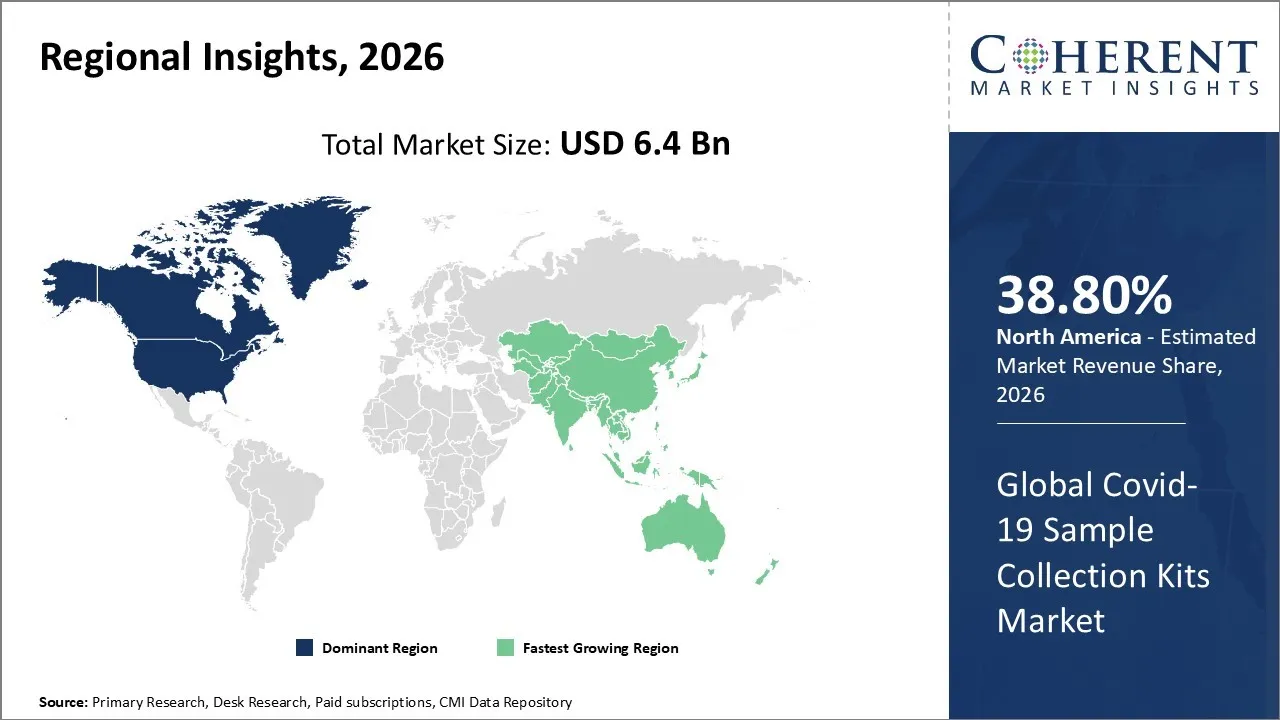
The Covid-19 Sample Collection Kits Market is estimated to be valued at USD 6.4 Bn in 2026 and is expected to reach USD 12.3 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 17.3% from 2026 to 2033.
The global need for accurate and large-scale testing drove the rapid expansion of the COVID-19 Sample Collection Kits Market during the pandemic. These kits typically include swabs, viral transport media, and collection tubes, which healthcare professionals use to collect respiratory samples for diagnostic procedures. Hospitals, diagnostic laboratories, and dedicated testing centers increased their demand for these kits. Governments and healthcare organizations also invested heavily in testing infrastructure, further driving market growth. Ongoing efforts in infectious disease surveillance and preparedness for future outbreaks continue to sustain the demand for reliable sample collection solutions.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Innovations and Digital Integration |
|
|
Epidemiological Trends and Public Health Responses |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Diagnostic expected to hold largest market share of 52.4% in 2026. The increasing need for timely and accurate disease detection drives the growth of the diagnostic segment in the COVID-19 Sample Collection Kits Market. Diagnostic laboratories and healthcare facilities use properly collected specimens to conduct molecular and antigen tests. The expansion of testing capabilities and improvements in laboratory infrastructure have increased the demand for reliable sample collection kits. Healthcare systems also conduct continuous surveillance of infectious diseases and adopt advanced diagnostic technologies, which encourages the consistent use of standardized sample collection systems in clinical and public health environments. For instance, Minister of State for Education Shri Sanjay Dhotre launched a COVID-19 Rapid Antigen Test kit developed by IIT Delhi researchers led by Dr. Harpal Singh of the Centre for Biomedical Engineering.
Swabs hold the largest market share of 42.2% in 2026. The need for reliable specimen collection for diagnostic testing significantly increased the demand for swabs in the COVID-19 Sample Collection Kits Market. Healthcare professionals used nasopharyngeal and oropharyngeal swabs to collect accurate respiratory samples for laboratory analysis. The expansion of testing centers, hospitals, and diagnostic laboratories further increased the use of these products. Manufacturers also improved swab materials and designs to enhance sample absorption and release efficiency. Ongoing monitoring of respiratory infections and preparation for future outbreaks continue to support the demand for medical swabs. For instance, Amazon launched an at-home COVID-19 test kit, letting customers quickly and easily collect samples using gentle lower nasal swabs, with free Amazon Prime delivery.
COVID-19 Testing Centers acquired the prominent market share of 38.4% in 2026. COVID-19 testing centers have strongly driven the sample collection kits market by serving as primary locations for large-scale specimen collection. Hospitals, drive-through sites, and community testing facilities use swabs, transport media, and collection tubes to handle high testing volumes efficiently. Government initiatives and public health programs have expanded these centers, while mobile and temporary facilities have increased testing accessibility. Rising public awareness and the demand for rapid, reliable testing continue to sustain the use of sample collection kits at these centers. For instance, Genomics company Mapmygenome opened a drive-through COVID-19 testing facility yesterday at Cyient Foundation MP Primary and ZP High School in Gachibowli, Hyderabad, allowing individuals and families to park and get tested on-site.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. Widespread testing and strong healthcare infrastructure have driven notable growth in North America’s COVID-19 Sample Collection Kits Market. Hospitals, diagnostic laboratories, and dedicated testing centers actively use swabs, viral transport media, and collection tubes to obtain accurate specimens. Innovations in kit design and the growing adoption of home-testing solutions continue to shape market trends. Governments and public health programs have promoted large-scale testing, while ongoing efforts to prepare for infectious disease outbreaks sustain demand and encourage continuous advancements in sample collection technologies. For instance, Biogen Inc., Broad Institute of MIT and Harvard, and Partners HealthCare formed a consortium to create a COVID-19 biobank, enabling scientists to study de-identified biological and medical data to advance vaccines and treatments.
Rising testing demand and strengthening healthcare infrastructure have driven rapid growth in the Asia Pacific COVID-19 Sample Collection Kits Market. Countries actively expanded hospitals, diagnostic laboratories, and community testing centers to enhance early infection detection. Healthcare providers and consumers increasingly use innovative swab designs and user-friendly kits for both clinical and home-based testing, shaping market dynamics. Governments implement mass testing campaigns, public awareness programs, and invest in laboratory capacity, while ongoing preparedness for future outbreaks sustains demand and promotes continuous development of reliable sample collection solutions. For instance, Dr. VK Saraswat, NITI Aayog member and President of SCTIMST, announced the commercial launch of Agappe Chitra Magna, a magnetic nanoparticle-based RNA extraction kit for COVID-19 testing, in a program attended virtually by Prof. Ashutosh Sharma, Dr. Asha Kishore, Dr. HK Varma, and SCTIMST scientists.
Extensive testing programs and a well-established healthcare infrastructure have driven significant growth in the United States COVID-19 Sample Collection Kits Market. Hospitals, diagnostic laboratories, and testing centers actively use swabs, viral transport media, and collection tubes to collect accurate specimens. Innovations in kit design and the expansion of at-home testing solutions continue to shape market developments. Federal and state government initiatives, together with public health campaigns, promote widespread testing, while ongoing monitoring of infectious diseases encourages healthcare providers to adopt advanced, reliable sample collection kits in both clinical and community settings. For instance, Quest Diagnostics launched an at-home COVID-19 rapid antigen test for purchase through its consumer platform, QuestDirect.
Large-scale testing initiatives and strong government support for public health have driven rapid growth in China’s COVID-19 Sample Collection Kits Market. Hospitals, diagnostic laboratories, and community testing centers actively collect accurate specimens using swabs, viral transport media, and collection tubes. Healthcare providers increasingly adopt innovative collection kits and streamlined testing processes to improve efficiency. Government-led mass testing campaigns, investments in laboratory infrastructure, and rising public awareness continue to boost demand, while ongoing preparedness for infectious disease outbreaks encourages the development and widespread use of reliable sample collection solutions.
The market is moving toward home-testing solutions, allowing individuals to collect samples safely and conveniently. Self-collection kits include swabs, transport tubes, and clear instructions to ensure accurate specimen collection without professional supervision. This trend reduces hospital congestion, supports telehealth initiatives, and enhances accessibility in remote or high-risk areas. Manufacturers are innovating user-friendly designs to increase adoption, making home testing a sustainable part of ongoing COVID-19 and infectious disease management.
Advancements in swab design, such as flocked tips and flexible shafts, improve sample absorption and patient comfort. Viral transport media and preservative solutions are also being enhanced for longer stability and safer handling. These innovations increase the reliability of diagnostic results, reduce sample degradation, and support broader adoption in hospitals, laboratories, and testing centers. Continuous research and material improvements remain a key trend to meet evolving testing requirements.
Rising consumer preference for convenient and safe testing creates opportunities for self-collection kits. Companies can design user-friendly kits with clear instructions, pre-labeled tubes, and secure packaging for accurate home specimen collection. Expanding telehealth services and remote diagnostics further drive this opportunity. By offering kits compatible with both PCR and rapid antigen tests, manufacturers can tap into growing demand for accessible testing solutions beyond clinical settings, improving reach in urban and remote communities.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.4 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 17.3% | 2033 Value Projection: | USD 12.3 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
MyLab Discovery Solutions, Bhat Bio-Tech, Camtech Diagnostics, Angstrom Biotech Pvt. Ltd., Medsource Ozone Biomedicals Pvt Ltd., Neuromics, Oscar Medicare Pvt. Ltd., GenBody Inc., Alimetrix, Binx Health, Clinical Enterprise, Bio-Rad Laboratories, Ethos Laboratories, GetMyDNA, Kroger, LabCorp, RapidRona, EKF Diagnostics, Everlywell, Inc., and Copan Diagnostics. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients