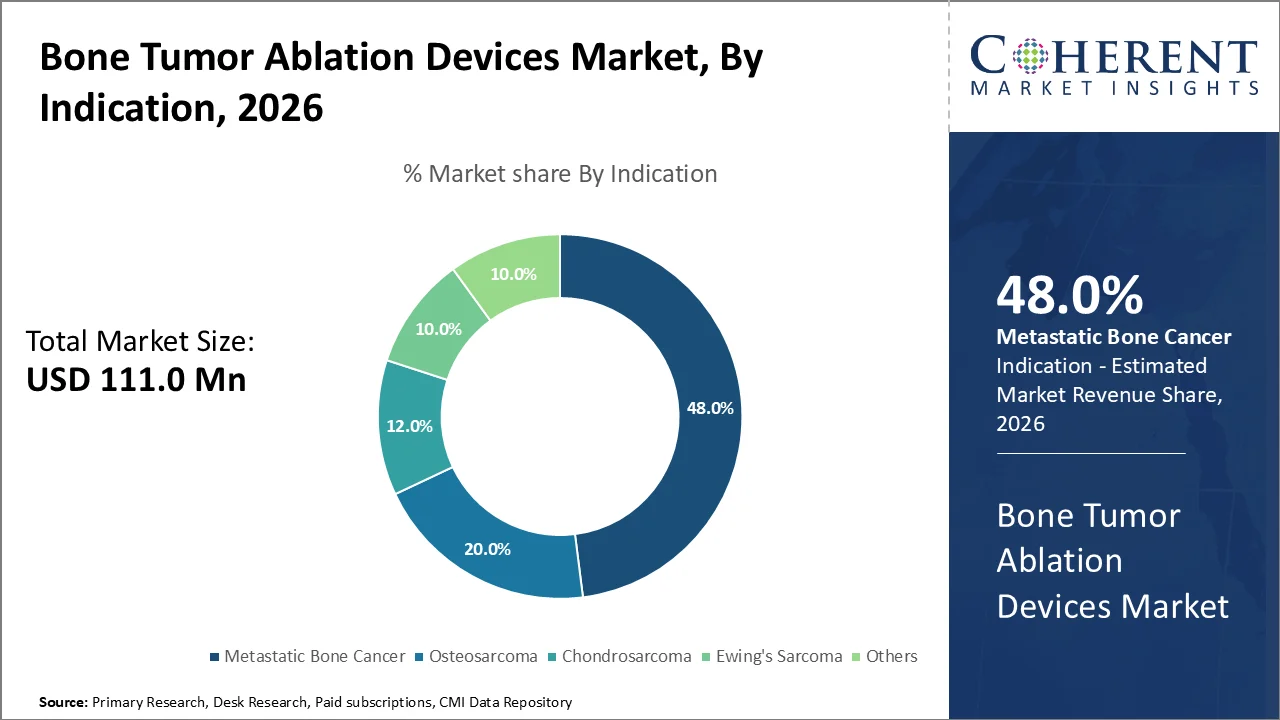
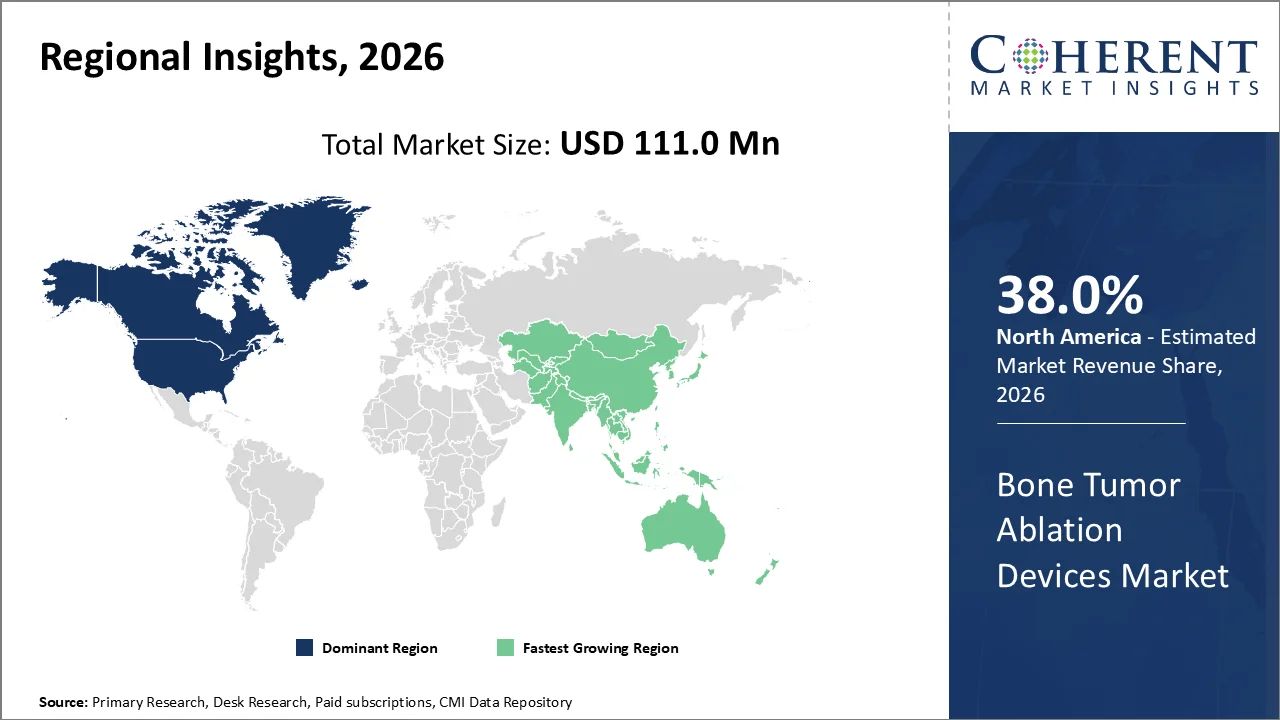
The global bone tumor ablation devices market size is projected to grow from USD 111.0 Mn in 2026 to USD 219.3 Mn by 2033, registering a compound annual growth rate (CAGR) of 10.2% during the forecast period (2026–2033). The key factors that drive this expansion include the growing incidence of bone cancers, metastatic bone conditions, and the increased use of minimally invasive surgeries along with continuous innovations in technologies like radiofrequency ablation, microwave ablation, and cryoablation.
The radiofrequency ablation segment of the market is expected to lead with about 42.5% market share in the forecast period of 2026. Radiofrequency ablation is popular owing to its ease of implementation, cost-effectiveness, and efficacy in treating diseases like osteoid osteoma and bone metastases. The popularity of radiofrequency ablation is attributed to its precise nature and effectiveness in relieving pain among patients affected by bone tumors.
Additionally, increasing adoption of minimally invasive procedures across hospitals and oncology centers is accelerating the use of radiofrequency ablation. Radiofrequency ablation offers a faster recovery period, reduced complications, and economical treatment options, compared to traditional surgery techniques.
In March 2024, Medtronic announced that it received U.S. FDA 510(k) clearance for its OsteoCool 2.0 Bone Tumor Ablation System, a next-generation radiofrequency ablation (RFA) platform designed specifically for treating painful bone metastases and benign bone tumors such as osteoid osteoma.
To learn more about this report, Request Free Sample
The metastatic bone cancer segment is anticipated to dominate the market with the highest market share in 2026, occupying about 48.0% of the entire market. The dominance can be attributed to the prevalence of secondary bone tumors that arise from the spread of cancer cells from the primary site, such as breast cancer, lung cancer, or prostate cancer, to the bone sites.
In May 2025, Stryker announced FDA clearance and expansion of its OptaBlate RF-based pain and tumor ablation portfolio, strengthening its interventional oncology offerings for bone metastases and vertebral tumor pain management.
Hospitals are anticipated to dominate the bone tumor ablation devices market in 2026, capturing approximately 64.0% of the total market share. This dominance is driven by the availability of advanced imaging technologies such as CT and MRI, well-established oncology departments, and the presence of highly skilled healthcare professionals required to perform complex ablation procedures.
As per the American Hospital Association, hospitals are responsible for carrying out most of the complicated and minimally invasive procedures, hence making them the ideal place for the ablation of bone tumors due to their full-fledged infrastructure.
In April 2024, Medtronic highlighted increased hospital adoption of its RF ablation platform for treating metastatic bone cancer pain, particularly in spine, ribs, and extremities. The system is widely used in hospital interventional radiology and oncology unit.
Advances towards minimally invasive procedures have been the key drivers contributing to the growth of the bone tumor ablation device market. With the help of image-guided tumor ablation, it has become easier to target tumors without damaging the surrounding tissue, hence helping in quicker recovery and less post-procedural pain for the patient.
A study in the National Library of Medicine indicates that minimal-invasive tumor ablation surgery reduces the hospitalization period by 30% to 50%, and the chance of developing complications during treatment.
The constant improvement of ablation techniques such as radio frequency ablation, microwave ablation, and cryoablation makes bone tumor treatment even more effective and safe for patients. This improvement includes more accurate temperature regulation and the ability to monitor the procedure more effectively. It also contributes to the reduction of damage to healthy tissues while treating complex tumors.
According to the Radiological Society of North America, newer ablation technologies have demonstrated local tumor control rates exceeding 80–90% in certain bone tumor cases, reinforcing their growing role in oncology care. Increased integration of image-guided systems and navigation tools further enhances procedural success rates and expands clinical applications.
|
Current Event |
Description and its Impact |
|
2026 Medicare Reimbursement and Coding Updates for Interventional Oncology Procedures in the U.S. |
|
|
Regulatory Approvals and Policy Support for Minimally Invasive Cancer Treatments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
The North American market is forecasted to have the highest share of 38.0%, owing to well-developed healthcare systems, high adoption of minimally invasive oncology surgeries, and the presence of many major medical device manufacturers. The prevalence of cancers and the usage of image-guided interventional procedures contribute to market dominance in the region.
Another factor behind the rising demand for ablation treatments includes the increased cases of metastatic bone disease. According to the American Cancer Society, breast cancer, prostate cancer, and lung cancer are some of the most frequent types of cancer that result in bone metastasis.
In December 2024, AngioDynamics received FDA 510(k) clearance for its NanoKnife® irreversible electroporation system, expanding ablation options for cancer treatment in North America.
The Asia Pacific region is anticipated to be the fastest-growing region for the forecast period from 2026 to 2033 owing to an increase in the incidence of cancer, improvements in health care facilities, and an increased preference for minimally invasive procedures.
As per the World Health Organization, there is an accelerating trend of cancer prevalence across the Asia Pacific region, especially in large populations like China and India, leading to rising demands for sophisticated cancer treatments that include bone tumors ablation.
The U.S. bone tumor ablation device market is experiencing rapid growth due to an increase in cancer incidences and bone metastasis as well as growing use of minimally invasive interventional oncology therapies. The U.S. market is considered as a major market for cancer around the globe, with breast, prostate, and lung cancers accounting for most of the bone metastasis that are typically treated via radiofrequency or microwave ablation.
Strengthens hospital-based interventional oncology capacity in the U.S., improves procedural efficiency, and increases adoption of radiofrequency ablation for metastatic bone cancer pain management.
The Japan bone tumor ablation devices market will continue growing at a consistent pace because of the rapid increase in the number of elderly people suffering from various diseases like cancers. Japan has become one of the most super-aged countries in the world with a high percentage of elderly people who have a high risk of developing cancer.
According to WHO (World Health Organization) reports, one of the major contributing factors for increasing rates of cancer among the global population is an aging population, which includes developed countries such as Japan.
Some of the major key players in Bone Tumor Ablation Devices are Medtronic Plc., Merit Medical Systems Inc., MedWaves Inc., AngioDynamics Inc., Olympus Corporation, Varian Medical Systems, Inc., Mermaid Medical A/S, RF Medical Co., Ltd., and Misomix Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 111.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.2% | 2033 Value Projection: | USD 219.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic Plc., Merit Medical Systems Inc., MedWaves Inc., AngioDynamics Inc., Olympus Corporation, Varian Medical Systems, Inc., Mermaid Medical A/S, RF Medical Co., Ltd., and Misomix Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients