Burkitt Lymphoma Treatment Market Size and Forecast – 2026 – 2033
The Global Burkitt Lymphoma Treatment Market size is estimated to be valued at USD 1.45 billion in 2026 and is projected to reach USD 2.75 billion by 2033, growing at a compound annual growth rate (CAGR) of 9.2% during the forecast period from 2026 to 2033.
Global Burkitt Lymphoma Treatment Market Overview
The Burkitt lymphoma treatment market is driven by rising incidence of non-Hodgkin lymphomas, increasing awareness of early cancer diagnosis, and advancements in targeted therapies and immunotherapies. Growing adoption of combination chemotherapy regimens, improved access to healthcare infrastructure, and expanding oncology research investments are accelerating market growth. Government initiatives and support from organizations such as World Health Organization and American Cancer Society also contribute to better treatment accessibility. Additionally, increasing clinical trials, development of personalized medicine, and demand for effective therapies in pediatric and adult populations are encouraging pharmaceutical companies to introduce innovative treatment options, further boosting the global Burkitt lymphoma treatment market.
Key Takeaways
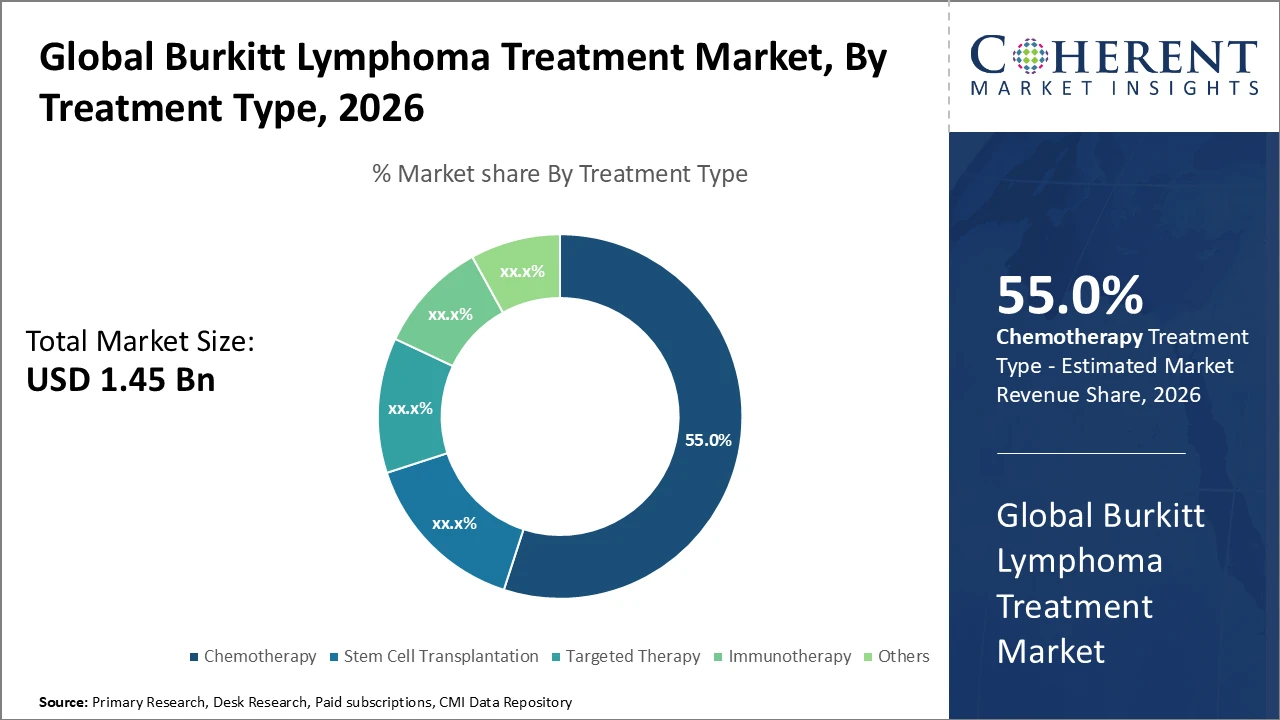
Chemotherapy segment dominates the treatment landscape, holding 55% market share, while immunotherapy is the fastest growing due to improved efficacy and reduced toxicity.
Pediatric patient type is increasingly prioritized for innovative clinical trials, contributing to a diversified patient population approach.
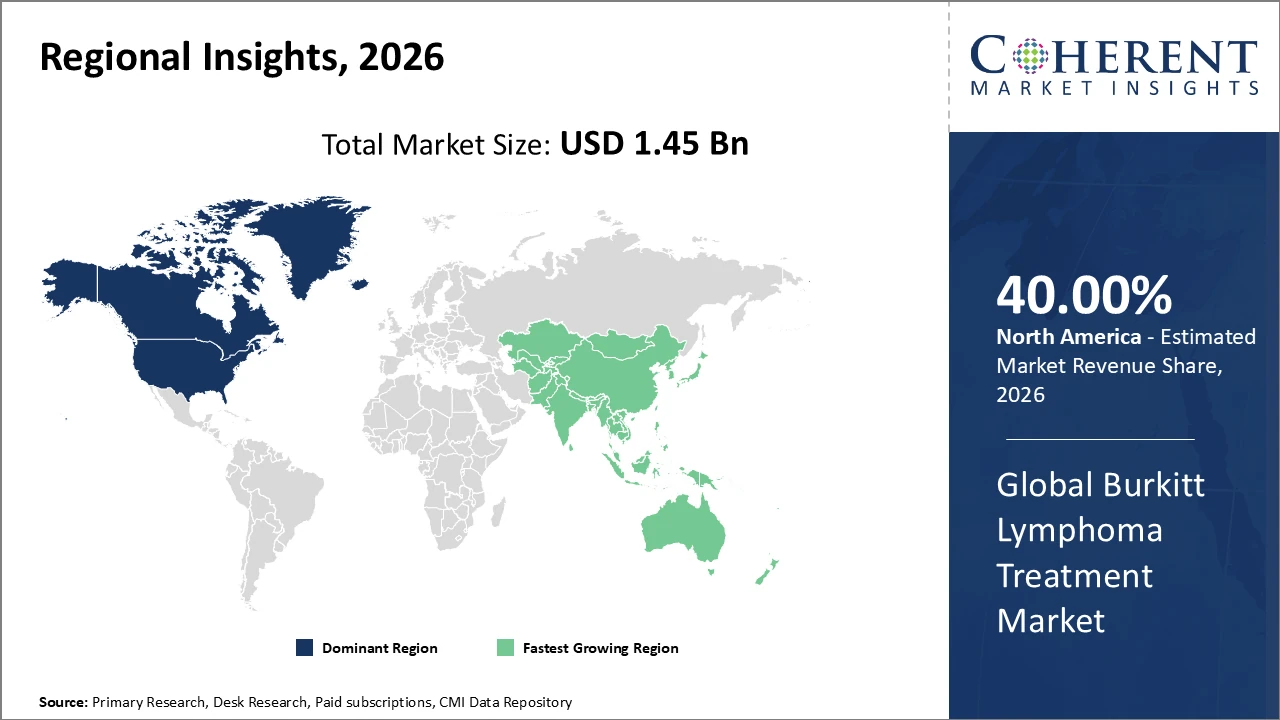
North America leads the Burkitt lymphoma treatment market, with advanced healthcare infrastructure and reimbursement policies accounting for over 40% industry share.
Asia Pacific exhibits the fastest growth trajectory, driven by increasing disease prevalence and government-led healthcare reform accelerating market penetration.
Burkitt Lymphoma Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Burkitt Lymphoma Treatment Market Insights, By Treatment Type
Chemotherapy dominates with a 55% market share due to its proven efficacy and widespread clinical use in frontline treatment. Immunotherapy is the fastest-growing segment, offering durable remission and fewer side effects, especially in relapsed or refractory cases. Targeted therapies, including small molecule inhibitors, support personalized approaches and address unmet clinical needs. Stem cell transplantation serves high-risk patients but has limited market penetration. Other emerging experimental treatments contribute to incremental growth, reflecting an evolving therapeutic landscape driven by innovation and patient-focused strategies.
Burkitt Lymphoma Treatment Market Insights, By Patient Type
Pediatric patients dominate the Burkitt lymphoma treatment market due to the high incidence in children, especially in endemic regions, and established pediatric oncology protocols. The adult segment is the fastest-growing, driven by advancements in targeted therapies and immunotherapies that enable tailored regimens with improved tolerability. Growth in the geriatric segment is limited by comorbidities and higher treatment-related toxicity, though emerging less intensive approaches are gaining attention. The “others” category includes immunocompromised patients requiring specialized care. This demographic segmentation highlights how age- and condition-specific clinical strategies influence treatment outcomes, patient management, and overall market demand.
Burkitt Lymphoma Treatment Market Insights, By End User
Hospitals dominate the Burkitt lymphoma treatment market, driven by the need for complex inpatient procedures such as intensive chemotherapy and stem cell transplantation. Specialty clinics are the fastest-growing subsegment, reflecting a shift toward outpatient care and personalized treatment delivery. Ambulatory care centers and research institutes play key roles in treatment innovation and clinical trials but hold smaller market shares compared to hospitals and clinics. The “others” category includes home care and telemedicine platforms that support patient monitoring and adherence. This end-user segmentation underscores how evolving healthcare infrastructure shapes treatment accessibility, patient management, and strategic growth opportunities in the Burkitt lymphoma market.
Burkitt Lymphoma Treatment Market Trends
Rise of immunotherapies is reshaping Burkitt lymphoma treatment protocols, offering alternatives to conventional chemotherapy.
Growing incorporation of AI-powered diagnostic tools is driving precision medicine and more personalized treatment strategies.
Clinical trials using bispecific antibodies increased by 28% between 2024 and 2026, expanding therapeutic options for patients.
Liquid biopsy techniques have improved minimal residual disease monitoring, allowing for dynamic treatment adjustments and better outcome tracking.
Burkitt Lymphoma Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Burkitt Lymphoma Treatment Market Analysis and Trends
North America dominates the Burkitt lymphoma treatment market, accounting for around 40% of the market share in 2026. This leadership is driven by advanced healthcare infrastructure, widespread insurance coverage, and substantial investments in pharmaceutical research and development. Streamlined regulatory pathways, including those by the FDA, have accelerated the approval and launch of innovative therapies, enhancing patient access. Major pharmaceutical companies such as Roche and Novartis have introduced advanced CAR-T cell therapies, strengthening the region’s treatment portfolio. The combination of technological innovation, clinical expertise, and supportive policies sustains robust market growth and positions North America as a key global leader in Burkitt lymphoma care.
Asia Pacific Burkitt Lymphoma Treatment Market Analysis and Trends
Asia Pacific is projected to exhibit the fastest CAGR of 11.4% in the Burkitt lymphoma treatment market, driven by rising disease incidence, improved diagnostic infrastructure, and government-led initiatives enhancing healthcare access. Countries such as China and India have implemented awareness campaigns and subsidized oncology programs, significantly boosting treatment adoption. Strategic collaborations and licensing agreements among biopharmaceutical companies have facilitated local manufacturing, lowering costs and improving accessibility for a broader patient population. Additionally, increasing investment in healthcare infrastructure and training programs supports early diagnosis and effective management, positioning the region as a rapidly growing market for Burkitt lymphoma therapies.
Burkitt Lymphoma Treatment Market Outlook for Key Countries
USA Burkitt Lymphoma Treatment Market Analysis and Trends
The USA is a key market within North America for Burkitt lymphoma treatment, driven by its concentration of research institutions and biopharmaceutical companies focused on oncology. Approximately 35% of global Burkitt lymphoma clinical trials are conducted in the USA, supporting the early adoption of novel therapies. Comprehensive insurance coverage and strong regulatory frameworks further facilitate market expansion. Leading companies such as Bristol-Myers Squibb and Amgen are introducing innovative immunotherapies, enhancing treatment options and improving patient outcomes. This combination of clinical research leadership, supportive healthcare policies, and pharmaceutical innovation positions the USA as a central driver of growth in the Burkitt lymphoma treatment market.
Germany Burkitt Lymphoma Treatment Market Analysis and Trends
Germany represents a significant Burkitt lymphoma treatment market in Europe, supported by advanced healthcare infrastructure, high standards of clinical care, and well-established oncology networks. The country emphasizes early diagnosis and guideline-driven treatment protocols, with chemotherapy remaining the backbone of therapy, complemented increasingly by immunotherapies and targeted agents. Rising investment in clinical trials, including CAR-T and bispecific antibody studies, reflects a focus on innovative therapies. Growing awareness programs and reimbursement coverage enhance patient access to cutting-edge treatments. The German market trends indicate steady growth, driven by precision medicine adoption, strong R&D activities, and integration of advanced diagnostics and personalized treatment strategies.
Analyst Opinion
Chemotherapy remains the primary treatment, holding approximately 55% market share in 2026. Immunotherapy grew by nearly 18% between 2024 and 2025, reflecting a shift toward precision medicine. Adoption of CAR-T cell therapies in clinical trials increased by 23% in 2025, offering new options for refractory cases.
North America saw a 12% rise in treatment uptake in 2026 due to better healthcare infrastructure and insurance coverage. Asia Pacific experienced a 15% increase in new patient diagnostics, driven by government awareness campaigns and improved laboratory services.
Advances in drug development led to an 8% rise in average treatment costs in 2024, mainly from biologics. Expanded reimbursement policies in developed markets improved patient access to high-cost therapies by 10% in 2026.
Ongoing clinical trials targeting Burkitt lymphoma increased by 30% between 2024 and 2026, focusing on bispecific antibodies and small molecule inhibitors. A novel PI3K inhibitor reported a 40% overall response rate in Phase II trials in 2025, showing promise for future treatment options.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.45 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.2% | 2033 Value Projection: | USD 2.75 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Novartis AG, Roche Holding AG, Amgen Inc., AbbVie Inc., Sanofi, Bayer AG, AstraZeneca Plc, Merck & Co. Inc., Pfizer Inc., Johnson & Johnson | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Burkitt Lymphoma Treatment Market Growth Factors
The Burkitt lymphoma treatment market is driven by rising incidence rates in endemic regions, particularly in Africa, and improved diagnosis through advanced imaging and molecular testing. The integration of immunotherapy and targeted therapies into standard care has enhanced treatment outcomes, contributing to a 20% increase in survival rates from 2024 to 2026. Increased investment in oncology research has led to a 30% rise in clinical trials, supporting pipeline innovation and expanding therapeutic options. Additionally, government initiatives and expanding healthcare infrastructure in emerging economies like India and Brazil have improved patient access and treatment adoption, driving overall market growth and revenue.
Burkitt Lymphoma Treatment Market Development
In February 2026, Neuberg Sehgal Path Lab launched advanced cytogenetics testing technology at its Mumbai facilities, enhancing precision in cancer diagnostics and prenatal testing through more accurate and personalized diagnostic solutions.
Key Players
Leading Companies of the Market
Novartis AG
Roche Holdings AG
Merck & Co.
Pfizer Inc.
AstraZeneca plc
Amgen Inc.
Bayer AG
AbbVie Inc.
Johnson & Johnson
Sanofi
Leading companies in the Burkitt lymphoma treatment market have pursued focused growth strategies to strengthen their positions. Roche integrated next-generation sequencing into treatment personalization, leading to a 15% increase in clinical trial enrollments by 2026. Novartis formed strategic partnerships with biotech firms to accelerate the development of CAR-T therapies, achieving regulatory milestones ahead of schedule. Bristol-Myers Squibb expanded its immunotherapy portfolio through aggressive licensing agreements, increasing its global market share by 7% in 2025. These strategic initiatives highlight the emphasis on innovation, collaboration, and portfolio diversification to drive competitive advantage and market growth.
Burkitt Lymphoma Treatment Market Future Outlook
The future outlook of the Burkitt Lymphoma treatment market is promising, driven by advancements in precision medicine and immunotherapy. The integration of CAR-T cell therapies, bispecific antibodies, and other targeted biologics is expected to improve survival rates and reduce treatment-related toxicity. Ongoing clinical trials are exploring novel combination regimens and personalized approaches, expanding options for refractory and high-risk patients. Additionally, growing awareness, early diagnosis, and increasing healthcare infrastructure in emerging markets will boost accessibility. The market is projected to experience steady growth as pharmaceutical companies continue investing in research and development to deliver more effective and patient-centric therapies.
Burkitt Lymphoma Treatment Market Historical Analysis
Historically, the Burkitt Lymphoma treatment market has been dominated by intensive chemotherapy regimens, including CODOX-M/IVAC and Hyper-CVAD, which have been the standard of care for decades. The introduction of monoclonal antibodies, particularly rituximab, in the early 2000s significantly improved patient survival and remission rates when combined with chemotherapy. Over time, supportive care therapies, such as antiemetics and growth factors, became integral to managing treatment-related side effects. Market growth during this period was driven by increasing disease awareness, advancements in combination therapy protocols, and gradual adoption of immunotherapy, setting the stage for current innovations in targeted and cellular therapies.
Sources
Primary Research Interviews:
Hematologists, oncologists, and pediatric oncologists directly involved in Burkitt Lymphoma diagnosis and treatment.
Hospital administrators, oncology department managers, and procurement specialists responsible for acquiring chemotherapy, immunotherapy, and targeted therapy drugs.
Product managers, clinical researchers, and regulatory experts from pharmaceutical companies developing monoclonal antibodies, CAR-T cell therapies, and chemotherapy regimens for B-cell lymphomas.
Magazines:
Oncology Times – Updates on lymphoma treatments, clinical studies, and market trends.
Hematology/Oncology Today – Insights into blood cancers, treatment innovations, and therapeutic guidelines.
Cancer Therapy Advisor – Focus on drug therapies, clinical trials, and treatment protocols for hematologic malignancies.
Journals:
Blood – Studies on Burkitt Lymphoma, treatment efficacy, and clinical research.
Journal of Clinical Oncology – Research on chemotherapy, immunotherapy, and targeted therapies in lymphomas.
Leukemia & Lymphoma – Investigations on novel therapies, molecular targets, and patient outcomes.
Newspapers:
The Financial Times – Global healthcare trends and pharmaceutical investments in oncology.
Reuters – Updates on drug approvals, innovations, and market expansion in lymphoma treatments.
Bloomberg – Market performance, corporate strategies, and developments in oncology sectors.
Associations:
American Society of Hematology – Guidelines, research, and clinical standards in lymphoma care.
Leukemia & Lymphoma Society – Research, conferences, and treatment updates for B-cell malignancies.
International Agency for Research on Cancer – Standards, epidemiology, and global cancer treatment data.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients