Ankylosing spondylitis (AS) is a rare type of inflammation that mainly affects spine and can damage large joints of body. It causes inflammation in the spinal joints (vertebrae), thereby causing severe pain and discomfort. Over time, ankylosing spondylitis can cause some bones in the spine to fuse together, as a result of which the spine becomes less flexible. Symptoms of ankylosing spondylitis include pain and stiffness in the hips and lower back.
The global ankylosing spondylitis market is estimated to be valued at US$ 4,864.3 million in 2021 and is expected to exhibit a CAGR of 8.2% during the forecast period (2021-2028).
Figure 1.Global Ankylosing Spondylitis Market Share (%) in Terms of Value, by Drug Type, 2021
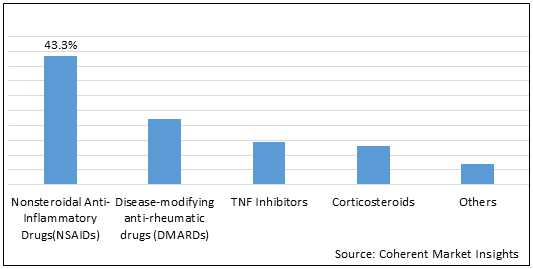
To learn more about this report, Request Free Sample
Increasing prevalence of ankylosing spondylitis is expected to drive the market growth during the forecast period.
The increasing prevalence of ankylosing spondylitis is expected to drive the global ankylosis spondylitis market growth during the forecast period. For instance, according to an article published by ‘Frontiers in Medicine’ journal in March 2021, the epidemiological data in Taiwan stated that the prevalence of ankylosing spondylitis is 96.9 per 100,000 people.
Ankylosing Spondylitis Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 4,864.3 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 8.2% | 2028 Value Projection: | US$ 8,460.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Amgen Inc., AbbVie Inc., Pfizer Inc., Novartis AG, Bristol-Myers Squibb Company, CELGENE CORPORATION, Eli Lilly and Company, Cipla Inc., Emcure Pharmaceuticals, Torrent Pharmaceuticals Ltd., UCB S.A., Suzhou Zelgen Biopharmaceuticals Co., Ltd., HENGRUI USA, and Johnson & Johnson Services, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Ankylosing Spondylitis Market Share (%), by Distribution Channel, 2021
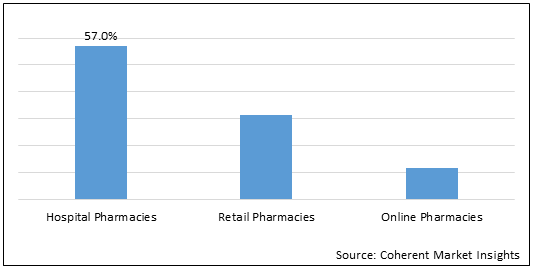
To learn more about this report, Request Free Sample
Increasing strategic alliances between the companies to develop or to expand the availability of drugs is expected to drive the market growth during the forecast period.
Key players in the market are focusing on inorganic growth strategies, such as partnerships and collaborations which is expected to drive the market growth during the forecast period.
For instance, in August 2021, Inmagene Biopharmaceuticals, a leading biotech company focused on the development of therapeutics for immunology-related diseases, and Affibody AB, a company developing innovative bi- and multi-specific next-generation biopharmaceuticals, working in a partnership, announced the successful dosing of the first patient in the global ASPIRE phase 2 trial evaluating izokibep (IMG-020 or ABY-035) for treating ankylosing spondylitis (AS).
Global Ankylosing Spondylitis Market – Impact of Coronavirus (COVID-19) Pandemic
Accessing medical services can be challenging for people with ankylosing spondylitis during the COVID-19 pandemic. While some drugs for ankylosing spondylitis can be self-administered, some are given through an IV in hospitals. Similarly, physical therapy requires a visit to the physical therapist, which has been difficult due to the COVID-19 pandemic.
Biologics such as TNF (Tumor Necrosis factor) inhibitors, used in ankylosing spondylitis treatment, may increase the risk of infections and lower the body's ability to fight infections, including COVID-19, because these medications cause a suppressing effect on the immune system. Biologics including IL-17 and IL-12/23 inhibitors are used for treating ankylosing spondylitis, but have the same immunosuppressive effect that may increase the risk of infections. Thus, due to this, the growth of the global ankylosing spondylitis market is expected to be hampered.
Ankylosing spondylitis is an immune-mediated inflammatory disease (IMID). According to a study conducted in Ontario, Canada, in 2020, it was found that people with IMIDs were tested for COVID-19 at a 20% higher rate, as compared to the general population.
Global Ankylosing Spondylitis Market: Restraint
The major factors that are expected to hinder growth of the global ankylosing spondylitis market include high cost of treatment and lack of skilled surgeons and physiotherapists.
Key Players
Major players operating in the global ankylosing spondylitis market include Amgen Inc., AbbVie Inc., Pfizer Inc., Novartis AG, Bristol-Myers Squibb Company, CELGENE CORPORATION, Eli Lilly and Company, Cipla Inc., Emcure Pharmaceuticals, Torrent Pharmaceuticals Ltd., UCB S.A., Suzhou Zelgen Biopharmaceuticals Co., Ltd., HENGRUI USA, and Johnson & Johnson Services, Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients