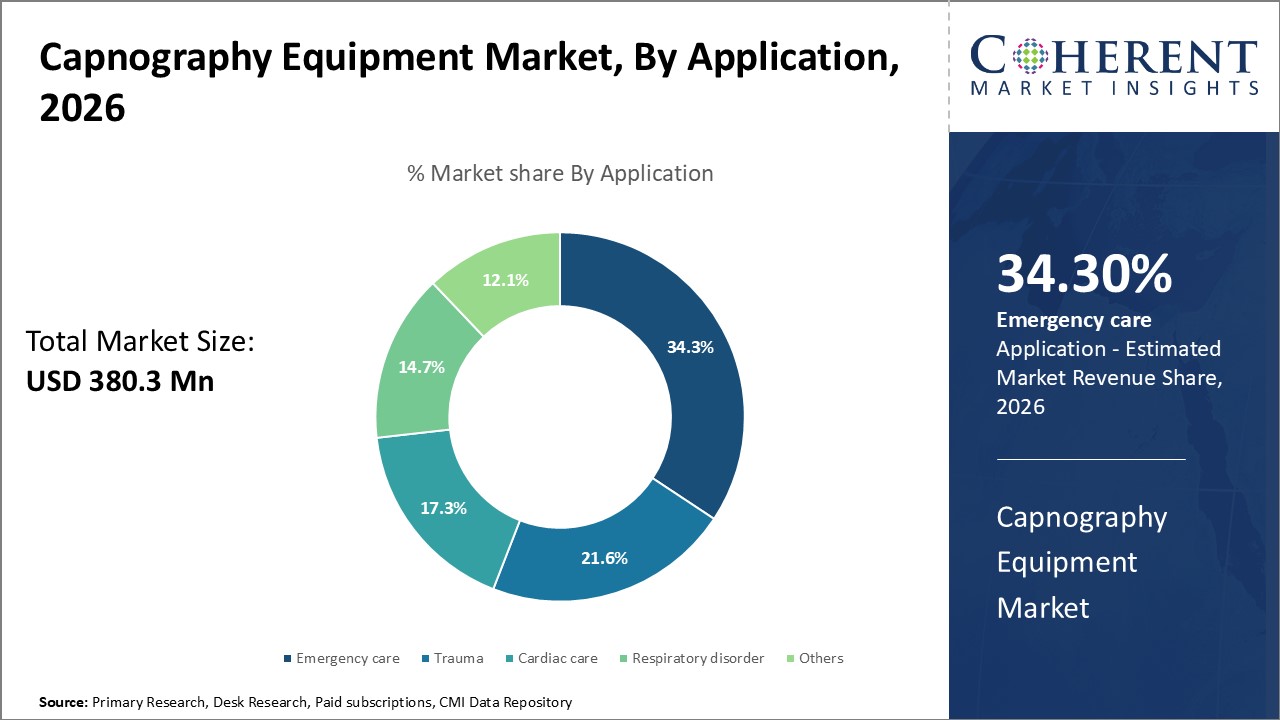
The global capnography equipment market size was valued USD 380.3 Mn in 2026 and is expected to reach USD 546.0 Mn by 2033 growing at a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
The capnography equipment market is growing steadily as it provides vital, non-invasive monitoring of carbon dioxide (CO₂) levels in respiratory gases. It is an essential tool to assess a patient's ventilatory status. Widely used in operating rooms, ICUs, emergency care, and ambulances, capnography is now a standard of care for anesthesia and sedation.
Rising surgical procedures, chronic respiratory diseases, and global emphasis on patient safety are major factors fueling capnography equipment market demand. Technological advancements and portable solutions are expanding its use across pre-hospital and outpatient settings. As healthcare systems prioritize early intervention, demand for capnography continues to accelerate worldwide.
|
Current Event |
Description and its Impact |
|
Regulatory Shifts in Key Markets |
|
|
Clinical Guideline Expansions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product, the mainstream capnographs segment is expected to contribute 66.0% share of the market in 2026, owing to its ability to provide with real-time and continuous monitoring. Capnography can detect subtle changes in ventilation, such as apnea, hypoventilation, or airway obstruction, earlier than other methods like pulse oximetry, allowing for timely intervention and potentially preventing adverse events. During the measurement with mainstream capnographs, there is no time delay in measurement and measurement encompasses the entire volume of air.
In August 2023, Medical device innovator Capno‑Med introduced its new mainstream capnography sensor, designed for seamless integration with ventilators and monitors from leading global manufacturers. It was constructed from medical-grade TPU plastic and gold‑plated connectors, the sensor delivers stable, accurate EtCO₂ waveforms within three seconds and supports respiratory rates up to 150 bpm. It employs non‑dispersive infrared optics for reliable CO₂ detection and withstands splashes (IPX4), while being corrosion‑resistant and durable. With reusable and single‑use adapters available, the device strengthens real‑time respiratory monitoring in anesthesia and critical care settings. This is further accelerating the capnography equipment market demand.
To learn more about this report, Request Free Sample
In terms of application, the emergency care segment is expected to contribute the highest share of the market in 2026, due to the rising demand in pre-hospital and acute care settings. It is also supported by strong clinical guidelines and technological advancements. The rising emergency case such as road traffic accidents, opioid overdoses, cardiac events, and respiratory failures is driving the capnography use in acute emergency care. The advanced technological development in capnography offers compact, battery-operated capnographs, making them ideal for rapid-response teams and ERs.
In February 2023, U.S. trial demonstrated that EtCO₂ measured at ED arrival outperformed traditional vitals in predicting ICU admission and mortality.
In terms of end user, the hospital segment is expected to contribute the largest share of the market in 2026, due to the high patient volume handled by hospital, mostly in ICU, emergency and surgical procedures where capnography is essential for monitoring ventilation and detecting respiratory failure in real time. Along with this, in hospitals, capnography is also used as a standard monitoring tool during general anesthesia, particularly in operating rooms and post-anesthesia care units. This is further accelerating the capnography equipment market growth.
To learn more about this report, Request Free Sample
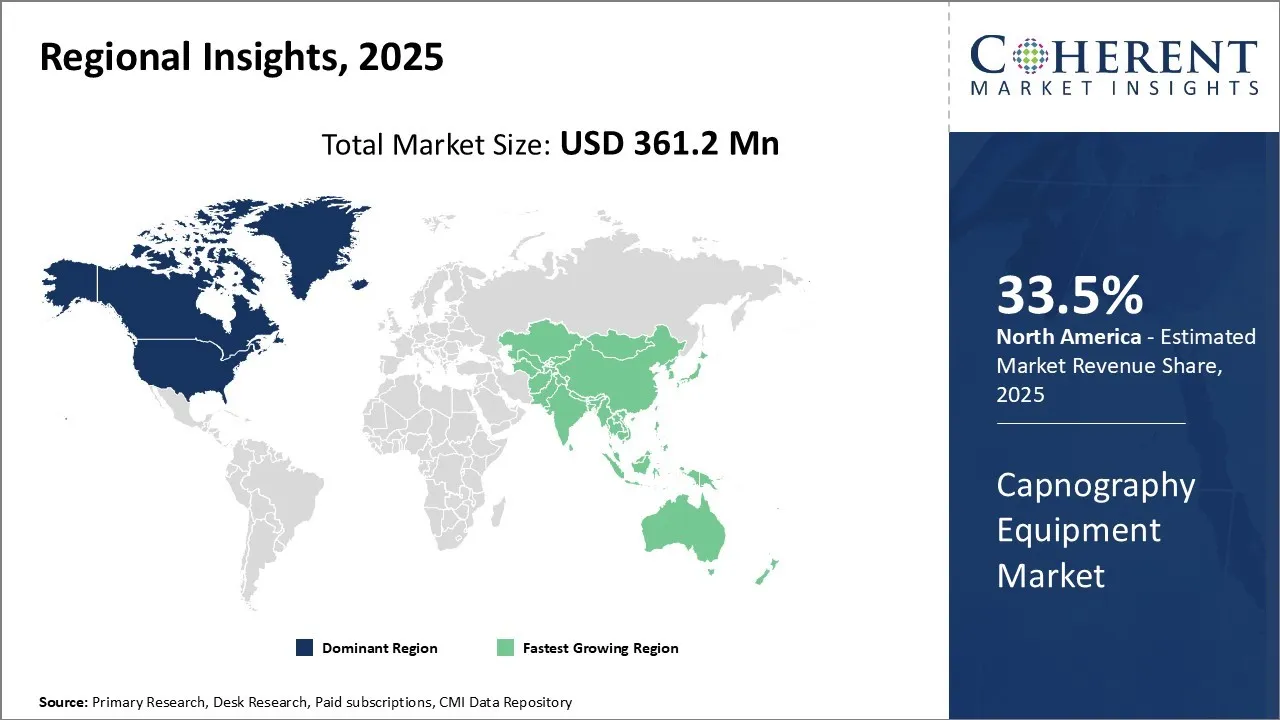
North America is the largest market for capnography equipment, accounting for a share of over 33.5% in 2026. North America, has well-established healthcare systems and access to advanced techniques. Increasing usage of capnography equipment in hospitals and emergency medical service is expected to drive the growth of the region over the forecast period. North America is surging for continuous advancements in capnography devices, including enhanced accuracy and portability, have increased their adoption across various healthcare settings.
Europe is the second-largest market for capnography equipment, accounting for a share of over 26.1% in 2026. Technological advancements and rising prevalence of respiratory diseases such as COPD and asthma, increasing patient safety concerns, and advancements in monitoring technologies in capnography equipment are expected to drive the growth of the region over the forecast period. UK and European guidelines recommend continuous capnography monitoring for all ICU patients with artificial airways.
Asia Pacific is the fastest-growing market for capnography equipment, accounting for a share of over 20.3% in 2026. The development of integrated and multi-parameter monitors with capnography is expected to drive the growth of the region over the forecast period. The demand for multi-parameters monitors is observed to be due to a growing prevalence of chronic disease, an expanding and aging population, increasing healthcare expenditure and advancements in healthcare infrastructure and technology.
In April 2026, Omron Healthcare India partnered with Tricog Health to introduce KeeboHealth, a multi-parameter remote cardiac monitoring solution featuring home ECG devices, blood pressure monitors, and weight scales with AI-driven analytics. It is designed for heart failure management, the system enables clinicians to remotely track vital signs, deliver personalized care, and adjust treatment protocols.
The U.S. capnography equipment market is a world leader, dominated by its mandate regulations by AHA and ASA in anesthesia, procedural sedation and during CPR. In the U.S., it is widely used in the ORs, ICUs, emergency rooms, and ambulances. The Increasing prevalence of COPD, sleep apnea, and opioid overdose cases in the United States is boosting the demand for capnography as it helps in early detection and monitoring respiratory issues. COPD affects over 16 million Americans, with millions more undiagnosed. Another factor that drives the market is a strong push from patient safety and hospital accreditation bodies to reduce adverse respiratory events. The Joint Commission, accredits over 22,000 U.S. healthcare organizations, emphasizes on continuous respiratory monitoring for patients receiving opioids or under moderate sedation. This is further proliferating the capnography equipment market share.
Germany represent one of the largest growth opportunities in the capnography equipment market, driven by its high standard of care and widespread adoption of advanced monitoring technologies across healthcare setting. German hospitals are undergoing digital transformation, investing in smart ICUs, AI-integrated operating rooms, and connected monitoring systems, where installation of capnography is commonly observes, as it is a non-invasive, continuous respiratory care monitoring tool, which fits well into this ecosystem of advanced real-time diagnostics. Germany also has an aging population with higher surgical volumes and ventilator use which further contributes in the growth of the market.
Japan is characterized by the increasing number of surgeries in aging population. Japan has over 29% of Japan’s population is aged 65 and above which is the highest in the world. Elderly people undergo surgeries such as cardiovascular, orthopedic, and gastrointestinal conditions. These elder patients are more vulnerable respiratory complications under anesthesia or sedation, making capnography essential for monitoring. Japanese government initiatives are working towards improving anesthesia safety and ICU monitoring. The Ministry of Health, Labour and Welfare (MHLW) mandates comprehensive monitoring, including capnography, ECG, pulse oximetry, and non-invasive blood pressure, for all general anesthesia cases. Guidelines from the Japanese Society of Anesthesiologists (JSA) recommend routine use of capnography, especially in the elderly and during sedation.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 380.3 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 546.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Drägerwerk AG & Co. KGaA, Masimo Corporation, Medtronic, Inc., Nihon Kohden Corporation, Nonin Medical, Inc., Philips Healthcare, Smiths Medical, and Welch Allyn, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The capnography equipment market value is entering a crucial phase of technological refinement and clinical necessity, and it is a firm view that this segment is under-leveraged relative to its demonstrated impact on patient outcomes. Despite being a standard of care in anesthesiology and intensive care in developed healthcare systems, capnography remains vastly underutilized in emergency and sub-acute settings globally. This presents both a challenge and a strategic inflection point for device manufacturers and policymakers.
Capnography's utility as an early warning indicator is unmatched among non-invasive monitoring modalities. Studies published in the Journal of Emergency Medical Services have shown that continuous waveform capnography reduces unrecognized esophageal intubations to nearly zero when used consistently, an alarming contrast to conventional auscultation methods, which miss up to 17% of such events in high-pressure settings like ambulances and trauma bays. Moreover, real-time end-tidal CO₂ (EtCO₂) trends offer a sensitive proxy for ventilation, perfusion, and metabolic status, leading to data pointing towards pulse oximetry simply cannot provide with the same immediacy or specificity.
The market’s under-penetration in step-down units and general wards is especially concerning. A 2023 multicenter study in Critical Care Medicine showed a 30% reduction in rapid response team activations on general wards equipped with capnography monitoring, underscoring its predictive value for respiratory decompensation. This suggests that the current perception of capnography as an "anesthesia-only" tool is outdated and misaligned with clinical evidence.
Equally important is the evolution of capnography in procedural sedation. As sedation outside the OR, such as in GI suites, radiology, and dental clinics becomes more widespread, capnography is increasingly viewed as a legal safeguard rather than a mere monitoring accessory. For instance, the American Society of Anesthesiologists and the Joint Commission have both reinforced guidelines requiring capnography in moderate to deep sedation procedures, and failure to comply has led to documented liability cases.
On the innovation front, leading OEMs such as Medtronic and Masimo are differentiating through integrated, multi-parameter modules that bundle EtCO₂ with oxygenation and pulse rate data, driving compact and scalable deployment. However, true disruption is likely to come from software: AI-based waveform analysis for early hypoventilation detection, alarm fatigue reduction, and closed-loop ventilation are not hypothetical anymore, they are already being piloted in top-tier hospitals in the U.S. and Germany.
Interviews with clinical engineers, respiratory therapists, anesthesiologists, biomedical technicians, ICU heads, ER doctors, and hospital procurement officers across major global markets.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients