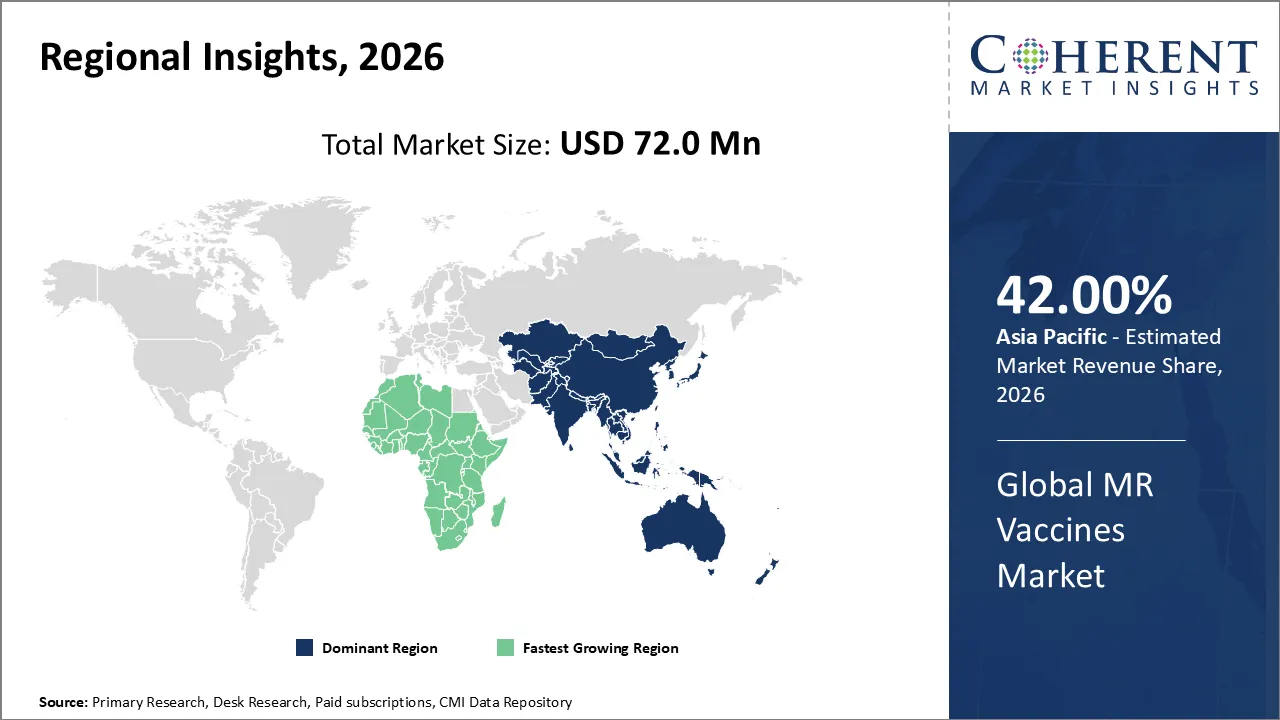
The MR vaccines market is estimated to be valued at USD 72.0 Mn in 2026 and expected to reach USD 101.6 Mn by 2033, witnessing a CAGR of 2.1% over the forecast period (2026-2033).
The measles and rubella (MR) vaccine, which protects against measles and rubella, is recommended by the Centers for Disease Control and Prevention (CDC). MR vaccine should be given to children in two doses, the first at 12 to 15 months of age and the second at 4 to 6 years of age. Measles is a highly contagious virus that continues to be one of the leading vaccine-preventable causes of childhood death. High fever and severe skin rash are among the symptoms of measles.
Rubella is a virus-borne infectious disease. Rubella causes a mild sickness in most people, with symptoms such as a low-grade fever, sore throat, and a rash that begins on the face and spreads to the rest of the body. The majority of people who are vaccinated against measles and rubella (MR) will be protected for the rest of their lives. Vaccines and high immunization rates have made these diseases much less common in the U.S.
|
Current Event |
Description and its Impact |
|
Regulatory Harmonization and Market Access Changes |
|
|
Climate Change and Disease Pattern Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Indicator |
Data (2025–26) |
Notes |
|
Contagiousness (R₀) |
12–18 |
One of the highest among infectious diseases; a single case can infect up to 18 unvaccinated individuals. |
|
Global Measles Deaths |
>95% in low-income countries |
Mortality concentrated where health systems are weakest. |
|
India (Jan–Mar 2025) |
332 districts reported zero measles cases; 487 districts reported zero rubella cases |
Reflects progress under the National Zero MR Elimination Campaign. |
|
India MR Vaccine Coverage (2024–25) |
93.7% (first dose), 92.2% (second dose) |
High coverage but still short of 100% elimination target. |
|
United States (Feb 2026) |
1,136 confirmed measles cases across 28 jurisdictions |
Shows resurgence despite high-income setting, linked to vaccine hesitancy. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of distribution channel, the public segment is expected to lead the market with 82.7% share in 2026. Government-led vaccination campaigns, subsidized immunization programs, and international procurement systems that make sure everyone has equal access are what make this system work. Private channels, on the other hand, are less important and only serve urban and higher-income populations.
For instance, in February 2026, Noida started a vaccination campaign for schoolchildren in Classes 1 to 5 against measles and rubella. The campaign lasted ten days and aimed to make sure that everyone could get free and safe vaccines, prevent outbreaks, and improve public health by making sure that all government and private schools had a lot of immunization coverage.
In terms of vaccine brand, the MR vaccine segment is expected to hold 68.4% share of the market in 2026, because it is used in national immunization programs, is consistently bought by global health organizations, and is more widely available than MRBEV, which is still mostly used in one region.
For instance, in May 2025, The National Zero Measles-Rubella Elimination Campaign will start during World Immunization Week 2025, according to the Ministry of Health and Family Welfare. The goal of the program is to give everyone two doses of the MR vaccine so that everyone is fully protected. India wants to get rid of measles and rubella completely by 2026.
To learn more about this report, Request Free Sample
Asia Pacific is expected to dominate the MR vaccines market with 42% share in 2026, because of the region's large population, frequent outbreaks of measles and rubella, and strong government-supported vaccination programs. Support from WHO and UNICEF speeds up adoption even more. A greater amount spent on healthcare and campaigns to raise awareness make sure that Asia-Pacific stays the main place where vaccines are needed.
For instance, in October 2025, Malaysia's Ministry of Health announced a nationwide campaign to vaccinate against measles and rubella. The program gives free vaccines to kids between the ages of six and 59 months. Its goals are to raise coverage above 95 percent, strengthen herd immunity, and help the country reach its goal of getting rid of measles and rubella by 2030.
Africa is expected to exhibit the fastest growth, because there are a frequent measles and rubella outbreaks, not enough people getting vaccinated, and a high rate of people getting sick. Strong immunization campaigns backed by WHO and UNICEF, along with government programs and international funding, lead to widespread use. This makes Africa the second-largest and fastest-growing market.
India's demand for the MR (Measles-Rubella) vaccine is growing in 2026 because of government immunization campaigns, high rates of measles and rubella, and WHO-backed goals to eliminate the diseases. More healthcare infrastructure, local vaccine production, and public awareness campaigns all help the market grow even more, making MR vaccines a top public health priority.
For instance, in April 2026, India has started the National Zero Measles-Rubella Elimination Campaign 2025–26 to make sure that everyone is fully immunized. Children will get two doses of the MR vaccine as part of the Universal Immunisation Programme. The goal of the campaign is to get rid of measles and rubella in all states and union territories by 2026.
In 2026, Nigeria's demand for the MR (Measles-Rubella) vaccine is high due to ongoing measles outbreaks, WHO-supported goals for eliminating the disease, and UNICEF-supported programs to buy the vaccine. More immunization campaigns, more awareness of public health, and more growth in regional markets in Africa all increase demand. This makes MR vaccines extremely significant for Nigeria's disease control strategy.
For instance, in January 2026, In Lagos State, Nigeria, a campaign to vaccinate 10.5 million children between the ages of 9 and 59 months against measles and rubella has begun. The Lagos State Government is in charge of the program, which aims to lower the number of diseases that can be avoided with vaccines and increase the number of people who get vaccinated across the state by widespread MR vaccine administration.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 72.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.1% | 2033 Value Projection: | USD 101.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
PT Bio Farma, Bio-Manguinhos, BE Vaccines, Serum Institute of India, and Crucell Switzerland AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Government-led immunization campaigns are a cornerstone of the MR Vaccines Market growth. Many low- and middle-income countries have launched nationwide vaccination drives, often supported by WHO and UNICEF, to eliminate measles and rubella. These initiatives not only increase vaccine accessibility but also strengthen healthcare infrastructure. By prioritizing immunization in public health agendas, governments create sustained demand for MR vaccines, ensuring that populations at risk are protected and that global eradication goals remain achievable.
The persistent prevalence of measles and rubella continues to fuel MR Vaccines Market demand. Outbreaks in Africa and Asia highlight the vulnerability of regions with low vaccination coverage, underscoring the urgent need for widespread immunization. The high morbidity and mortality associated with these diseases drive public health authorities to intensify vaccination efforts. As awareness grows and healthcare systems respond to outbreaks, the demand for MR vaccines rises, making them essential tools in reducing disease burden and preventing future epidemics.
New advances in vaccine technology are opening up new possibilities for the MR Vaccines Market forecast. Thermostable formulations allow vaccines to work without strict cold-chain requirements, reducing logistical barriers in remote and resource-limited regions. Combination vaccines, which protect against both measles and rubella in a single dose, make immunization schedules easier to follow and more likely to be followed. These improvements not only make vaccines easier to get, but they also make global immunization programs stronger, which means that MR vaccines will be in higher demand in both developed and emerging markets.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients