expand. Increasing demand for treatments targeting cancer, rare diseases, and genetic disorders further fuels innovation and adoption across healthcare systems.
Advanced Therapy Medicinal Products Market Outlook for Key Countries
USA Advanced Therapy Medicinal Products Market Analysis and Trends
The U.S. Advanced Therapy Medicinal Products market is rapidly expanding. This growth is fueled by strong gene and cell therapy innovation, high clinical trial activity, and supportive regulatory frameworks like accelerated pathways from the U.S. Food and Drug Administration (FDA). Gene therapies lead the landscape, holding roughly ~49% share, while cell therapies are among the fastest-growing segments. Outsourcing to CDMOs and significant R&D investments further bolster commercialization and adoption.
Germany Advanced Therapy Medicinal Products Market Analysis and Trends
Germany’s Advanced Therapy Medicinal Products market is expanding rapidly. Gene therapies currently lead revenue, while cell therapies are the fastest-growing product segment. Germany hosts strong manufacturing infrastructure and research activity, with increasing numbers of clinical studies and high availability of ATMPs relative to other EU countries (≈89% availability). However, application rates and reimbursement frameworks remain evolving, driving demand for supportive policies and wider adoption.
Analyst Opinion
Analysts project a 18% CAGR through 2033, driven by gene and cell therapy innovations and increasing rare disease applications.
Expedited approvals in the U.S., EU, and select APAC countries are accelerating market entry, though differing reimbursement frameworks pose challenges.
Gene therapies hold the largest market share (48%), while CAR-T and stem cell therapies are rapidly expanding globally.
Production costs, vector supply, and complex logistics remain critical hurdles for commercialization.
Partnerships between biotech firms, hospitals, and CDMOs are essential for R&D acceleration, clinical adoption, and global market expansion.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 30 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 18% | 2033 Value Projection: | USD 100 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Novartis AG, Gilead Sciences, Inc., Bristol Myers Squibb, Pfizer Inc., Bluebird Bio, Lonza Group AG | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Advanced Therapy Medicinal Products Growth Factors
The growth of the Advanced Therapy Medicinal Products market is driven by several factors. Rapid advancements in gene editing and cell therapy technologies enable targeted and personalized treatments, particularly in oncology and rare genetic disorders. Increasing regulatory support in the U.S., EU, and Asia accelerates approvals, while rising healthcare investments and venture funding bolster R&D. Expanding CDMO and manufacturing infrastructure ensures scalability and global reach. Growing awareness among clinicians and patients about regenerative and curative therapies further fuels adoption. Additionally, the rising prevalence of chronic, degenerative, and genetic diseases creates a strong unmet need for innovative ATMP solutions.
Advanced Therapy Medicinal Products Market Development
In May 2025, Berlin Institute of Health at Charité (BIH) partnered with BioLabs to launch a Clinical Incubator focused on translating Advanced Therapy Medicinal Products (ATMPs) into clinical practice. Through this agreement, BioLabs will support operations of the full-service BIH Clinical Incubator (CLIC), which aims to accelerate the development of innovative advanced therapies and promote entrepreneurship in regenerative medicine.
Key Players
Leading Companies of the Market
Novartis AG
Gilead Sciences, Inc.
Bristol Myers Squibb
Pfizer Inc.
Bluebird Bio
Lonza Group AG
Key players in the Advanced Therapy Medicinal Products (ATMP) market include Novartis AG, Gilead Sciences, Inc. (Kite Pharma), Bristol Myers Squibb, Pfizer Inc., Bluebird Bio, and Lonza Group AG, focusing on gene therapies, cell therapies, and contract development/manufacturing services.
Advanced Therapy Medicinal Products Market Future Outlook
The future outlook for the Advanced Therapy Medicinal Products (ATMP) market is highly promising, with a projected CAGR of 18% through 2033. Gene and cell therapies will continue to dominate, supported by innovations in CRISPR, CAR-T, and stem cell technologies. Expansion of CDMO capacities, automated manufacturing, and global distribution networks will enhance accessibility and scalability. Regulatory incentives and faster approval pathways in the U.S., EU, and APAC will accelerate market entry. Increasing prevalence of rare genetic, oncology, and degenerative diseases, coupled with rising patient awareness and investment, positions ATMPs as a cornerstone of personalized and regenerative medicine worldwide.
Advanced Therapy Medicinal Products Market Historical Analysis
The Advanced Therapy Medicinal Products (ATMP) market has experienced significant growth over the past decade. From 2010 to 2025, the market expanded, driven primarily by the emergence of gene and cell therapies. Early regulatory approvals in the U.S. and EU, such as CAR-T and first-generation gene therapies, laid the foundation for commercialization. Investments in biotech R&D, clinical trials, and CDMO infrastructure accelerated development. Initial challenges, including high manufacturing costs, complex logistics, and limited awareness, gradually diminished as stakeholders adopted scalable processes, strategic collaborations, and targeted reimbursement models, setting the stage for rapid future growth.
Sources
Primary Research Interviews:
Biopharmaceutical Companies
Contract Development and Manufacturing Organizations
Healthcare Providers
Regulatory Authorities
Databases:
PubMed / MEDLINE
ClinicalTrials.gov
EMBASE
Journals:
Nature Biotechnology
Cell Stem Cell
Molecular Therapy
Cytotherapy
Newspapers:
Reuters Health
Financial Times
The Guardian
The New York Times
Associations:
Alliance for Regenerative Medicine (ARM)
International Society for Cell & Gene Therapy (ISCT)
European Society of Gene & Cell Therapy (ESGCT)
Cell & Gene Therapy Catapult
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients