Medical Bionic Implants Market Size and Forecast – 2026 – 2033
The Global Medical Bionic Implants Market size is estimated to be valued at USD 14.5 billion in 2026 and is projected to reach USD 29.3 billion by 2033, growing at a compound annual growth rate (CAGR) of 10.2% during the forecast period from 2026 to 2033.
Global Medical Bionic Implants Market Overview
The growth of the medical bionic implants market is driven by the rising prevalence of chronic diseases, disabilities, and age-related conditions such as hearing loss, limb loss, and organ failure. Increasing demand for advanced prosthetics and neuroprosthetic devices is improving patient mobility and quality of life. Technological advancements in robotics, brain-computer interfaces, and materials science are enhancing implant functionality, durability, and biocompatibility. Growing healthcare expenditure, supportive government initiatives, and rising awareness of assistive technologies further support market expansion. In addition, the increasing adoption of minimally invasive surgeries and integration of AI-enabled smart implants are accelerating the development and adoption of medical bionic solutions globally.
Key Takeaways
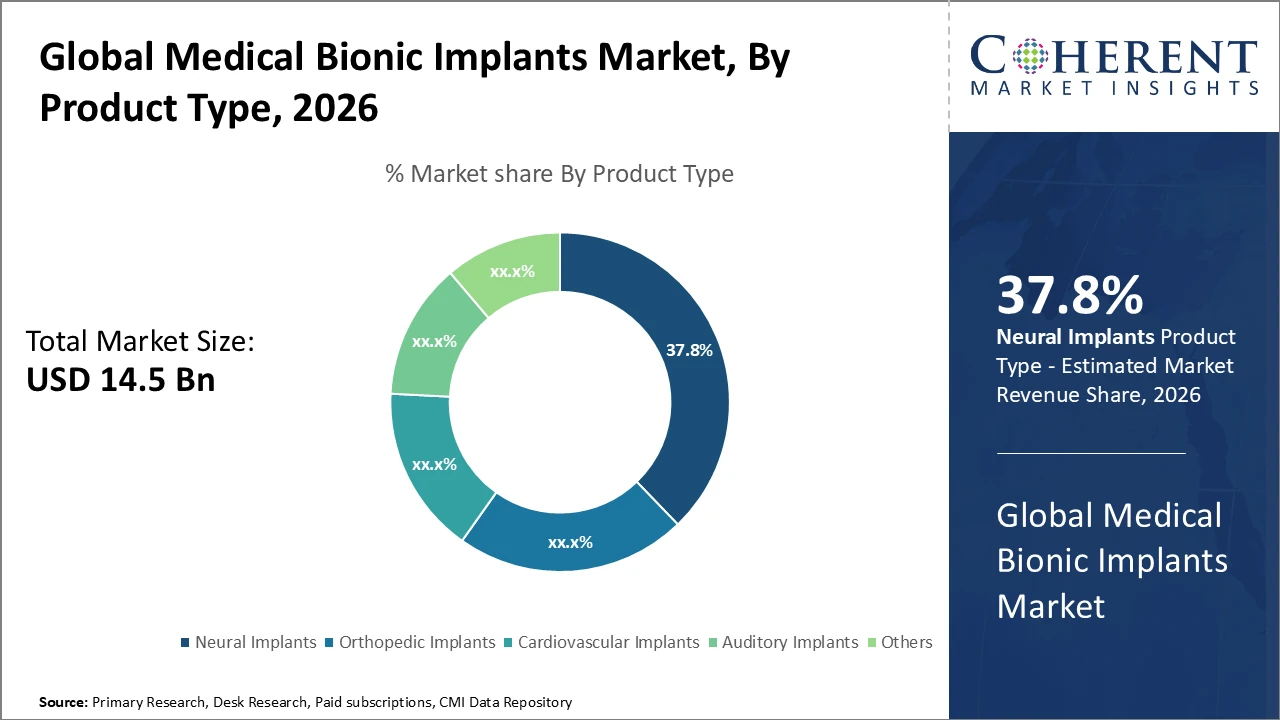
Neural implants segment commands a dominant market share amid rising neurological disorder cases globally, with ongoing R&D driving innovative product launches.
Orthopedic implants are the fastest-growing subsegment, fueled by escalating orthopedic surgeries and enhanced implant longevity.
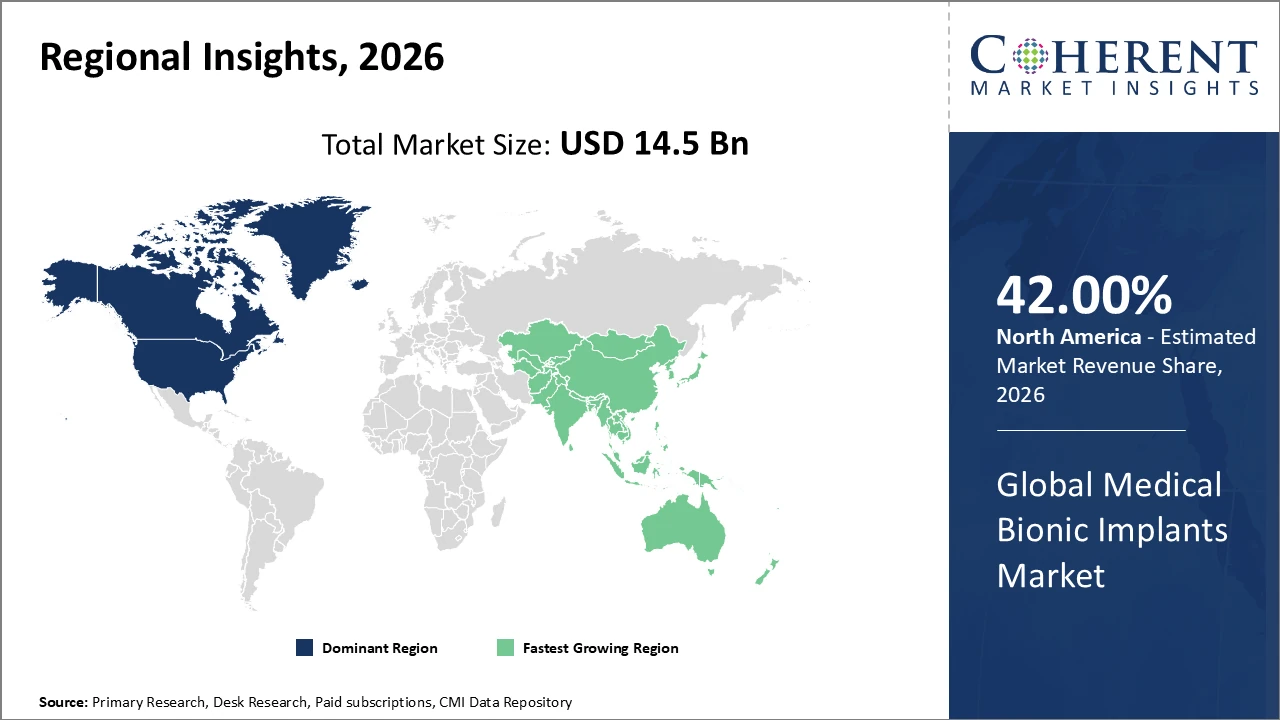
North America maintains dominance with approximately 42% market share, led by substantial healthcare spending and advanced technological integration.
Asia Pacific emerges as the fastest-growing region, reflecting a CAGR exceeding 11%, supported by expanding healthcare infrastructure and rising patient awareness in countries like China and India.
Medical Bionic Implants Market Segmentation Analysis
To learn more about this report, Request Free Sample
Medical Bionic Implants Market Insights, By Product Type
Neural implants dominate the medical bionic implants market due to rising neurological disorder cases and rapid technological advancements enabling effective brain-computer interfaces and sensory restoration. These devices generate substantial revenue through high-value contracts and accelerated clinical adoption. Orthopedic implants are the fastest-growing subsegment, driven by increasing degenerative joint diseases and trauma-related surgeries, supported by advanced materials and minimally invasive techniques. Cardiovascular implants, including pacemakers and defibrillators, grow steadily within a niche application base. Auditory implants address hearing loss, showing moderate growth due to rising sensorineural conditions. Other segments, such as dental and cosmetic implants, maintain smaller but stable contributions to the market.
Medical Bionic Implants Market Insights, By Application
Neurology dominates the medical bionic implants market, driven by the rising prevalence of conditions such as Parkinson’s, epilepsy, and spinal cord injuries, which increase demand for neuroprosthetics. Advances in implantable devices offering precise neurostimulation further strengthen this segment’s leadership. Orthopedics is the fastest-growing segment, supported by the rising number of orthopedic surgeries and improvements in implant materials that enhance durability and biointegration. Cardiovascular implants show steady growth due to an aging population and increasing cardiovascular disease incidence. Ophthalmology applications, including retinal and corneal implants, gain traction through vision restoration innovations. The ‘Others’ category, such as cosmetic implants, presents emerging growth opportunities.
Medical Bionic Implants Market Insights, By Technology
Electroactive bionic implants hold a leading market share due to their precision in replicating biological functions and broad applications, including neural prosthetics and advanced cardiac devices. Their high performance and adaptability continue to attract substantial investment and clinical adoption. Hybrid implants, combining mechanical and electroactive functionalities, represent the fastest-growing segment, driven by personalized solutions and multi-modal therapy potential. Mechanical bionic implants provide durable, reliable options, especially in orthopedic applications, sustaining steady demand through proven clinical efficacy. The Others segment includes emerging technologies utilizing bioresorbable materials and nanotechnology, advancing through early-stage research and development, signaling future market innovation opportunities.
Medical Bionic Implants Market Trends
Market trends show a shift toward personalized bionic implants enhanced by AI and advanced imaging, improving implant survival rates and patient outcomes.
Smart implants with integrated biosensors demonstrated a 15% reduction in postoperative complications in 2025, highlighting their clinical effectiveness.
Evolving regulatory frameworks support faster approvals for breakthrough implant technologies, enabling quicker product launches and influencing market growth.
Medical Bionic Implants Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Medical Bionic Implants Market Analysis and Trends
In North America, the medical bionic implants market is dominated by high healthcare spending, well-established infrastructure, and strong patient affordability. The United States holds the largest share, driven by pioneering clinical trials, advanced research facilities, and the presence of leading bionic implant manufacturers. Government initiatives promoting biomedical innovation, coupled with favorable reimbursement policies, further enhance implant accessibility and adoption. The region’s market share is estimated at around 42%, reflecting its technological leadership and investment in healthcare innovation. Continuous advancements in implant design, regulatory support, and integration of smart and AI-enabled devices are expected to sustain North America’s dominance in the coming years.
Asia Pacific Medical Bionic Implants Market Analysis and Trends
The Asia Pacific region is experiencing the fastest growth in the medical bionic implants market, with countries like China and India leading the surge at a CAGR exceeding 11%. This rapid expansion is driven by increasing healthcare investments, rising prevalence of chronic and degenerative diseases, and a growing middle-class population seeking advanced medical care. Strengthening healthcare infrastructure, including modern hospitals and specialized clinics, supports greater access to bionic implants. Additionally, the adoption of innovative medical technologies, such as AI-enabled implants, neural prosthetics, and minimally invasive devices, is accelerating market penetration. These factors collectively position Asia Pacific as a key growth hub.
Medical Bionic Implants Market Outlook for Key Countries
USA Medical Bionic Implants Market Analysis and Trends
The USA’s medical bionic implants market is driven by significant R&D investment and advanced healthcare infrastructure. Accounting for over 60% of reported neuroimplant cases globally, the country leads in clinical trials and early adoption of technologies such as AI-enabled implants and smart neuroprosthetics. Major U.S.-based companies leverage extensive patent portfolios to maintain a competitive advantage, while government incentives for innovation further support market growth. Comprehensive reimbursement frameworks covering most implant procedures enhance patient accessibility and adoption. These factors collectively contribute to steady business expansion, strong market share, and reinforce the United States’ position as a global leader in the medical bionic implants sector.
Germany Medical Bionic Implants Market Analysis and Trends
Germany is a key leader in Europe’s medical bionic implants market, driven by strict regulatory standards and high-quality manufacturing practices. In 2025, the country recorded a 10% year-on-year increase in implant surgeries, fueled by an aging population and growing demand for orthopedic interventions. Leading domestic companies actively collaborate with research institutions to advance innovation, improving implant durability and patient outcomes. Government initiatives supporting healthcare digitization and medical device research further stimulate market growth. These factors create a strong business ecosystem, attracting investment and fostering technological advancement, resulting in stable revenue streams and reinforcing Germany’s position as a European market frontrunner.
Analyst Opinion
Increasing adoption of next-generation neural implants is a major quantitative driver, with advanced cochlear and retinal implants deployment rising by 18% in North America in 2025, highlighting growing application diversity and expanding market revenue.
Production scalability and supply chain optimizations are boosting manufacturer market share, with several companies increasing production capacities by 25% in 2024 to meet rising clinical demand, resulting in significant revenue growth and stronger competitive positioning.
Pricing dynamics impact market growth, exemplified by a 12% reduction in average implant costs in Europe in 2026 due to improved manufacturing efficiency and economies of scale, driving higher adoption.
Import-export trends influence expansion, as Asia Pacific countries saw a 20% increase in specialized bionic implant imports in 2025, reflecting rapid technology transfer and rising regional healthcare investments.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14.5 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.2% | 2033 Value Projection: | USD 29.3 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Medtronic plc, Stryker Corporation, Cochlear Ltd., Nevro Corp, LivaNova PLC, NuVasive Inc., Cala Health Inc., Inspire Medical Systems, Synapse Biomedical, Abbott Laboratories | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Medical Bionic Implants Market Growth Factors
The growth of the medical bionic implants market is primarily driven by the rising prevalence of chronic neurological and musculoskeletal disorders, with over 275 million orthopedic cases reported globally in 2024 requiring implant interventions. Advances in bioelectronics and implant miniaturization have enhanced patient outcomes and acceptance, further accelerating market expansion. Regulatory approvals for novel devices in 2025 have eased entry barriers, enabling faster commercialization. Additionally, the extension of reimbursement policies by healthcare providers in developed markets has incentivized adoption, evidenced by a 22% increase in insurance coverage for implant surgeries in North America during 2026, supporting sustained market growth.
Medical Bionic Implants Market Development
In July 2025, Neuralink partnered with researchers in Spain and California to initiate a clinical trial for a “Smart Bionic Eye” project.
Key Players
Leading Companies of the Market
Medtronic plc
Stryker Corporation
Abbott Laboratories
Cochlear Ltd.
Nevro Corp
LivaNova PLC
NuVasive Inc.
Cala Health Inc.
Inspire Medical Systems
Synapse Biomedical
Several market players are pursuing strategic mergers and collaborations to expand product portfolios and accelerate clinical trials. For instance, in 2025, a leading neurological device manufacturer partnered with a biotechnology firm to develop next-generation neuroprosthetics, resulting in a 15% increase in market share within the neuroimplant segment. Companies are also integrating AI-driven design and precision manufacturing techniques, which have reduced implant failure rates by over 7% in recent years. These advancements enhance device reliability, boost user adoption, and contribute to significant revenue growth, reinforcing competitive positioning and driving innovation across the medical bionic implants market.
Medical Bionic Implants Market Future Outlook
The future of the medical bionic implants market is poised for significant growth, driven by technological advancements, aging populations, and rising prevalence of chronic diseases. Innovations in neural interfaces, bioelectronic devices, and smart implants are expected to improve precision, functionality, and patient outcomes. Integration with artificial intelligence and wireless technologies will enable more personalized and minimally invasive treatments. Expanding healthcare infrastructure in emerging markets and increasing regulatory support for innovative devices will further fuel adoption. The market is likely to see broader applications across cardiology, neurology, ophthalmology, and orthopedics, enhancing quality of life and creating new opportunities for patient care worldwide.
Medical Bionic Implants Market Historical Analysis
The medical bionic implants market has experienced steady growth over the past decade, driven by advancements in biomedical engineering and increasing demand for restorative healthcare solutions. Early developments focused on cochlear implants and cardiac pacemakers, which laid the foundation for more complex neural and retinal prosthetics. Growing awareness of implantable medical technologies, coupled with rising incidences of chronic conditions such as heart disease, hearing loss, and mobility impairments, accelerated adoption. Technological innovations, including biocompatible materials, miniaturized electronics, and wireless connectivity, improved device safety and functionality. Historically, North America and Europe dominated the market, supported by strong healthcare infrastructure and favorable regulatory frameworks.
Sources
Primary Research Interviews:
Engineers, R&D managers, and project leaders from companies manufacturing bionic implants, neural prosthetics, and orthopedic devices.
Technical specialists from hospitals and rehabilitation centers using bionic implants for patient care.
Industry experts from firms developing AI-enabled, wireless, and smart bionic implants, as well as companies focused on regulatory compliance and clinical trials.
Magazines:
Medical Device and Diagnostic Industry (MD+DI) – Market updates, innovations in bionic implants, and device performance trends.
MedTech Outlook – Insights on emerging technologies in prosthetics, neurostimulation, and retinal implants.
Implant & Biomaterials Magazine – Coverage of new materials, implant design, and clinical applications.
Journals:
Journal of Neural Engineering – Research on neural prosthetics, brain-computer interfaces, and bioelectronic implants.
Biomedical Engineering Online – Studies on bionic devices, sensors, and integration with human physiology.
Journal of Prosthetics and Orthotics – Clinical efficacy, rehabilitation outcomes, and implant innovations.
Newspapers:
The Wall Street Journal – Company strategies, mergers, and investments in bionic implant manufacturers.
Financial Times – Global market developments, regulatory changes, and adoption trends.
Reuters – News on technological breakthroughs and FDA or EMA approvals.
Bloomberg – Corporate financial performance, market forecasts, and emerging regions in bionic implants.
Associations:
International Society for Prosthetics and Orthotics (ISPO) – Standards, training, and research in bionic and prosthetic devices.
American Academy of Orthopaedic Surgeons (AAOS) – Guidelines, innovations, and clinical best practices for implants.
World Health Organization (WHO) – Safety protocols, global standards, and ethical considerations for medical implants.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients