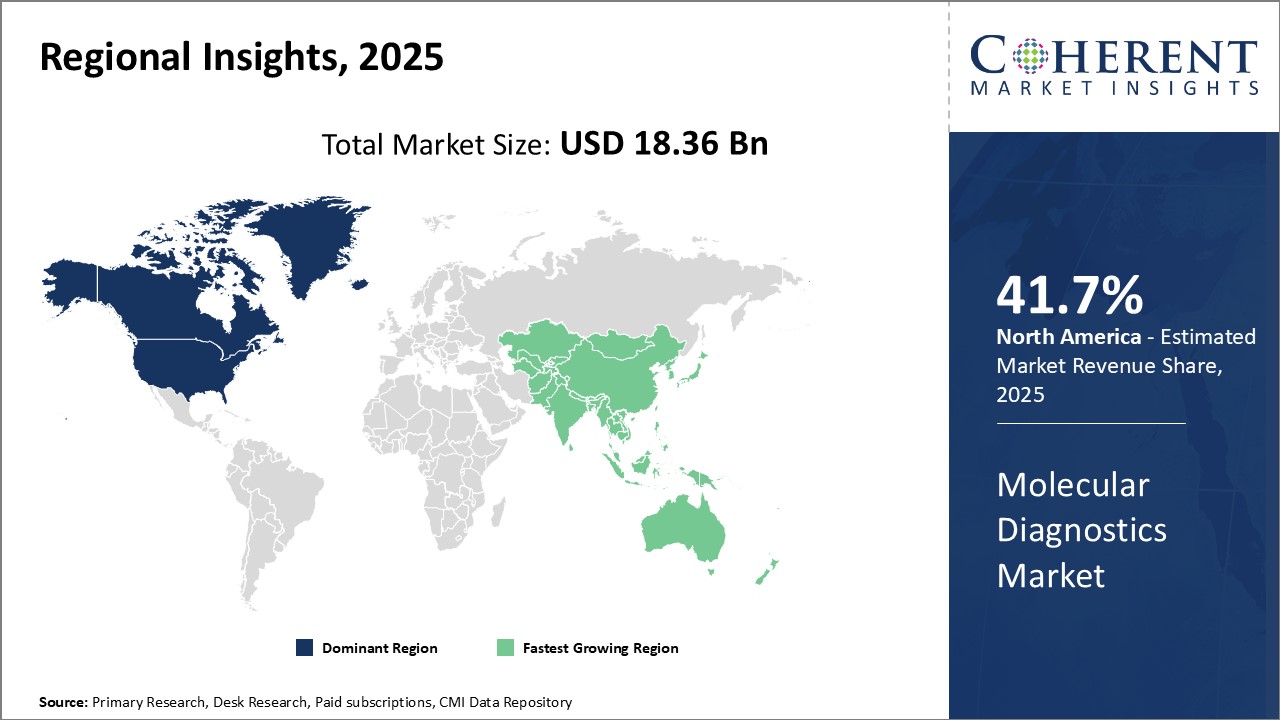
The global molecular diagnostics market is estimated to be valued at USD 18.36 Bn in 2025 and is expected to reach USD 41.63 Bn by 2032, exhibiting a compound annual growth rate (CAGR) of 12.4% from 2025 to 2032.
To learn more about this report, Download Free Sample
The increasing prevalence of infectious diseases and genetic disorders, technological advancements in molecular diagnostics, and growing application areas of polymerase chain reaction (PCR) and next-generation sequencing (NGS) are significantly boosting molecular diagnostics market demand. These factors collectively enhance the need for rapid, accurate, and scalable diagnostic solutions, fueling sustained market growth across clinical and research settings.
|
Current Event |
Description and its Impact |
|
Regional Healthcare Investment Shifts |
|
|
Economic Pressures and Healthcare Spending |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial intelligence (AI) is transforming the molecular diagnostics market by enhancing the speed, accuracy, and scalability of disease detection and analysis. AI algorithms are increasingly used to interpret complex genomic and biomarker data generated from techniques like PCR and next-generation sequencing (NGS). These tools help identify patterns, predict disease risks, and personalize treatment plans with greater precision. AI also streamlines workflows by automating data processing, reducing manual errors, and accelerating diagnostic turnaround times. In oncology, infectious disease control, and genetic screening, AI-powered platforms are enabling real-time insights and supporting point-of-care diagnostics. As AI integration deepens, it is expected to significantly boost the molecular diagnostics market’s value and global adoption.
For instance, in July 2025, PathAI launched the Precision Pathology Network to advance AI-powered pathology and precision medicine. The initiative connects leading labs and biopharma partners to deliver scalable, digital pathology solutions. By integrating AI with biomarker analysis and clinical workflows, PathAI aims to enhance diagnostic accuracy, support clinical trials, and accelerate personalized treatment development.
In terms of product type, the reagents & kits segment is expected to contribute the highest share of the market with 65.5% in 2025 driven by the continuous need for consumables in diagnostic workflows. Molecular tests rely heavily on reagents, probes, and primers to process samples and deliver accurate results. As testing volumes and complexity rise, demand for these materials grows steadily. Ready-to-use kits simplify procedures and ensure reproducibility, making them indispensable for labs. Unlike instruments, reagents have short replacement cycles and are consumed per test, ensuring recurring purchases. Their role in automation and workflow optimization, combined with stable end-user demand, supports consistent growth and market leadership.
For instance, in May 2024, Polaris launched a premium line of IVD-certified molecular diagnostics kits, enhancing clinical testing capabilities with high-quality reagents. Designed for precision and reliability, these kits support a wide range of diagnostic applications. The new product line reflects Polaris’s commitment to advancing molecular diagnostics through innovation and compliance with international standards.
In terms of application, the virology segment is expected to contribute the highest share of the market in 2025 due to its vital role in detecting and managing viral infections. The global focus on public health, intensified by recent pandemics, has driven widespread adoption of molecular tools like PCR and next-generation sequencing for accurate viral detection. Diseases such as HIV, hepatitis, and influenza continue to pose significant threats, requiring ongoing surveillance and rapid diagnostics. Additionally, advancements in point-of-care testing and multiplex assays have made virology diagnostics more accessible and efficient, reinforcing its dominance in clinical settings and public health initiatives worldwide.
For instance, September 2024, Roche launched the cobas® Respiratory Panel with its novel TAGS technology, enabling simultaneous detection of 12 common respiratory viruses, including SARS-CoV-2, influenza, and RSV. Designed for use on the cobas 5800/6800/8800 systems, this high-throughput molecular test enhances diagnostic accuracy and efficiency, supporting timely clinical decisions during respiratory infection seasons.
In terms of end user, the hospitals and diagnostic laboratories segment is expected to contribute the highest share of the market with 46.5% in 2025 driven by centralized testing of complex assays. These facilities consolidate tests to achieve economies of scale, leveraging automation, multiplexing, and skilled technologists for high-volume, rapid processing. Accreditation from bodies like AABB and CAP enhances credibility, while expanded test menus attract referrals. Hospitals absorb capital costs better and negotiate favorable terms with IVD suppliers, offering competitive pricing. Their ability to provide one-stop diagnostic services boosts demand, and centralized labs ensure faster turnaround, real-time reporting, and consistent quality, key factors fueling their leadership in molecular diagnostics adoption.
For instance, in October 2025, Fortis Hospital Mulund inaugurated a new Cancer and Gastrosciences wing, enhancing its capabilities in oncology and gastrointestinal care. The facility aims to provide advanced diagnostics, personalized treatment, and multidisciplinary expertise. With cutting-edge infrastructure and specialized teams, the new wing strengthens Fortis’s commitment to comprehensive, high-quality care for complex medical conditions.
To learn more about this report, Download Free Sample
North America dominates the global molecular diagnostics market, and is expected to hold 41.7% share of the market in 2025. The U.S. accounts for the largest share primarily due to advancements in genomic and protein-based technologies. Local companies have been investing heavily in the research and development of novel molecular diagnostic tools and platforms. Additionally, favorable regulatory policies have enabled companies to introduce innovative products at a rapid pace. The presence of several leading global companies has created an ecosystem that promotes technological advancement in molecular diagnostics. These industry leaders have large distributions networks that ensure wide access and adoption of new techniques across the region.
For instance, in October 2025, Roche received FDA clearance for its Elecsys® pTau181 blood test, a breakthrough in Alzheimer’s diagnostics. Designed to detect phosphorylated Tau protein in plasma, the test helps rule out Alzheimer’s-related amyloid pathology in patients with early cognitive symptoms. This minimally invasive, high-throughput solution enhances diagnostic accuracy and expands molecular testing in neurology.
Asia Pacific represents the fastest growing regional market for molecular diagnostics. The growth can be attributed to rising healthcare investments by governments as well as expanding private sector involvement. Countries such as China and India offer low-cost manufacturing environments that have attracted global companies to establish production facilities. This has enhanced the availability of affordable molecular diagnostic solutions and made advanced techniques more accessible. Furthermore, increasing awareness regarding personalized medicine and companion diagnostics among doctors as well as patients has boosted demand. The large patient populations and growing incidence of chronic and infectious diseases also present lucrative opportunities. Export opportunities are rising as quality standards improve and regulatory systems become more robust.
For instance, in October 2025, NUH and NUHS launched the Molecular Imaging and Theranostic Centre in Singapore to advance patient care and research. The facility integrates cutting-edge imaging and targeted therapies, supporting precision medicine across oncology and other specialties. This initiative strengthens diagnostic capabilities and fosters innovation in molecular diagnostics and personalized treatment strategies.
The U.S. Molecular Diagnostics Market is thriving due to high disease prevalence, advanced healthcare infrastructure, and strong adoption of technologies like PCR and NGS. Government support, robust R&D, and widespread clinical integration drive demand for rapid, accurate testing. Personalized medicine and early disease detection further fuel growth across hospitals, labs, and research institutions.
For instance, in July 2025, Orochem Technologies Inc. and Vision Biotechnology launched Vision Diagnostics, a new venture focused on advancing molecular diagnostics. Based in the U.S., the initiative aims to deliver innovative solutions for infectious disease detection and genetic testing. Combining biotechnology expertise with scalable manufacturing, Vision Diagnostics supports precision medicine and global healthcare needs.
China’s Molecular Diagnostics Market is booming due to rising infectious disease cases, cancer prevalence, and demand for early detection. Government investments in biotech, expanding healthcare access, and adoption of advanced technologies like PCR and NGS fuel growth. The push for precision medicine and local manufacturing further accelerates diagnostic innovation and accessibility.
For instance, in July 2025, Paragon Genomics and Genecast announced a global partnership to advance precision oncology through next-generation sequencing. By integrating Paragon’s CleanPlex® technology with Genecast’s Focus Assay, the collaboration aims to deliver high-sensitivity molecular diagnostics for solid tumors. The initiative spans the U.S. and China, enhancing access to personalized cancer testing worldwide.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2024 | Market Size in 2025: | USD 18.36 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2025 To 2032 |
| Forecast Period 2025 to 2032 CAGR: | 12.4% | 2032 Value Projection: | USD 41.63 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
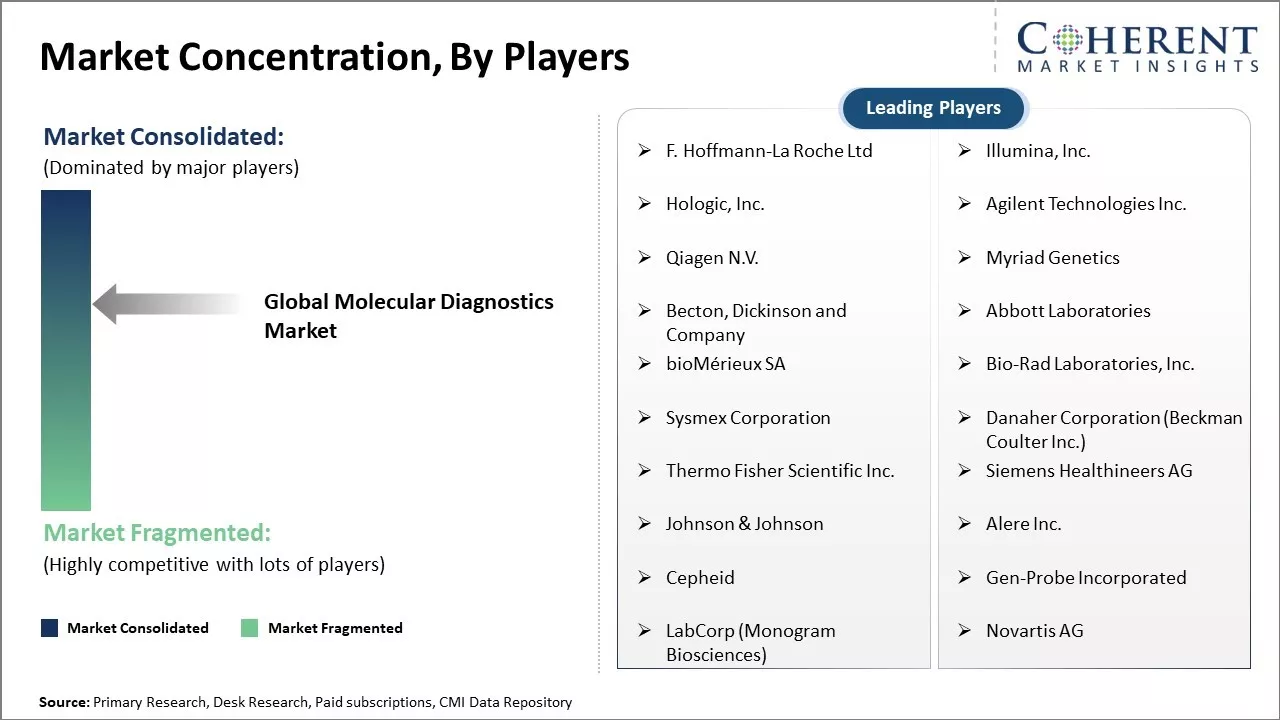
F. Hoffmann-La Roche Ltd, Illumina, Inc., Hologic, Inc., Agilent Technologies Inc., Qiagen N.V., Myriad Genetics, Becton, Dickinson and Company, Abbott Laboratories, bioMérieux SA, Bio-Rad Laboratories, Inc., Sysmex Corporation, Danaher Corporation (Beckman Coulter Inc.), Thermo Fisher Scientific Inc., Siemens Healthineers AG, Johnson & Johnson, Alere Inc., Cepheid, Gen-Probe Incorporated, LabCorp (Monogram Biosciences), and STNovartis AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
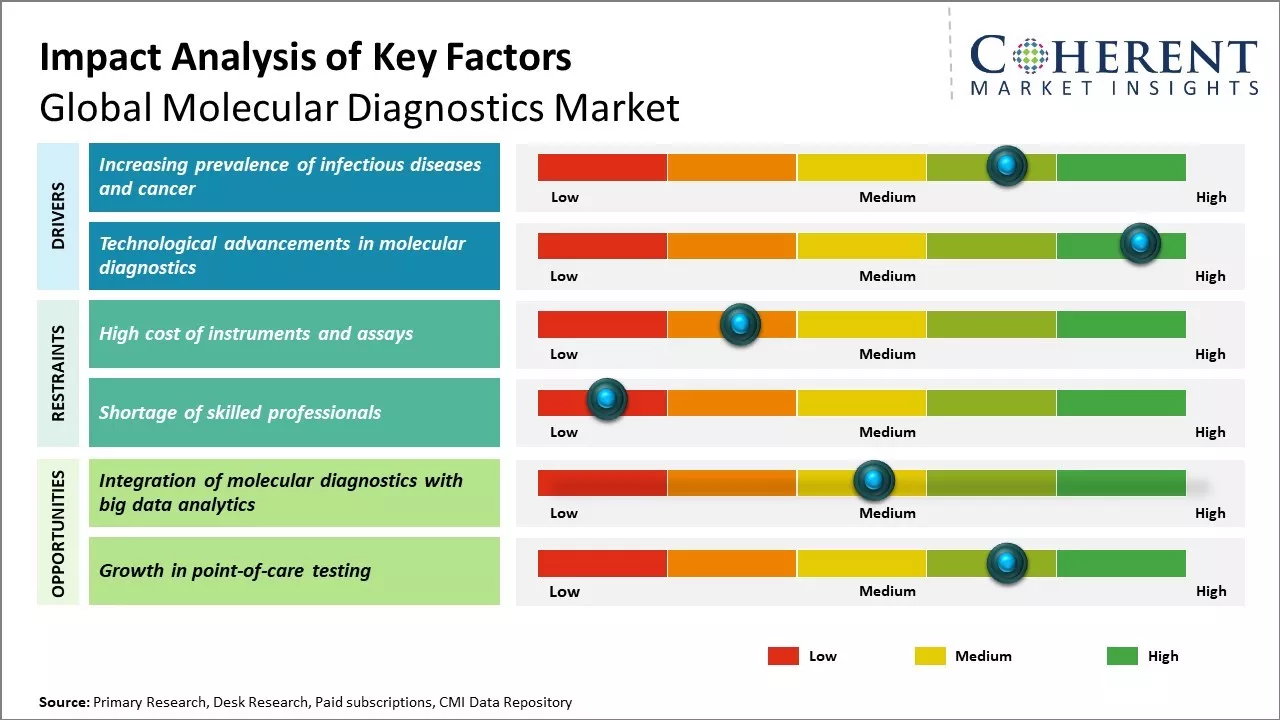
Rising cases of infectious diseases like HIV, hepatitis, and tuberculosis, along with increasing global cancer rates, are driving demand for advanced molecular diagnostic techniques. These tests offer rapid, highly sensitive detection, identifying TB in hours and pinpointing cancer at the genetic level for early intervention. Traditional methods often fall short in speed and accuracy. Molecular diagnostics enable timely, personalized treatment, improving survival rates. With the global cancer burden expected to rise, oncologists increasingly rely on these tools. As a result, the molecular diagnostics market share is expanding rapidly, fueled by innovation and the need for precise, early disease detection.
To learn more about this report, Download Free Sample
Technological innovations are rapidly expanding the molecular diagnostics market value. Next-generation sequencing enables fast, comprehensive DNA/RNA profiling, while synthetic biology and machine learning enhance biomarker detection and data analysis. Miniaturized devices using microfluidics and nanotech offer portable, on-site testing with rapid results. Examples include handheld PCR analyzers and mini-labs. These tools support decentralized healthcare, especially in infectious disease control. Automation boosts efficiency and reduces errors, while smartphone integration is being explored for point-of-care diagnostics. Declining costs and improved accessibility make molecular diagnostics more powerful, convenient, and affordable, driving global adoption across clinical and research settings.
The integration of molecular diagnostics with big data analytics offers major growth potential for the molecular diagnostics market forecast. Molecular testing generates vast genomic and biomarker data, which big data tools help store, manage, and analyze. Linking diagnostics with EHRs and real-world data enables large-scale insights for personalized medicine, outbreak surveillance, and population health. This data-driven approach enhances clinical value, reduces costs, and improves access. It also supports diagnostics development and early disease detection. As stakeholders recognize these benefits, the adoption of molecular diagnostics is expected to accelerate globally, driving innovation and expanding market opportunities in the years ahead.
The molecular diagnostics market is undergoing structural transformation, where sustainable value increasingly resides in reagents and proprietary assays rather than instruments. Reagents and kits contribute nearly two-thirds of total market revenue, emphasizing the dominance of consumable-driven business models. Companies with integrated reagent supply chains and strong IP are better positioned to sustain profitability, while hardware-focused firms face intensifying margin pressure and must pivot toward recurring-revenue streams such as consumables, analytics, and services.
A clear bifurcation in innovation is evident: rapid, decentralized molecular point-of-care solutions compete alongside high-content sequencing and liquid-biopsy applications serving oncology and precision medicine. However, PCR continues to anchor routine testing, while sequencing gains traction only where clinical and reimbursement frameworks justify premium pricing.
Regional concentration remains pronounced, with North America accounting for roughly 40% of market revenues, creating both opportunity and exposure to regulatory and reimbursement volatility. Concurrently, portfolio realignments and diagnostics divestitures among major players signal growing investor preference for focused, data-driven models.
Success in this evolving landscape will depend on three factors: reagent monetization, clinical data integration, and payer-aligned evidence generation. Firms that blend technological capability with recurring consumable economics and clinically validated outcomes will emerge as the enduring leaders in molecular diagnostics.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients