Atherectomy Devices Market Insights
Atherectomy devices are indicated for removing plaque and thrombus from diseased arteries. Atherectomy devices are used in effective treatment of both coronary and peripheral arterial disease. Atherectomy devices are designed to either cut, shave, sand, or vaporize these plaques. There are mainly four types of atherectomy devices: Directional atherectomy, Rotational atherectomy, Laser atherectomy, and Orbital atherectomy. Directional atherectomy catheter have a cutting tip that shaves the plaque from blood vessels. It is used only for peripheral blood vessels. Rotational atherectomy have cutting tip, which spins at high speed and grinds the plaque into a powder. In Laser atherectomy, the catheter tip has a device that emits high-energy laser, which vaporize the plaque material. Although orbital atherectomy uses high-speed rotational device similar to the rotational atherectomy, it has different tip.
Frequent approval of innovative atherectomy devices is expected to drive growth of global atherectomy devices market in near future
Frequent approval from different regulatory bodies to new atherectomy devices in key regions such as North America and Europe is expected to drive growth of this market. In recent past, innovative atherectomy devices received U.S. FDA approval and CE marking. For instance, in 2016, Medtronic Plc. received FDA clearance of new HawkOne 6F directional atherectomy system. In 2017, Avinger Inc. received FDA 510(k) clearance for new modified version of Pantheris Lumivascular atherectomy system. Company has modified design of previous Pantheris Lumivascular atherectomy system in order to enhance its performance. In 2017, Cardiovascular Systems, Inc. received approval for Diamondback 360 coronary orbital atherectomy system in U.S. Increasing adoption for these innovative products is expected to fuel growth of the global atherectomy devices market over the forecast period.
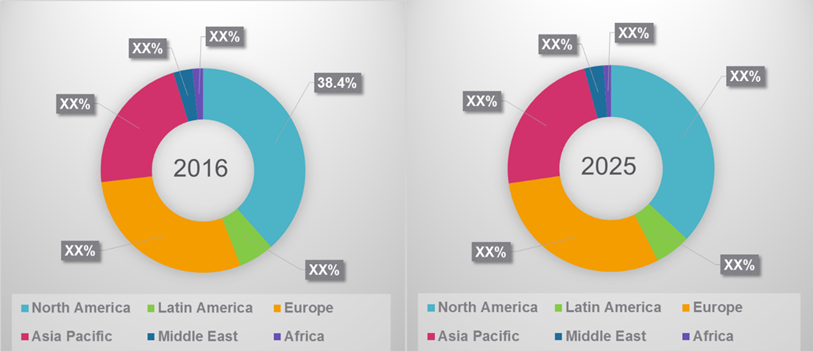
The Global Atherectomy Devices Market was valued at US$ 504.5 million in 2016 and is expected to witness a CAGR of 6.2% over the forecast period (2017 – 2025).
Figure 1. Global Atherectomy Devices Market Share, By Region, 2016 - 2025
To learn more about this report, Request Free Sample
Increasing prevalence of cardiovascular diseases is expected to drive growth of the global atherectomy devices market
Increasing prevalence of cardiovascular diseases (CVDs) is due to use of Tobacco, unhealthy diet, and physical inactivity may lead to increase in demand for atherectomy devices for the treatment of CVDs. For instance, according to research published by World Health Organization (WHO) in 2017, around 17.7 million people died from cardiovascular diseases in 2015, which is around 31% of all global deaths. Among these death around 7.4 million death were due to coronary heart disease and 6.7 million were due to stroke. According to Centers for Disease Control and Prevention in 2017, Coronary heart disease (CHD) is one of the common heart disease, which causes around 370,000 deaths in the U.S. annually. According to study published in Heart Foundation in 2015, cardiovascular diseases affects one in six Australians or 4.2 million. In 2015, around 490,000 peoples in Australia are suffering from cardiovascular diseases.
The presence of alternative devices such as balloon angioplasty may hamper the growth for atherectomy devices market. Balloon angioplasty is also used to widen blocked or narrowed blood vessel same as atherectomy devices.
The industry has witnessed multiple mergers & acquisitions in recent years. For instance, Boston Scientific Corporation acquired the interventional business unit of Bayer AG in 2014. Medtronic plc acquired Covidien plc in 2014 to strengthen its position in global atherectomy devices market.
Key players operating in the global atherectomy devices market include Medtronic plc, Koninklijke Philips N.V., Cardiovascular Systems, Inc., Boston Scientific Corporation, Avinger, Inc, C. R. Bard, Inc, Cardinal Health, Inc., Minnetronix, Inc., Straub Medical AG, and Biotronik.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients