The Viral Vectors and Plasmid DNA Manufacturing Market is anticipated to grow at a CAGR of 23% with USD 1,798.6 Mn share in 2026 and is expected to reach USD 7,571.1 Mn in 2033. Rising gene and cell therapy pipelines (According to American Society of Gene & Cell Therapy (ASGCT) as of Q4 2025, 1,888 gene therapy clinical trials were open globally, rising clinical trial activity, increasing GMP manufacturing needs, and advances in vector design and production technologies are driving the growth of the Viral Vectors and Plasmid DNA Manufacturing Market, supported by broader therapeutic applications and expanding commercialization of advanced therapies.
Increasing number of clinical trials has significantly driven the viral vectors and plasmid DNA manufacturing market growth. A significant increase in clinical trials for gene therapy and advanced biologics has increased the demand for the viral vectors and plasmid DNA manufacturing. Hece, due to increasing number of clinical trials for new drug discovery there is rise in demand for GMP-grade vectors and plasmids. For instance, in April 2025, according to the data published by the National Library of Medicine (NLM) reported in April 2025 that since ClinicalTrials.gov's establishment in 2000, over 573,913 clinical studies have been registered. According to ClinicalTrials.gov, 37,597 new studies were uploaded to the registry in 2025 alone, demonstrating a high level of continuous registration activity. According to a different source, more than 7,000 new studies were registered on ClinicalTrials.gov in 2026, maintaining the high yearly trial posting pattern. There have been about 106 new trials registered in 2026 that combine gene therapy (79) and non-genetically modified cell treatment (27).
Favorable government support and regulation have estimated to create significant opportunity for the growth of the viral vectors and plasmid DNA manufacturing market in the near future. Policies promoting biopharma innovation and manufacturing infrastructure has driven the viral vectors and plasmid DNA manufacturing market demand. Increasing regulatory approvals encourage commercialization of the viral vectors and plasmid DNA for drug discovery purpose.
Increasing adoption of inorganic growth strategies like partnership and collaboration has accelerated the manufacturing of plasmid and viral vectors for development of new gene therapies. The growth of gene therapies including those aimed at treating rare and chronic diseases is one of the primary drivers of the viral vectors and plasmid DNA manufacturing market size.
Source: Lonza
|
Current Event |
Description and its Impact |
|
Increasing Launch of Favorable Regulatory Policies |
|
|
Increasing Government Support Boosting Gene and Cell Therapy Growth |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Viral vector is projected to account for the largest share of viral vectors and plasmid DNA manufacturing in 2026, representing approximately 68.2% of the total volume. Most commercially approved gene therapies rely on viral vectors (AAV, lentivirus). This directly drives large-scale manufacturing demand. Viral vectors have high transduction efficiency (they deliver genes into cells very effectively). More reliable compared to plasmid DNA and non-viral systems. A large share of ongoing clinical trials in gene and cell therapy use viral vectors. This ensures sustained demand in the near future. Viral vectors are widely used in cell therapies like CAR-T especially lentiviral vectors. These therapies are rapidly expanding in oncology. Viral vector-based products have more regulatory approvals and better-defined GMP manufacturing processes. This gives them a maturity advantage. Viral vectors are complex and high-value products. Even smaller volumes generate higher revenue share compared to plasmid DNA. Increasing funding and collaborations are focused on viral vector production capacity expansion. Many CDMOs specialize in viral vector manufacturing. Growing demand of viral vectors in manufacturing precision and targeted therapies has supported the dominance of segment in the market.
Based on therapeutic application, cancer dominate the market, accounting for a significant 47.5% share in 2026, owing to a significant share of gene therapy clinical trials focused on cancer. Due to more trials focused on developing cancer treatments there is higher demand for vector and plasmid manufacturing. Increasing incidence of cancer worldwide is driving the need for innovative treatments. This boosts investments and product development in oncology. Continuous innovation in immune-oncology and Advancements in gene-modified immune cells and oncolytic viral therapies has supported segment dominance. These approaches rely on viral vectors and plasmid DNA. Cancer research receives significant funding from governments and biotech companies. This accelerates R&D and production needs. For instance, in an effort to accelerate advancements in cancer prevention, detection, and treatment, Worldwide Cancer Research announced financing for 26 fresh, cutting-edge research projects in April 2026. These initiatives are spearheaded by "curestarters" (supporters and researchers) who focus on high-risk, high-reward concepts in an effort to eventually discover cures. They are chosen from applications submitted internationally.
To learn more about this report, Request Free Sample
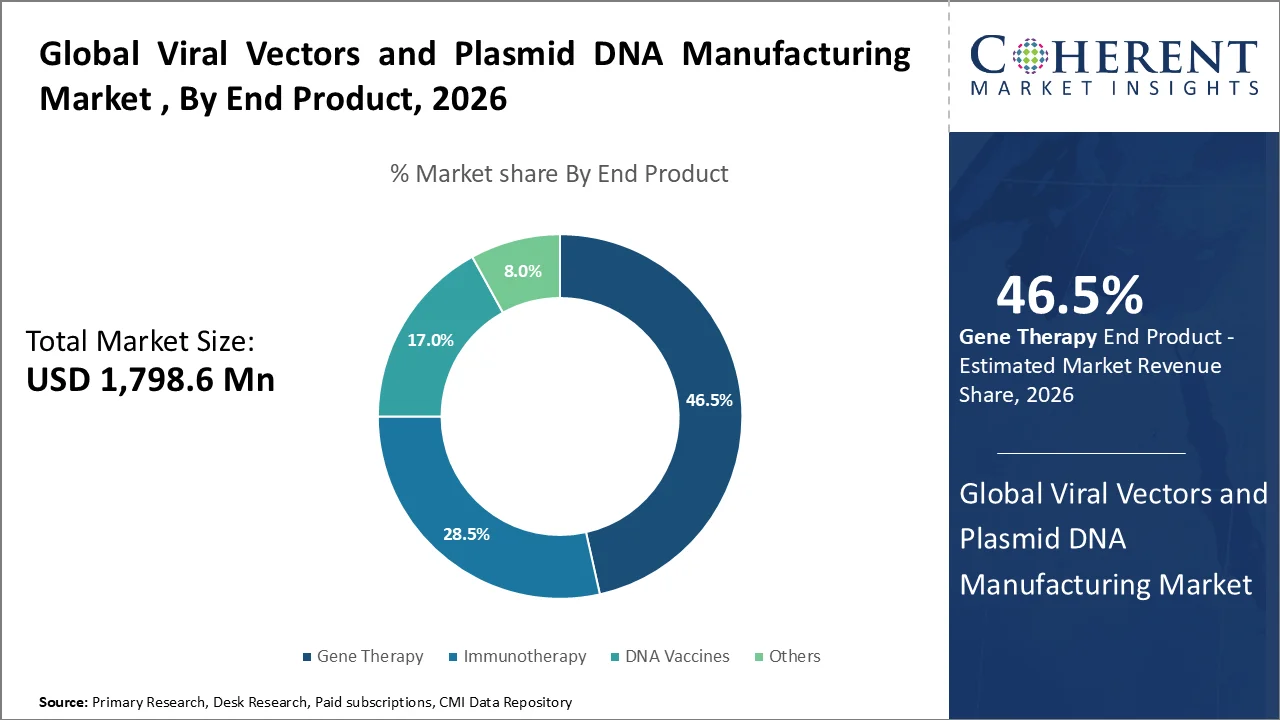
Gene Therapy account for the largest share of 46.5% in 2026 due to high reliance on viral vectors and plasmid DNA and growing number of approved therapies. Gene therapies directly depend on viral vectors (AAV, lentivirus) for gene delivery. Plasmid DNA is also widely used as a starting material in vector production. Increasing approvals of gene therapies for rare genetic disorders and certain cancers. Commercialization drives large-scale manufacturing demand. A large proportion of ongoing clinical trials are focused on gene therapy which ensures continuous and long-term demand.
For instance, in 2026, according to the data published by the National Centers for Biotechnology Information, it has been estimated that with more than 3,900 studies and more than 2,150 treatments under development worldwide as of early 2026, the gene therapy industry is rapidly growing. In 2026, the sector will concentrate on handling U.S.FDA holds on advanced studies and filing INDs for in vivo gene therapies (like Siren Biotechnology). AI integration and decentralized trial models are important trends, and the market for cell and gene therapy is expected to expand dramatically by 2035. Furthermore, in March 2026, the U.S. Food and Drug Administration revealed the authorization of the Kresladi (marnetegragene autotemcel), the first gene therapy for severe Leukocyte Adhesion Deficiency Type I (LAD-I). When there is no human leukocyte antigen (HLA)-matched sibling donor available for an allogeneic hematopoietic stem cell transplant, Kresladi is recommended for the treatment of juvenile patients with severe leukocyte adhesion deficit I (LAD-I) caused by biallelic mutations in ITGB2.
Source: Novartis
Source: Bayer
To learn more about this report, Request Free Sample
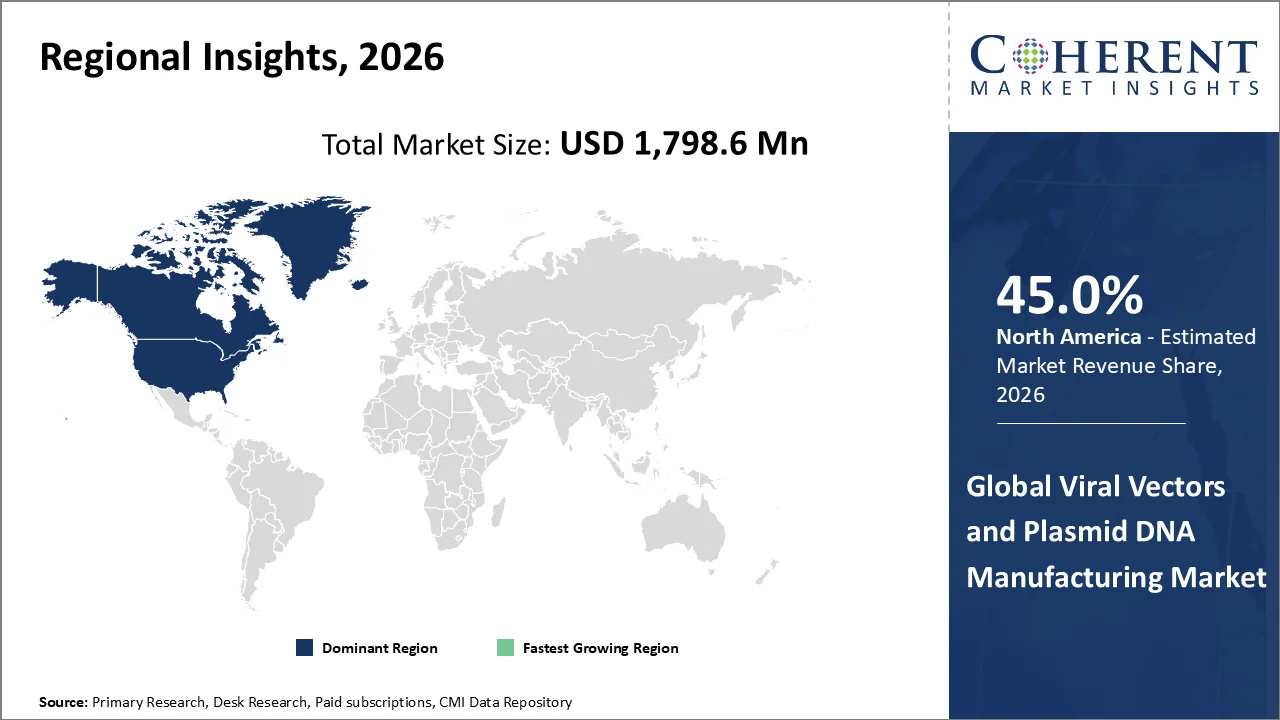
North America account 45.0% market share in 2026, supported by favorable regulatory environment and significant funding & investments. North America is home to major gene therapy developers and vector manufacturers which creates high and consistent demand for viral vectors and plasmid DNA. Advanced healthcare and research infrastructure in North America and well-established biopharma ecosystem, GMP facilities, and research labs enables large-scale and high-quality manufacturing. The North America region (especially U.S.) conducts a large share of global gene therapy clinical trials. Regulatory bodies provide clear guidelines and fast-track approvals for advanced therapies which encourages faster development and commercialization. For instance, in April 2025, Thermo Fisher Scientific Inc., science and clinical research company, unveiled that the company will invest an additional USD 2 billion in the U.S. over next four years, boosting American manufacturing, innovation, and economic competitiveness in the life sciences industry. Thermo Fisher, one of the biggest producers of pharmaceuticals worldwide, makes it possible for biopharmaceutical businesses to create and manufacture their products in the United States. A robust U.S. healthcare supply chain is supported by these new investments in the company's American manufacturing capabilities, which will have a significant multiplier effect throughout the American economy. Over the following four years, the USD 2 billion investment consists of:
USD 1.5 billion in capital expenditures to improve and grow American manufacturing operations
US$500 million in high-impact innovation-focused research and development.
The Asia-Pacific region is poised to be as the fastest-growing region through 2026-2033, owing to rising prevalence of chronic and genetic diseases. Increasing cases of genetic disorders, cancer, and infectious diseases has driven the viral vectors and plasmid DNA manufacturing market demand in the Asia Pacific region. For instance, in 2026, according to the data published by the National Center for Biotechnology Information, over 9.8 million people in Asia are diagnosed with cancer each year, making it the most common disease in the Asia Pacific region. With 49% of all cancer cases worldwide, Asia has the highest cancer burden, highlighting the region's rising workforce and public health issues.
Furthermore, government bodies in Asia Pacific region are promoting biotech through regulatory reforms, funding programs, and national missions (e.g., biopharma initiatives). These policies accelerate manufacturing and innovation. For instance, in February 2026, according to the data published by the Press Information Bureau, nodal agency of Government of India it has been explained that the India's approach to pharmaceuticals has undergone a significant change with the Union Budget 2026–2027, which places biopharma and biologic medicines at the core of its production and healthcare strategy. This is in line with the Indian government's goal of making India a major player in the global biopharmaceutical industry and gaining 5% of the world market. launch of Biopharma SHAKTI, a committed national project to improve India's end-to-end biologics and biosimilars ecosystem that will cost Rs. 10,000 crores over five years. The initiative's goals are to increase India's competitiveness in international biologics supply chains, lessen reliance on imports, and boost domestic discovery and production of high-value biopharmaceutical goods and medications. Three new National Institutes of Pharmaceutical Education and Research (NIPERs) have been established, and seven existing NIPERs have been upgraded, strengthening and expanding the Biopharma-focused network. This proposal aims to address the increasing need for highly specialized human resources in the manufacturing, research, development, and regulation of biopharmaceuticals.
The U.S. viral vectors and plasmid DNA manufacturing market is undergoing a period of robust expansion. The U.S. country has leading universities and research centers actively working on molecular biology and gene editing which fuels continuous innovation. Moreover, the country has large number of specialized contract manufacturing organizations which supports outsourcing and scalability. Strong financial support from government agencies in U.S. like NIH, venture capital, and private biotech firms drives innovation and capacity expansion. For instance, in February 2026, the Platform Vector Gene Therapy (PaVe-GT) initiative, which aims to increase the effectiveness of rare-disease gene therapy development by utilizing the same AAV delivery system and manufacturing techniques across multiple programs, was highlighted by the National Center for Advancing Translational Sciences (NCATS) of the National Institutes of Health. Additionally, NCATS claims to be involved in the leadership of the Bespoke Gene Therapy Consortium (BGTC), a public-private collaboration aimed at creating standards and platforms to expedite the creation and distribution of gene therapies. Simultaneously, the U.S. FDA said in January 2026 that its adaptable strategy to chemistry, manufacturing, and control (CMC) regulations for gene and cell therapies has aided in accelerating product development and is anticipated to continue facilitating quicker commercialization. These advancements demonstrate how robust U.S. research institutions, government-sponsored initiatives, and regulatory backing are driving innovation and scale production in plasmid DNA and viral vectors.
The China viral vectors and plasmid DNA manufacturing market projected to grow significantly during the upcoming years. China has a fast-growing biotech and pharmaceutical sector. Increasing number of companies involved in gene therapy and vector production. Rapid growth of Contract Development and Manufacturing Organizations - CDMOs strengthens the supply chain and production capabilities. Moreover, China has lower operational and labor costs compared to Western countries which attracts both international and domestic outsourcing. For instance, in March 2026, AstraZeneca plc, pharmaceutical company, unveiled a USD 15 billion investment plan in China through 2030 to increase its capacity for production, research, and development there. This strategy includes building a cell therapy manufacturing and supply base and innovation center in Shanghai. This facility will boost China's biotech supply chain and local manufacturing capability for complicated biologics and gene-based treatments by supporting the production of cutting-edge therapeutic products including CAR-T cell therapies.
Lonza Group AG, FinVector Vision Therapies, Cobra Biologics and Pharmaceutical Services, Sigma-Aldrich Co. LLC, VGXI, Inc., VIROVEK, SIRION Biotech GmbH, FUJIFILM Diosynth Biotechnologies U.S.A., Inc., Sanofi, Cell and Gene Therapy Catapult, Brammer Bio, and MassBiologics.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,798.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 23% | 2033 Value Projection: | USD 7,571.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Lonza Group AG, FinVector Vision Therapies, Cobra Biologics and Pharmaceutical Services, Sigma-Aldrich Co. LLC, VGXI, Inc., VIROVEK, SIRION Biotech GmbH, FUJIFILM Diosynth Biotechnologies U.S.A., Inc., Sanofi, Cell and Gene Therapy Catapult, Brammer Bio, and MassBiologics. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients