Global embolization market is estimated to be valued at USD 2.61 Bn in 2026 and is expected to reach USD 4.71 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The market growth is primarily driven by increasing prevalence of cardiovascular, oncology and neurological disorders worldwide. Embolization procedures are minimally-invasive, and are less painful for patients as compared to conventional open surgeries. This is expected to boost adoption of embolization processes in the near future.
Market players are constantly investing in research and development to launch newer and more effective embolic agents. Increasing application of these procedures for treating liver cancer and other tumors can offer growth opportunities. However, high cost of embolization procedures and lack of reimbursement policies in some emerging nations can hamper the market growth.
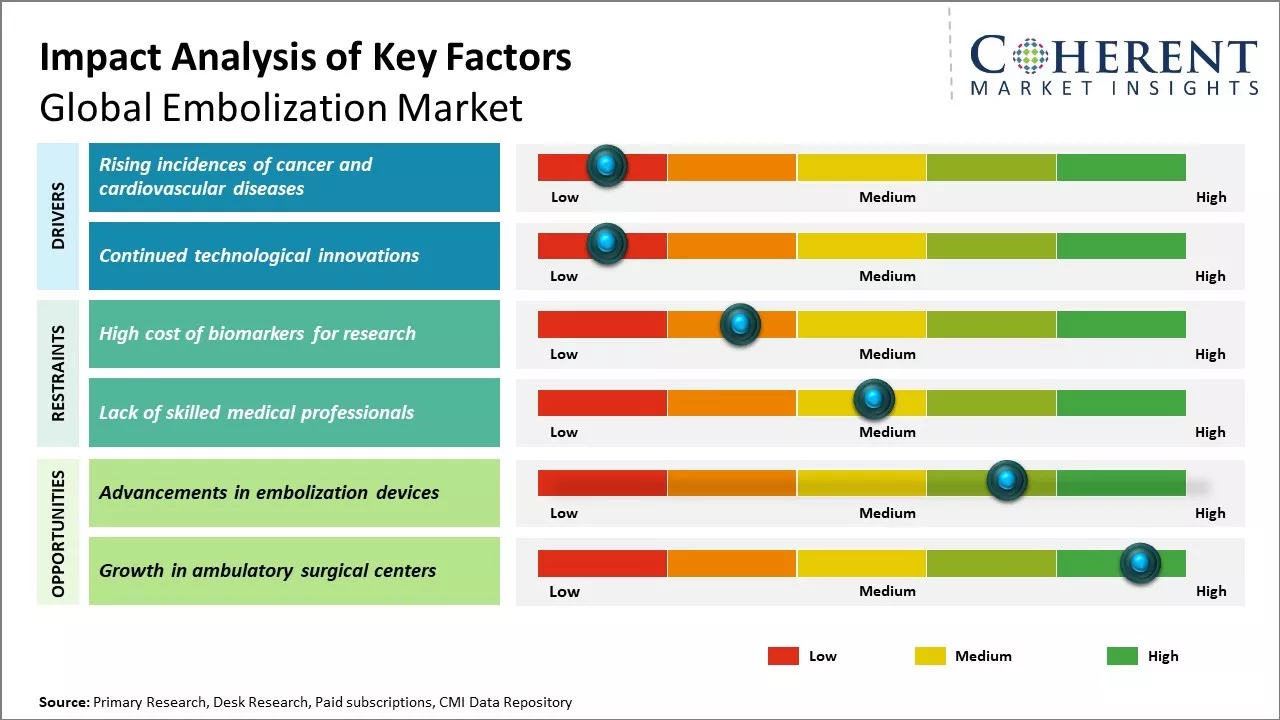
Rising incidences of cancer and cardiovascular diseases
Rising cases of cancer and cardiovascular diseases can drive the market growth. According to the World Health Organization, cardiovascular diseases are the number one cause of death globally, with an estimated 17.9 million deaths due to CVDs each year. Cancer also poses a major threat, with nearly 10 million new cases and 9.6 million deaths due to cancer in 2018. Advanced interventional procedures and minimally invasive treatment techniques have allowed physicians to treat these conditions. Embolization has emerged as a vital therapeutic option for treatment of cancerous and non-cancerous tumors as well as certain cardiovascular conditions. It is being widely used to restrict blood flow to tumors prior to surgery or radiation therapy, thus, aiding in their destruction. For cardiovascular applications, embolization treat aneurysms, uterine fibroids and arterial venous malformations by blocking blood supply to abnormal blood vessels. Growing prevalence of diseases , coupled with increasing adoption of minimally invasive procedures can boost demand for embolization devices and accessories. Global embolization market growth is driven by several factors, including increasing incidence of vascular diseases, advancements in embolization products and techniques, rising R&D efforts, and growing demand for minimally invasive treatments. For instance, in 2020, according to the World Health Organization, ischemic heart disease alone contributes to 16% of annual global deaths. Cardiovascular diseases cause around 17.9 million deaths worldwide each year, representing 35% of total global mortality. Out of these cardiovascular-related deaths, 85% are attributed to heart attacks and strokes. Ischemic heart disease is highlighted as a prominent cause of global mortality, affecting approximately 126 million individuals worldwide, and will increase to over 1,845 cases per 100,000 people by 2030. This underscores the significant impact of cardiovascular diseases, including coronary heart disease, rheumatic heart disease, cerebrovascular disease, and other cardiac conditions, as leading causes of death worldwide.
Get actionable strategies to beat competition: Request Free Sample
Continued technological innovations
Continual technological innovations expand the clinical applications of embolization, thus, driving the market growth. Vendors are developing new product formulations, delivery technologies and advanced techniques that enhance procedural safety, efficiency and outcomes. For instance, new detachable coils allow for more targeted embolization of aneurysms and malformations. Advancements like microspheres and drug-eluting beads permit prolonged localized drug delivery post-embolization. Biosurgical thrombin improves hemostasis during procedures. Improved imaging systems also assist in precise catheter navigation and placement. The availability of biocompatible materials allows for longer-term occlusion with reduced risks. Furthermore, vendors are exploring innovative methods like combination therapy and integration of embolic agents with diagnostic capabilities. Such technological advancements are supporting treatment of complex cases, improving success rates and enabling use of embolization in new clinical indications. These are likely to make embolization more effective and viable option for broader patient base over time, thus, driving the market growth.
Key Takeaways from Analyst:
Global embolization market growth is driven by rising geriatric population and increasing incidence of target diseases such as cancer and cardiovascular conditions. The market witness high potential in vascular embolization procedures used for hemorrhage control in trauma cases and uterine fibroid embolization. Lifestyle diseases can boost demand for minimally invasive procedures worldwide.
North America dominates the market due to ongoing technological advancements and favorable reimbursement policies for novel embolic agents. However, Asia Pacific is likely to emerge as the fastest growing region due to growing medical tourism, rising healthcare expenditure, and increasing focus of key players on untapped opportunities in developing nations.
Increasing introduction of innovative embolic products with better performance characteristics and adoption of advanced endovascular techniques for complex cases can drive the market growth. Lack of skilled radiologists and strong regulatory frameworks for approval of new entrants can hamper the market growth, especially in low and middle-income countries.
Market Challenges: High cost of Biomarkers
The high cost of biomarkers treatment procedures can hamper the global embolization market growth. Embolization procedures such as tumor embolization require the use of expensive medical devices and equipment. Due to various factors like advanced technologies involved, high manufacturing costs of these devices, specialized training required by physicians and long hospital stay times, the overall cost of embolization significantly adds to the economic burden on patients. This escalating cost of accessing these specialized treatment options can restrict their uptake, especially in developing regions and among lower-income groups in developed markets. According to the data published by WHO in 2022, over 100 million people worldwide are pushed into extreme poverty each year due to health expenses. The high out-of-pocket spending on specialized procedures denies equitable access to quality healthcare for all.
Market Opportunities: Advancements in embolization devices
Advancements in embolization devices can offer opportunities for global embolization market. Embolization procedures are minimally invasive techniques that is used to block blood vessels or abnormalities to cut off blood flow to an area of the body. Traditionally, embolization devices involved coils, liquid embolic agents and vascular plugs. However, emerging technologies allows clinicians to treat more complex conditions with minimally invasive therapies. Newly developed microspheres, beads, plugs and spacers are enabling physicians to embolize difficult to access vascular spaces and small target vessels with improved control, precision and efficacy compared to traditional options. This makes embolization procedures suitable for more complex cases and expands the range of treatable conditions.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
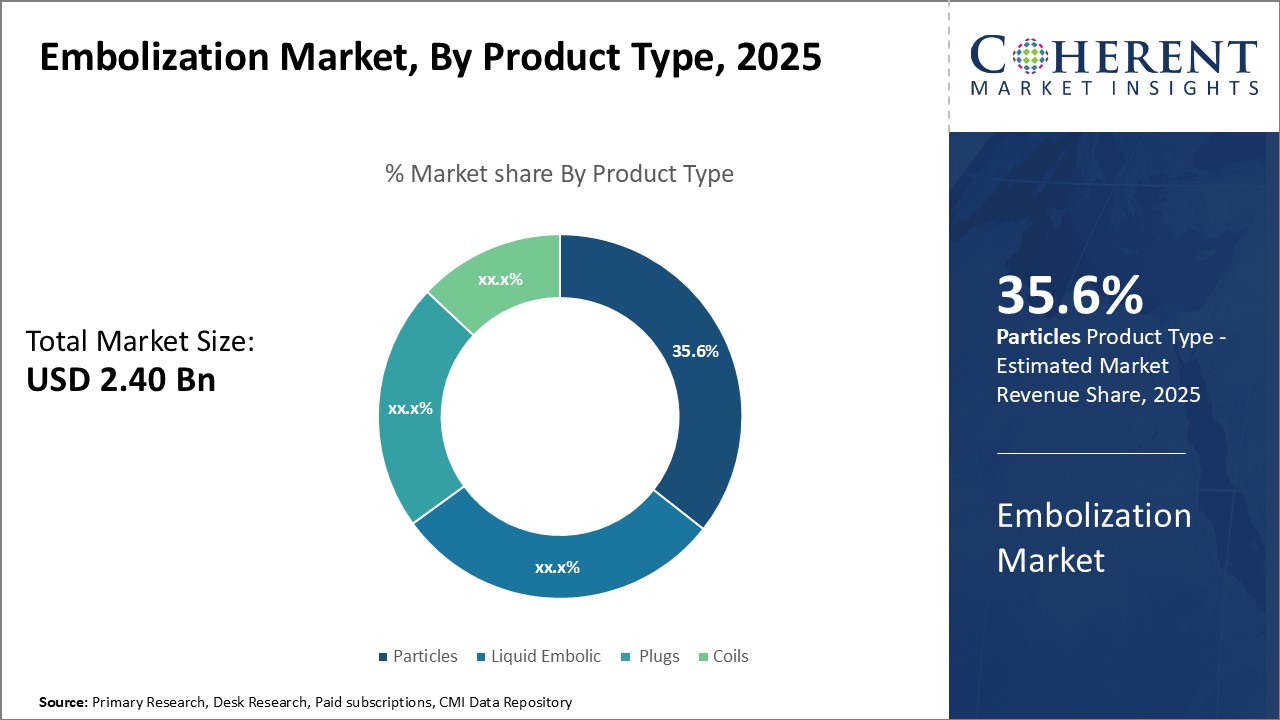
Insights, By Product Type- Adaptable applications boosts particles segment growth
In terms of product type, particles segment is estimated to contribute the highest market share of 36.6% in 2026, due to their diverse applications across different indications. Being made of various compounds like polyvinyl alcohol, tris-acryl gelatin microspheres, and microsphere combinations, particles can be tailored to meet the requirements of a wide range of treatments. Their customizable size, shape and material properties allow physicians to choose the appropriate particle type depending on the vascular anatomy, blood flow characteristics and other clinical factors of each individual patient. This adaptability makes particles effective for blocking blood vessels of various calibers in different areas of the body. Many particle types are non-absorbable and offer long-term occlusive effects, providing lasting relief to patients. Growing need for personalized embolization procedures and preference for adaptable products that can be used across indications can drive the segment growth.
To learn more about this report, Request Free Sample
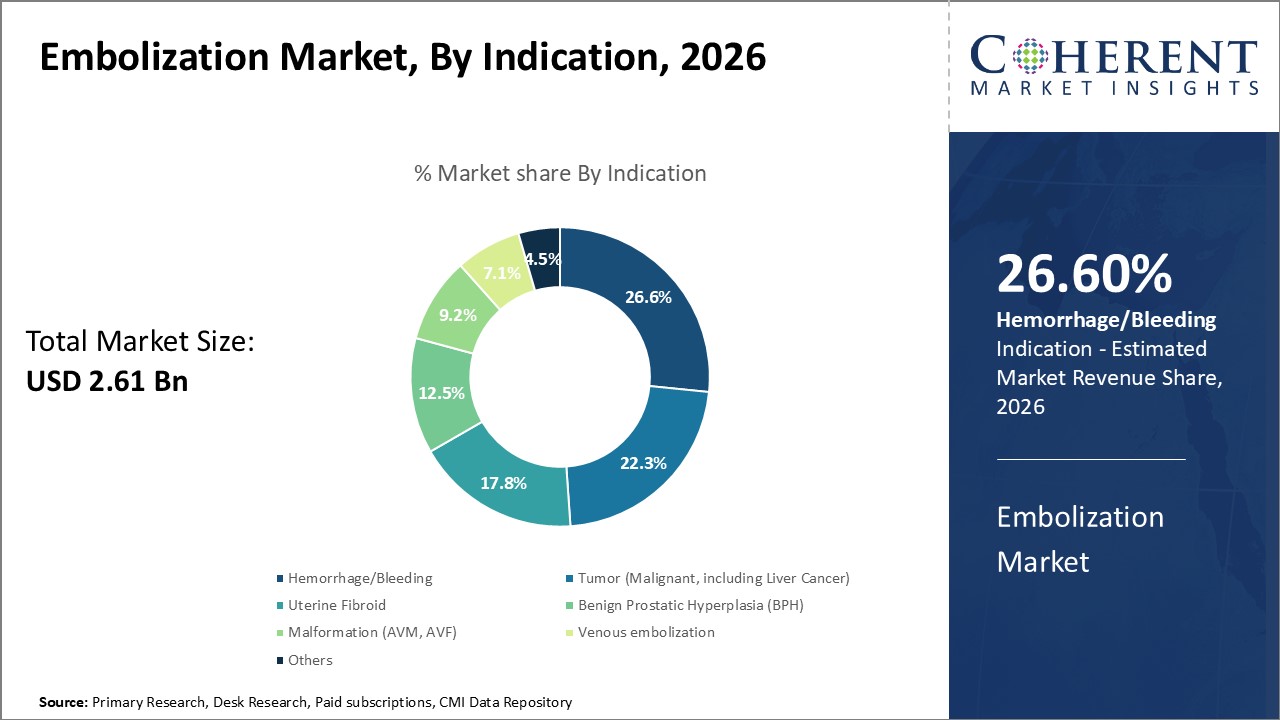
By Indication- Rising incidence fuels bleeding condition prevalence
In terms of indication, hemorrhage/bleeding segment is estimated to contribute the highest market share of 26.6% in 2024, due to increase in associated risk factors and conditions. Factors such as aging population, rising obesity levels, wider use of anticoagulants, and adoption of invasive diagnostic/therapeutic techniques are contributing to elevated bleeding risks. Conditions like peptic ulcers, trauma injuries, liver diseases and vascular malformations are becoming more prevalent. Interventional radiology is being utilized more often as a minimally-invasive alternative to surgeries for hemorrhage control. These trends have significantly raised the caseload of emergency and elective bleeding embolization procedures in recent years. Rising global incidence of underlying diseases and risk factors that may cause hemorrhage can drive the segment growth.
By End User- Provider preference cements hospital dominance
In terms of end user, hospitals & clinics segment is estimated to contribute the highest market share of 61.6% in 2026, as it is the preferred locations for embolization. As specialized interventional radiology and endovascular surgery services become widely available in hospitals, these are capturing a major share of these procedures away from ambulatory centers. Medical facilities possess the necessary infrastructures like dedicated interventional radiology suites, 24/7 catheterization lab support, intensive care backup and a multidisciplinary team of specialists. This enables them to safely and effectively handle complex cases as well as emergency bleeding situations better than other settings. Many patients opt for hospitals over ambulatory clinics as these provide a perception of higher quality care. Insurance coverage is often better for in-patient hospitalization. With provider and patient preferences leaning towards hospitals, this segment will be dominant in the global embolization market.
Need a Different Region or Segment? Request Free Sample
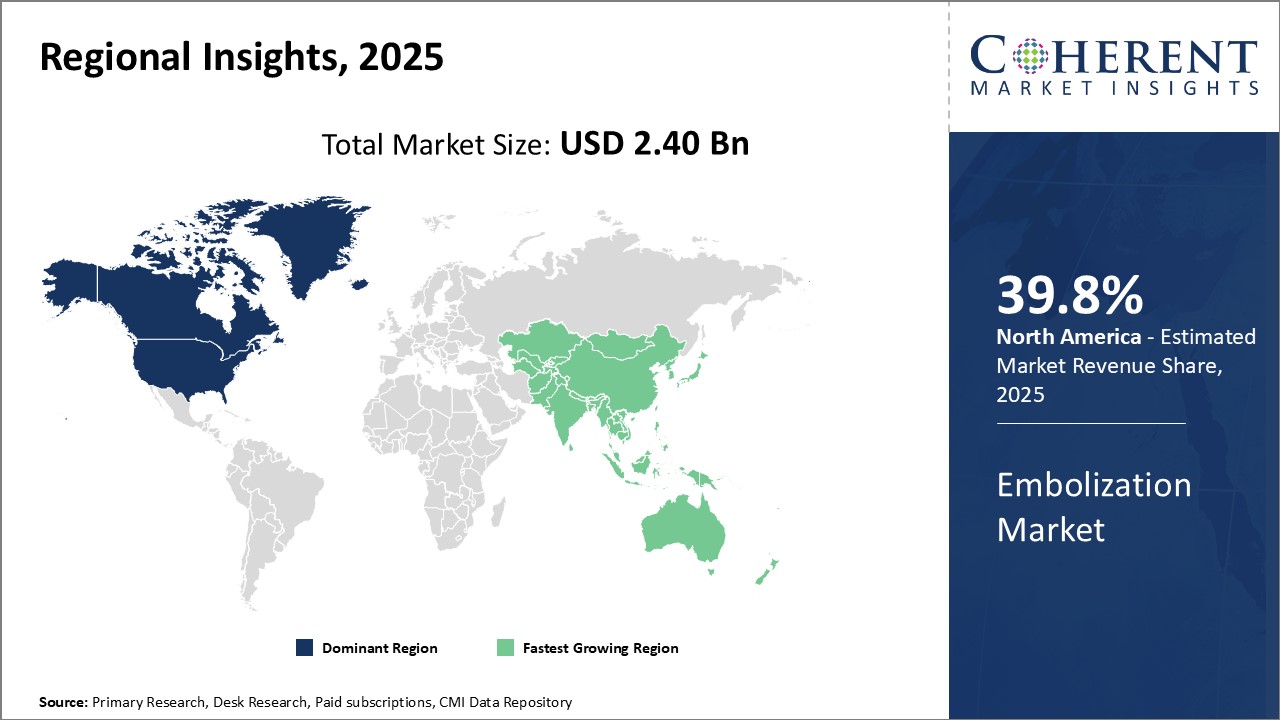
North America has established itself as the dominant region in the global embolization market with estimated market share of 39.8% in 2026 due to well-established healthcare and research infrastructure. Major pharmaceutical and biotechnology companies have their headquarters in the U.S. and Canada that provides them an edge in R&D for new embolization. Furthermore, the presence of top medical devices players engaged in neurobiology research and development can drive the regional growth market. Support from government bodies in the form of grants and funding for neuroscience projects has also propelled the market growth. Several universities and academic institutions in the U.S. and Canada are engaged in collaborative research with industry players to discover novel biomarkers.
Asia Pacific is poised to be the fastest growing region over the forecast period due to improving healthcare infrastructure and increasing expenditure on neurology care. Rapid economic development of countries such as China and India have increased access to advanced healthcare facilities. Rising focus on early disease diagnosis and availability of low-cost skilled labor for manufacturing are attracting biopharmaceutical players to establish production and R&D hubs in Asia Pacific. This has augmented investments in the regional embolization industry.
Embolization Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.61 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.8% | 2033 Value Projection: | USD 4.71 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
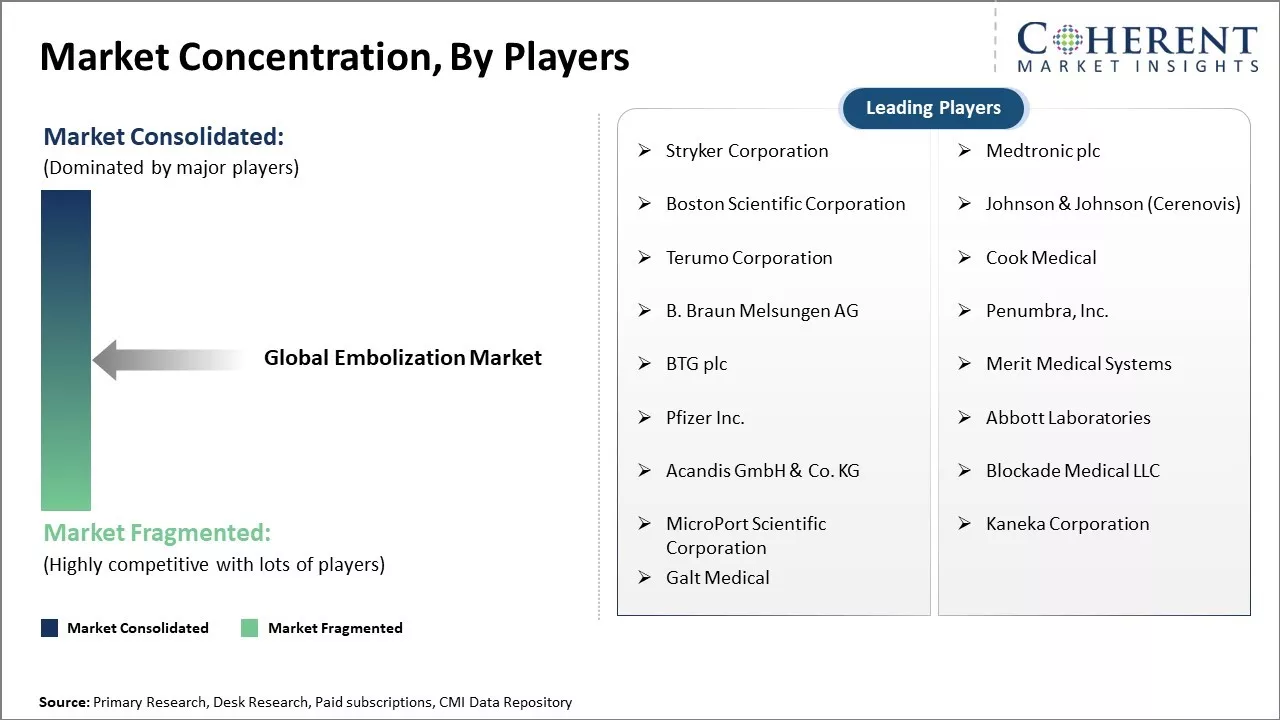
Stryker Corporation, Medtronic plc, Boston Scientific Corporation, Johnson & Johnson (Cerenovis), Terumo Corporation, Cook Medical, B. Braun Melsungen AG, Penumbra, Inc., BTG plc, Merit Medical Systems, Pfizer Inc., Abbott Laboratories, Acandis GmbH & Co. KG, Blockade Medical LLC, MicroPort Scientific Corporation, Kaneka Corporation, Galt Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients