Porphyria Treatment Market Size and Forecast – 2026 – 2033
The Global Porphyria Treatment Market size is estimated to be valued at USD 1.2 billion in 2026 and is expected to reach USD 2.1 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 8.5% from 2026 to 2033.
Global Porphyria Treatment Market Overview
The porphyria treatment market is driven by increasing awareness and improved diagnosis of rare genetic disorders, leading to higher treatment uptake. Growing prevalence of acute hepatic porphyrias and advancements in genetic testing are supporting early detection and intervention. The introduction of novel therapies, including RNA-based and targeted treatments, is significantly improving disease management outcomes. Rising investment in orphan drug development and favorable regulatory incentives such as fast-track approvals and orphan drug designations are further accelerating market growth. Additionally, increasing patient support programs and expanding research into safer, more effective long-term treatment options are contributing to sustained demand in this specialized market.
Key Takeaways
Acute Hepatic Porphyria treatment holds the dominant share of the Porphyria Treatment Market due to the availability of targeted therapies and extensive ongoing clinical research activities.
Oral administration remains the preferred route of treatment because of greater patient convenience, easier compliance, and expanding use of long-term maintenance therapies.
North America accounts for more than 40% of the global market share, supported by advanced healthcare infrastructure, strong rare disease awareness, and favorable reimbursement policies.
Asia Pacific is projected to witness the fastest CAGR owing to improving healthcare accessibility, rising diagnosis rates, and growing investments in rare disease treatment programs.
Europe maintains steady market growth through a strong regulatory framework, active rare disease research initiatives, and the presence of leading pharmaceutical and biotechnology companies.
Porphyria Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
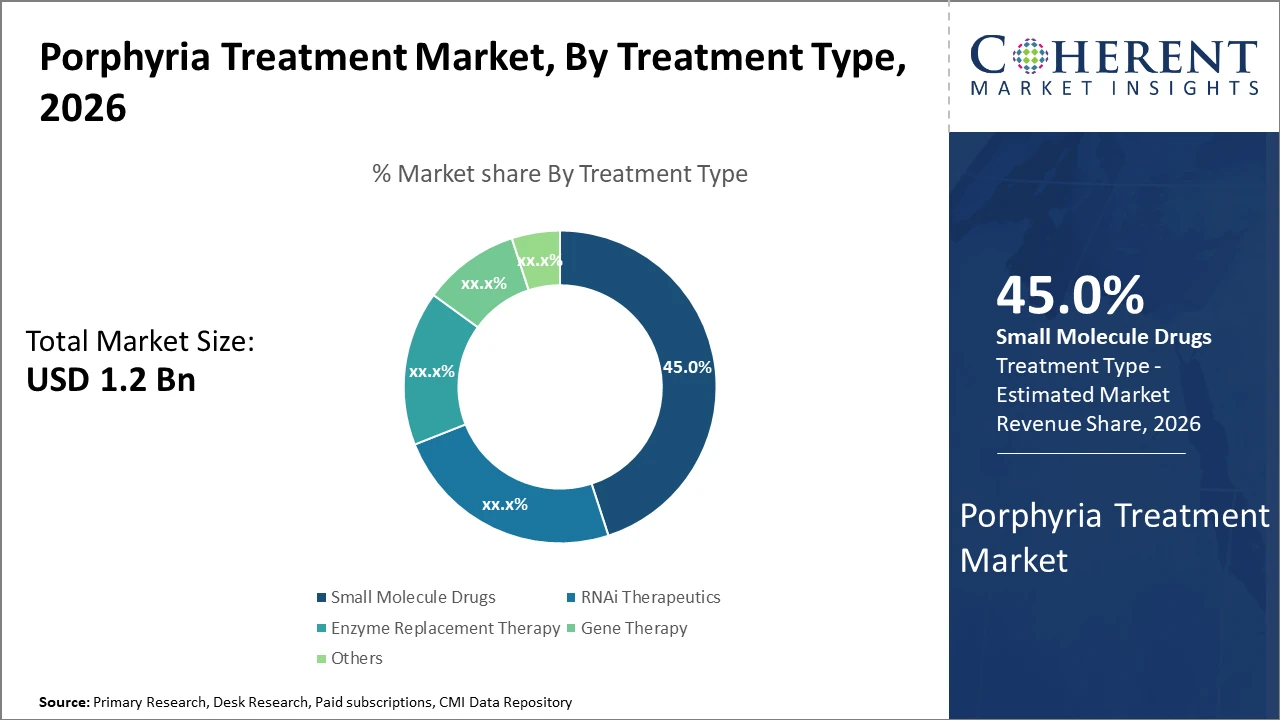
Porphyria Treatment Market Insights, By Treatment Type
Small Molecule Drugs account for nearly 45% of the Porphyria Treatment Market share due to their established safety profiles, affordability, and widespread use in managing symptoms associated with cutaneous porphyria. These therapies continue to serve as the foundation of conventional treatment approaches. RNAi Therapeutics are emerging as the fastest-growing segment, driven by highly targeted gene-silencing mechanisms and strong clinical efficacy in acute hepatic porphyria following the introduction of advanced therapies such as givosiran. Enzyme Replacement Therapy and Gene Therapy are promising emerging segments focused on modifying disease progression and improving long-term outcomes. Other segments include experimental therapies and combination treatment approaches targeting complex porphyria conditions.
Porphyria Treatment Market Insights, By Route of Administration
Oral administration accounts for nearly 52% of the Porphyria Treatment Market share due to its convenience, ease of use, and suitability for long-term disease management in chronic patients. Oral therapies are widely preferred because they improve patient adherence and reduce the need for frequent hospital visits. The subcutaneous route is the fastest-growing segment, primarily driven by the increasing adoption of RNA interference therapies that offer targeted treatment with improved patient compliance. Intravenous therapies remain clinically important for acute management but hold a smaller share because of invasive administration procedures and higher healthcare costs. Other administration routes include experimental and combination delivery approaches currently under clinical evaluation.
Porphyria Treatment Market Insights, By Porphyria Type
Acute Hepatic Porphyria (AHP) holds nearly 60% of the Porphyria Treatment Market share due to its severe clinical impact, higher diagnosis rates, and availability of advanced targeted therapies such as RNA interference treatments. Continuous research activities and growing awareness are further expanding therapy adoption and market growth. Cutaneous Porphyria remains an important segment with steady demand for symptom-management therapies, particularly small molecule drugs. Mixed Porphyria is emerging as a growing subsegment because of increasing interest in gene therapies and enzyme-based treatments aimed at addressing complex disease manifestations. Other rare porphyria variants continue to have limited treatment options and lower diagnostic visibility, restricting broader market penetration.
Porphyria Treatment Market Trends
RNA interference therapies are becoming a major trend in the Porphyria Treatment Market, with more than six advanced therapies introduced between 2024 and 2026 offering targeted gene silencing and improved safety profiles.
Digital health platforms and real-world data integration are enhancing personalized treatment approaches, enabling earlier diagnosis, better patient monitoring, and more effective clinical intervention.
A 2025 observational study reported nearly 15% improvement in clinical outcomes through the use of digital biomarker tracking and data-driven disease management tools.
Emerging markets are increasingly adopting advanced porphyria treatment options due to improving healthcare infrastructure, rising rare disease awareness, and growing international healthcare collaborations.
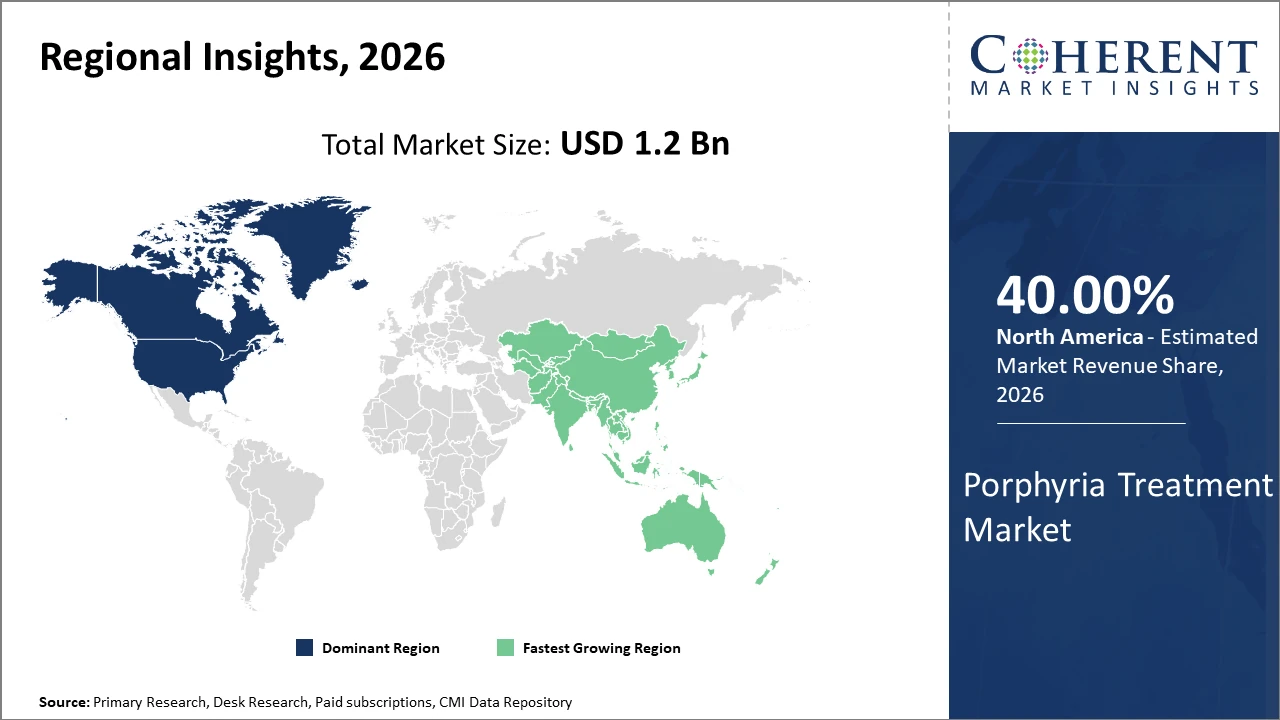
Porphyria Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Porphyria Treatment Market Analysis and Trends
North America dominates the Porphyria Treatment Market with a share of 40% due to strong research and development investments, advanced healthcare infrastructure, favorable reimbursement policies, and the presence of major pharmaceutical companies specializing in rare diseases. The United States contributes the majority of regional market revenue, supported by growing adoption of targeted therapies and continuous innovation from companies such as Alnylam Pharmaceuticals. Government incentives and orphan drug support programs have accelerated rare disease treatment development, contributing to nearly 30% growth in porphyria-related drug approvals between 2024 and 2026. Increasing awareness, improved diagnostic capabilities, and expanding patient access programs continue to strengthen regional market leadership and therapy adoption.
Asia Pacific Porphyria Treatment Market Analysis and Trends
The Asia Pacific Porphyria Treatment Market is projected to register the fastest growth, with a CAGR exceeding 10% during the forecast period. Growth is primarily driven by expanding diagnostic infrastructure, rising healthcare expenditure, and improving awareness of rare metabolic disorders in countries such as China and India. Increasing availability of advanced treatment options and greater access to specialized healthcare services are supporting market expansion across the region. Global pharmaceutical companies are also strengthening their presence through partnerships with local healthcare providers and distributors, improving therapy accessibility and patient support programs. These developments are positioning Asia Pacific as a key emerging market for innovative porphyria therapeutics and rare disease management solutions.
Porphyria Treatment Market Outlook for Key Countries
USA Porphyria Treatment Market Analysis and Trends
The United States remains the leading hub for Porphyria Treatment innovation, accounting for nearly 70% of the North American market share. The introduction of advanced RNA interference therapies such as Givosiran has significantly transformed treatment approaches, contributing to approximately 28% growth in therapy adoption rates between 2024 and 2026. Strong support from regulatory authorities, including FDA orphan drug incentives and priority review programs, has accelerated clinical development and commercialization of rare disease therapies. In addition, robust pharmaceutical research activity, increasing patient awareness, and expanding access to specialized treatment centers continue to reinforce the U.S. leadership position in the global Porphyria Treatment Market.
Germany Porphyria Treatment Market Analysis and Trends
The Germany Porphyria Treatment Market is experiencing steady growth driven by increasing awareness of rare metabolic disorders, advancements in diagnostic capabilities, and improved access to specialized healthcare services. The country’s strong healthcare infrastructure and emphasis on rare disease management support the adoption of advanced porphyria therapies, including RNA interference treatments and hemin-based products. Hospitals and specialty clinics remain the primary treatment centers due to the need for expert monitoring and personalized care. Growing investments in rare disease research and supportive government healthcare policies are further strengthening market development. Additionally, collaborations between pharmaceutical companies and research institutions are accelerating innovation in targeted therapies and improving patient outcomes across Germany.
Analyst Opinion
Over 15 Phase II and III clinical trials for RNA interference and enzyme replacement therapies were ongoing in 2025, accelerating Porphyria Treatment Market growth.
Improved genetic sequencing and biochemical diagnostic methods increased porphyria diagnosis rates by nearly 18% between 2024 and 2026, supporting higher treatment adoption.
Expanded reimbursement coverage for RNAi therapies in North America and Europe grew by around 30% in 2025, improving patient access to advanced treatments.
Public and private investments in rare disease research increased by approximately 12% annually from 2024 to 2026, strengthening innovation and future therapy development.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.2 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.5% | 2033 Value Projection: | USD 2.1 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Novartis AG, Amgen Inc., Paladin Labs, Vertex Pharmaceuticals, PTC Therapeutics, Horizon Therapeutics, Pfizer Inc., Roche Holdings AG, Johnson & Johnson | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Porphyria Treatment Market Growth Factors
The Porphyria Treatment Market is primarily driven by the rising prevalence of acute hepatic porphyrias, with diagnosed cases increasing by nearly 10% between 2024 and 2026. Growing adoption of next-generation RNA interference therapies has significantly improved treatment effectiveness, patient adherence, and long-term disease management outcomes due to targeted mechanisms and reduced side effects. Government support for orphan drug development, including accelerated regulatory approvals and financial incentives in the U.S. and Europe, is further encouraging innovation and new therapy launches. Additionally, increasing awareness campaigns, patient advocacy initiatives, and collaborations among healthcare stakeholders are improving early diagnosis rates and expanding access to specialized treatments worldwide.
Porphyria Treatment Market Development
In August 2022, Disc Medicine initiated the BEACON Phase 2 trial of oral bitopertin to evaluate its safety, tolerability, and efficacy across multiple doses in approximately 20 patients with erythropoietic protoporphyria (EPP) and X-linked protoporphyria (XLP) at study sites in Australia.
Key Players
Leading Companies of the Market
Novartis AG
Vertex Pharmaceuticals
Pfizer Inc.
Roche Holdings AG
Johnson & Johnson
Horizon Therapeutics
PTC Therapeutics
Amgen Inc.
Paladin Labs
Competitive strategies in the Porphyria Treatment Market are increasingly centered on pipeline expansion, advanced therapy development, and patient accessibility initiatives. Alnylam Pharmaceuticals strengthened its market position through strategic acquisitions and expansion of its rare disease therapy portfolio, contributing to nearly 20% growth in market share by 2025. At the same time, Recordati Rare Diseases enhanced its competitive presence through focused investment in RNA interference technologies, resulting in a successful FDA approval in late 2024. Additionally, Apellis Pharmaceuticals implemented patient access and support programs that increased treatment initiation rates by more than 15% across North America during 2025.
Porphyria Treatment Market Future Outlook
The Porphyria Treatment Market is expected to witness significant growth in the coming years due to increasing awareness of rare metabolic disorders, advancements in genetic therapies, and improved diagnostic capabilities. Growing adoption of targeted treatments such as RNA interference therapies and innovative biologics will enhance disease management and reduce acute attack frequency. Expanding healthcare access and supportive government initiatives for rare disease treatment are likely to strengthen market demand globally. Pharmaceutical companies are expected to increase investments in research and development to introduce safer and more effective therapies. Additionally, collaborations between biotechnology firms, research institutions, and specialty clinics will continue to accelerate innovation and improve patient outcomes worldwide.
Porphyria Treatment Market Historical Analysis
The Porphyria Treatment Market has evolved steadily over the years due to increasing recognition of rare metabolic disorders and improvements in diagnostic technologies. Initially, treatment options were limited mainly to supportive care, glucose therapy, pain management, and hemin administration for acute symptom control. As awareness of porphyria increased, healthcare systems and rare disease organizations promoted earlier diagnosis and specialized patient management. Over the past decade, advancements in genetic research and biotechnology enabled the development of targeted therapies, including RNA interference treatments, which significantly improved long-term disease management. Growing investments in rare disease research, expanding specialty care centers, and supportive regulatory policies have further contributed to market growth globally.
Sources
Primary Research Interviews:
Pharmaceutical manufacturers, rare disease specialists, hepatologists, dermatologists, hospital procurement managers, specialty pharmacy providers, and distributors involved in porphyria treatment supply chains and patient care services.
Genetic researchers, clinical toxicologists, regulatory experts, biotechnology innovators, and R&D directors specializing in RNA interference therapies, enzyme replacement technologies, rare metabolic disorders, and orphan drug development.
Magazines:
Rare Disease Advisor – Insights into rare disease therapies, orphan drug advancements, and porphyria treatment developments.
Pharmaceutical Executive – Coverage of biotechnology innovation, specialty therapeutics, and rare disease market trends.
BioPharma Dive – Updates on RNAi therapies, gene therapy advancements, and pharmaceutical regulatory developments.
Journals:
Orphanet Journal of Rare Diseases – Research on rare metabolic disorders, porphyria diagnosis, and innovative treatment approaches.
Journal of Inherited Metabolic Disease – Studies related to porphyria pathophysiology, genetic therapies, and disease management strategies.
The Lancet Gastroenterology & Hepatology – Research covering hepatic disorders, rare liver diseases, and advanced therapeutic interventions.
Newspapers:
Financial Times – Coverage of biotechnology investments, pharmaceutical industry expansion, and rare disease healthcare trends.
Reuters – Reporting on orphan drug approvals, RNA interference therapy developments, and healthcare policy updates.
Bloomberg – Insights into mergers, acquisitions, and strategic partnerships among global biotechnology and pharmaceutical companies.
Associations:
American Porphyria Foundation – Research updates, patient advocacy programs, and porphyria education resources.
National Organization for Rare Disorders (NORD) – Rare disease awareness initiatives, treatment access support, and policy advocacy.
European Porphyria Network – Clinical guidelines, diagnostic standards, and collaborative research developments related to porphyria management.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients