COVID-19 (Coronavirus Disease) is caused by the novel coronavirus SARS-CoV-2, which is a RNA virus. Few of the common symptoms of COVID-19 include fever, dry cough, fatigue, and body ache. COVID-19 disease can be transmitted from one person to another by the inhalation of infectious droplets. People with conditions such as hypertension, cancer, diabetes, old age, and others are at higher risk of getting severe COVID-19 infection. Thus, it is essential to develop efficient nasal swab tests for diagnosis of COVID-19. Rapid research is being carried out to make low cost and accurate nasal swab COVID-19 tests, which is expected to create growth opportunities for this market. Nasal swab tests involve the collection of secretions from the inner surface of nostril. These samples are employed by diagnostic systems such as molecular diagnostic, biosensors, immunoassays, and others. COVID-19 nasal swab tests are widely used by hospitals, clinics, laboratories, diagnostic centers and others.
Global COVID-19 Nasal Swab Test Market - Impact of the Coronavirus (COVID-19) Pandemic
Coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19, as pandemic on March 11, 2020.
According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 186,240,393 cases and 4,027,861 deaths due to coronavirus disease (COVID-19) were reported till July 11, 2021, across the globe.
Impact of COVID-19 on Demand and Supply of COVID-19 Nasal Swab Test
The COVID-19 pandemic and lockdown in various countries across the globe have negatively impacted the financial status of businesses across all sectors. The COVID-19 pandemic has impacted the entire supply chain of the biotechnology industry mainly due to strict lockdown in several regions. However, the demand for COVID-19 nasal swab test is increasing in this COVID-19 pandemic due to the growing need of diagnostics for the proper detection of COVID-19 infection and to stop its spread.
For instance, in May 2020, Aga Khan University’s Innovation Lab, Pakistan, developed an indigenous, low-cost, 3-D printed prototype of nasal swab for the detection of SARS-CoV-2. This new development would aid the country in meeting the growing demand for innovative diagnostics for COVID-19 amidst surge in coronavirus cases. In November 2020, the clinical trials for this 3-D printed nasal swab were completed, and had shown positive results for the safety and efficacy of these 3 D printed nasal swabs.
Moreover, in September 2020, the University of Melbourne, Australia, and 3D MEDiTech, an Australian medical devices company, accelerated the production of the first indigenous 3-D printed nasal swab for diagnosis of COVID-19. According to clinical trial data published by the Medical Journal of Australia in May 2020, these newly developed 3D printed nasal swabs were found to be at par with commercial nasal swabs. 3D printed nasal swabs would aid in providing a cost effective solution amidst shortage of nasal swabs due to rapid increase in laboratory testing for COVID-19.
Thus, impact of the coronavirus (COVID-19) pandemic is expected to drive the growth of the global COVID-19 nasal swab test market during the forecast period, owing to rising number of COVID-19 cases and rapid research in development of diagnostics for COVID-19 during the pandemic.
The global COVID-19 nasal swab test market is estimated to be valued at US$ 111.3 Mn in 2021, and is expected to exhibit a CAGR of 2.6% over the forecast period (2021-2028).
COVID-19 Nasal Swab Test Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 111.3 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2020 to 2027 CAGR: | 2.6% | 2028 Value Projection: | US$ 133.2 Million |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Roche Holding AG, Becton Dickinson & Company, 3M Corporation, BioMerieux SA, Lucira Health, Mesa Biotech, Inc.,Siemens Healthineers AG, QIAGEN N.V., Cepheid, LumiraDx Ltd., Fluidigm Corporation, Quidel Corporation, GenMark Diagnostics, Inc., Sherlock Biosciences, PrivaPath Diagnostics Limited, and OraSure Technologies, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 1: Global COVID-19 Nasal Swab Test Market Share (%) Analysis, By Diagnostics Type, 2021
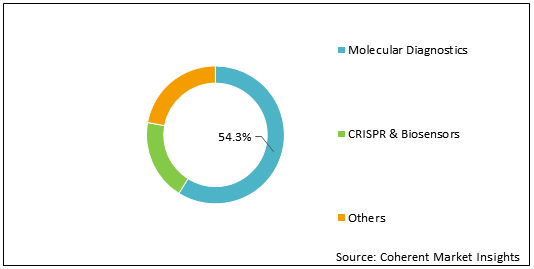
To learn more about this report, Request Free Sample
The increasing number of novel coronavirus cases is the major factor that is expected to drive the market growth over the forecast period.
The rising number of novel coronavirus cases is expected to drive growth of COVID-19 nasal swab test market over the forecast period. According to an article published by Healthline Media, an American medical information provider, on June 28, 2021, more than 70,000 new COVID-19 cases are added every week in U.S.
Moreover, increasing number of research and development activities for improving the effectiveness of COVID-19 nasal swab test is expected to aid in the growth of the market over the forecast period. For instance, according to an article published by the National Institutes of Health (NIH), U.S. in July 2020, Helix OpCo, LLC, a California based population genomic company, developed standardized nasal swab kits in bulk for the collection of nasal secretion samples to detect SARS-CoV-2. This will allow processing of thousands of nasal swab samples at once and provide results within 24-48 hours. These COVID-19 nasal swab kits would be shipped to public health departments, employers, healthcare systems, and others.
Rising government funding may support research and development activities, which in turn may support the research and development of new COVID-19 nasal swab tests, which is expected to drive the growth of the global COVID-19 nasal swab tests market over the forecast period. For instance, according to an article published by the National Institutes of Health (NIH), U.S. in July 2020, NIH plans to invest around US$ 248.7 million for making innovative diagnostics solutions for COVID-19.
Global COVID-19 Nasal Swab Test Market – Restraints
However, growth of the COVID-19 nasal swab test market may be hampered, owing to the increasing product recalls. For instance, in May 2020, rapid COVID-19 diagnostic kits manufactured by Spartan Bioscience, Inc., a Canadian biotechnology company, were recalled by Health Canada (department of Government of Canada) due to efficacy concerns of nasal/nasopharyngeal swabs. Thus, Health Canada directed the use of this diagnostic kit for research purposes only while its regulatory approval has been put on hold.
Global COVID-19 Nasal Swab Test Market – Regional Analysis
On the basis of region, the global COVID-19 nasal swab test market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa.
North America is expected to hold dominant position in the global COVID-19 nasal swab test market over the forecast period, owing to rising number of product launches. For instance, in March 2021, Roche Diagnostics, a part of the Swiss pharmaceutical corporation Roche Holding AG, launched a new rapid antigen COVID-19 nasal swab test in partnership with SD Biosensor Inc., a Korean technology solutions provider. This COVID-19 diagnostic kit is a less invasive nasal test that provides results within 15 minutes.
Key players are focused on marketing strategies such as partnerships, collaborations, and others, which is expected to drive growth of the COVID-19 nasal swab test market. For instance, in April 2020, Accumen Inc., an American healthcare company, entered into a partnership with EnvisionTec, a German technology company, to produce 3D printed nasal swabs for diagnosis of COVID-19.
Figure 2: Global COVID-19 Nasal Swab Test Market Value (US$ 39.5 Mn), by Region, 2021
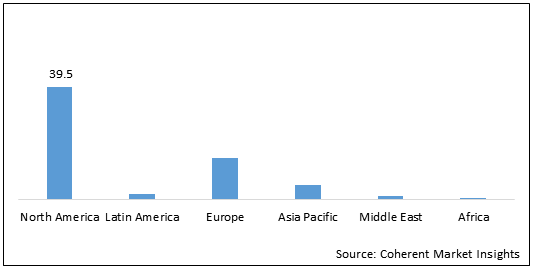
To learn more about this report, Request Free Sample
Global COVID-19 Nasal Swab Test Market – Competitive Landscape
Major players operating in the global COVID-19 nasal swab test market include Abbott Laboratories, Roche Holding AG, Becton Dickinson & Company, 3M Corporation, BioMerieux SA, Lucira Health, Mesa Biotech, Inc.,Siemens Healthineers AG, QIAGEN N.V., Cepheid, LumiraDx Ltd., Fluidigm Corporation, Quidel Corporation, GenMark Diagnostics, Inc., Sherlock Biosciences, PrivaPath Diagnostics Limited, and OraSure Technologies, Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients