Radioactive Tracer Market is estimated to be valued at USD 4.00 Bn in 2026 and is expected to reach USD 6.59 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.4% from 2026 to 2033.
Radioactive tracers are radioactive elements that are used to diagnose and treat a variety of diseases. These are administered into the body and monitored by using specialized cameras to image the body’s internal structures. Growing prevalence of chronic diseases such as cancer and cardiovascular diseases boosts demand for radioactive tracers. Key drivers of the market include rising adoption of nuclear medicines, increasing healthcare expenditure, and growing cancer research funding.
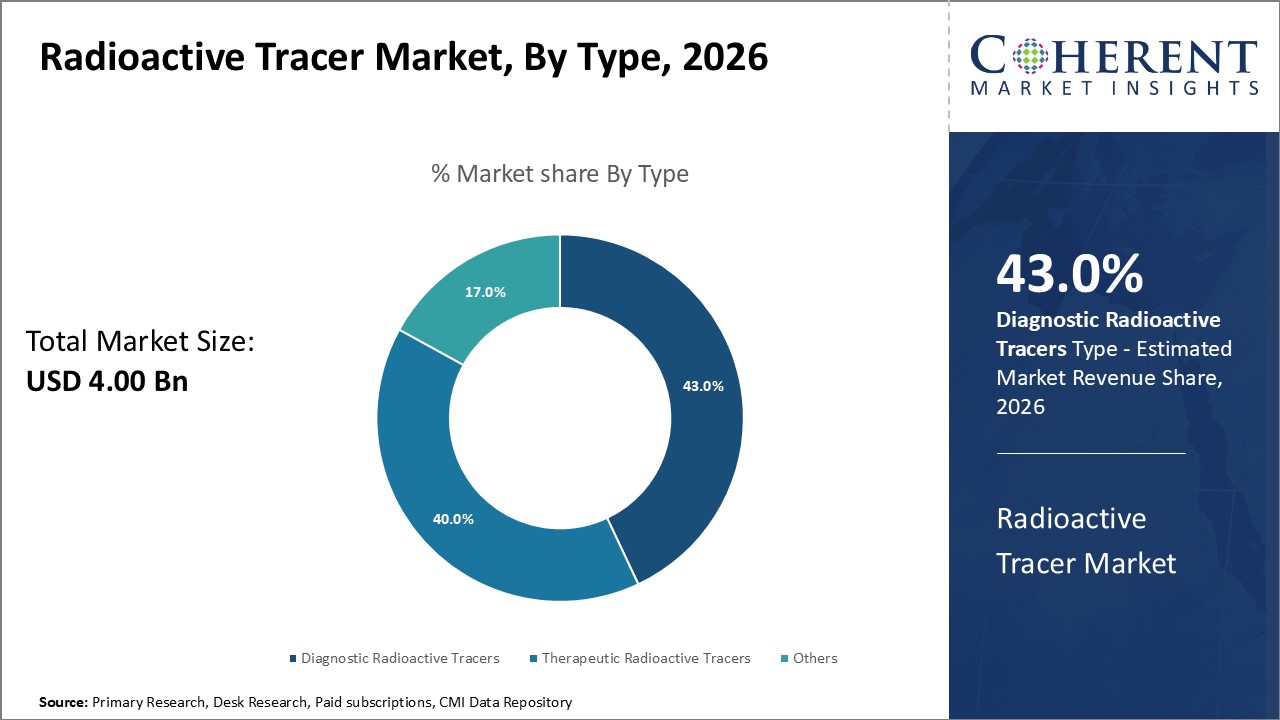
Global radioactive tracer market is segmented by type, application, end user, and region. By type, the market is segmented into diagnostic radioactive tracers, therapeutic radioactive tracers, and others. The diagnostic radioactive tracers segment accounts for the largest market share due to the increasing use of PET and SPECT imaging. By application, the oncology segment dominates the market due to high incidence of cancer globally.
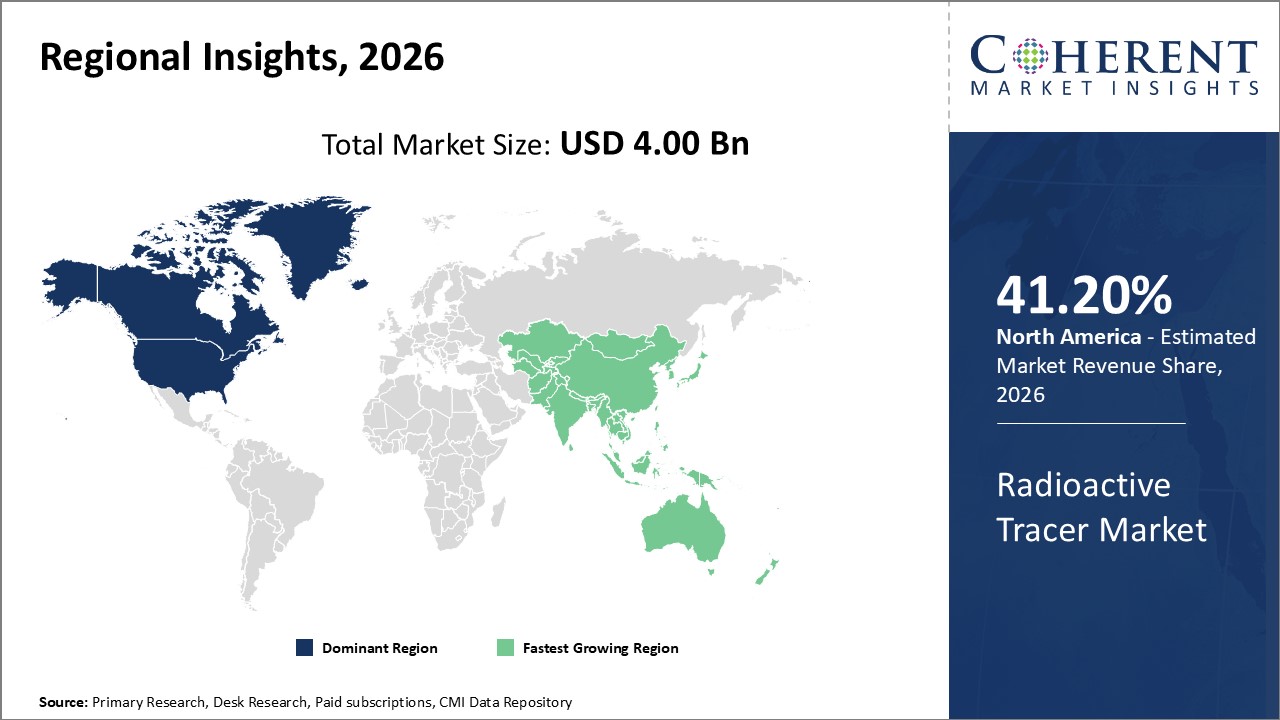
Global Radioactive Tracer Market- Regional Insights
- North America is expected to be the dominant in radioactive tracer market during the forecast period, accounting for over 41.2% of the market share in 2026. The growth of the market in North America is attributed to the high adoption of nuclear medicines, presence of major players, and increasing prevalence of chronic diseases.
- Europe is expected to be the second-largest market for radioactive tracer, accounting for over 28.7% of the market share in 2026. The growth of the market in Europe is attributed to the rising focus on alpha radio-immunotherapies and increasing research funding.
- Asia Pacific is expected to be the fastest-growing market for radioactive tracer, with a CAGR of over 22.3% during the forecast period. The growth of the market in Asia Pacific is attributed to the rapidly growing healthcare sector, increasing healthcare expenditure, and rising prevalence of cancer in the region.
Analyst View: Global radioactive tracer market is expected to offer promising growth opportunities in the near future. Rising prevalence of chronic diseases such as cancer is a major factor that boosts demand for radio pharmaceuticals that are used in diagnosis and treatment of cancer. Moreover, growing geriatric population prone to developing such diseases can bolster market revenues. Advancements in molecular imaging technologies have enhanced precision of radioactive tracers, and thus, enabling better diagnosis. This has increased clinical utility of tracers in varied applications. However, stringent regulations pertaining to radioactive material handling may hamper market expansion to some extent. North America currently dominates the market, and is likely to continue its leadership through the forecast period. Large patient population diagnosed with cancer and cardiovascular diseases drives the market growth. Meanwhile, Asia Pacific is projected to witness growth, owing to improving healthcare expenditures and healthcare infrastructure across developing nations. Growing public and private funding for academic research in radiopharmaceutical development as well as nuclear medicine programs drives the Asia Pacific market. Favorable reimbursement policies for medical isotope production and radiopharmaceutical application also drive the market growth. However, shortage of molybdenum-99, the parent isotope required for producing a host of common medical radioisotopes, remains a challenge to be addressed. Manufacturers are actively diversifying raw material sourcing and production capabilities to overcome this supply issue.
Figure 1. Global Radioactive Tracer Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Radioactive Tracer Market- Drivers
- Increasing prevalence of chronic diseases such as cancer, cardiovascular diseases, and neurological disorders is expected to boost demand for radioactive tracers. The growing burden of chronic illnesses globally has led to increase in diagnostic procedures involving nuclear medicine imaging techniques such as PET and SPECT. These utilize radioactive tracers to visualize, characterize and measure processes in the body. According to the WHO, by 2030, chronic disease prevalence is expected to rise by 58%. The high sensitivity of techniques like PET scans boosts its adoption for disease staging, monitoring treatment effectiveness and disease recurrence.
- Technological advancements in clinical imaging are anticipated to increase adoption of radioactive tracers. Ongoing developments to improve image quality, resolution and 3D reconstruction have enhanced the capability of diagnostic equipment using radioactive tracers. Vendors are focused on integrating imaging modalities and introducing multi-modal systems. For instance, the introduction of integrated PET-MRI systems enables superior imaging with the combination of anatomical and functional data. Furthermore, the development of novel diagnostic agents is expanding the applications of radioactive tracers.
- Growing geriatric population susceptible to chronic conditions is expected to drive the radioactive tracer market growth. According to UN population estimates, the global population aged 65 years or over is projected to grow from 727 million in 2020 to 1.5 billion in 2050. Aging is a significant risk factor for oncological, cardiac, neurological and other conditions. This is anticipated to boost usage of nuclear imaging tests and procedures using radioactive tracers for diagnosis and treatment monitoring.
- Investments to enhance production capacities and supply of radioisotopes are projected to favor radioactive tracer market expansion. Initiatives by governments and private players to increase isotope production and processing facilities to meet growing radiopharmaceutical demand offers opportunities for the market growth. For instance, in 2021, the U.S. Department of Energy (DOE) announced plans to invest US$ 45 million in building a technetium-99m production facility in North America by end of 2026. Such investments secure supplies improving tracer availability.
Global Radioactive Tracer Market- Opportunities
- Emerging applications in oncology such as theranostics provide significant growth opportunities. Targeted alpha therapy using alpha-emitting radioactive isotopes conjugated to tumor-specific molecules is an emerging therapeutic approach. Alpha emitters such as Actinium-225 have gained interest for the treatment of metastatic cancers. Advanced clinical trials assessing the efficacy of alpha-labeled radiopharmaceuticals will drive its adoption. Moreover, the use of matched diagnostic and therapeutic pairs allows personalized therapy.
- The growing focus on radioactive tracers for neurological applications presents future prospects. Beta-amyloid binding radiotracers have shown promise in detecting beta-amyloid plaque deposits in Alzheimer's disease. In 2020, U.S. FDA approved AVID Radiopharmaceutical's [18F] flutemetamol injection to estimate beta-amyloid neuritic plaque density. Tau imaging tracers that bind to tau protein aggregates have potential for diagnosis of tauopathies. Investments in R&D will expand radiotracer applications in neurology.
- Increasing demand for radioisotopes from emerging countries offers lucrative opportunities. Growing investments in healthcare infrastructure, rising income levels and increasing prevalence of lifestyle diseases in developing nations is escalating usage of nuclear medicine diagnostics. Isotope suppliers are through new production facilities and distribution partnerships. Localizing production and enhancing supply chains will support tracer adoption.
- Advancements in radioactive tracer using alternative isotope production methods could enable market expansion. Accelerator technology shows promise as an alternative for producing radioisotopes as it offers advantages like reduced nuclear waste. Approaches using cyclotrons and linear accelerators are being explored for wider commercial applications. Further technological improvements and decline in accelerator prices will support decentralized tracer production.
Radioactive Tracer Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.00 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.4% | 2033 Value Projection: | USD 6.59 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
PerkinElmer Inc., Cardinal Health, Bracco, NTP Radioisotopes, Nordion (Canada) Inc., Curium, IRE, Telix Pharmaceuticals Limited, NorthStar Medical Radioisotopes, LLC, Lantheus Holdings, Inc., Eckert & Ziegler, Siemens Healthineers, GE Healthcare, Mallinckrodt Nuclear Medicine LLC , Jubilant DraxImage Inc., Sumitomo Heavy Industries, Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Radioactive Tracer Market- Trends
- Precision diagnostic and therapeutic radiopharmaceuticals tailored to molecular targets are a key trend. Companies are developing tumor-specific radioactive tracers by conjugating radioisotopes with antibodies, peptides, small molecules, nanobodies, affibodies, and others. Emerging targeted radiotracers like prostate-specific membrane antigen (PSMA) ligands for prostate cancer improve diagnostic accuracy. Moreover, molecularly targeted alpha and beta-emitting therapies maximize dose to tumors while minimizing toxicity.
- Hybrid tracer systems combining radioactive materials with other moieties are gaining traction. Radiolabeled nanoparticles, vesicles, micelles and dendrimers are novel platforms being studied for enhanced tumor targeting and imaging sensitivity. Combination with MRI contrast agents, fluorescent materials, and others enables multimodal imaging. Combining radioisotopes with chemotherapeutics, photosensitizers or immune adjuvants is being explored for synergistic therapeutic effects.
- Implementation of automation and artificial intelligence is increasing for radioactive tracer synthesis, analysis and imaging applications. Automated radiochemistry systems and AI-based image reconstruction solutions help to improve efficiency, standardization and accuracy. Key players are integrating automation and AI through partnerships or in-house development. Adoption of big data analytics and blockchain for tracking radioactive materials supply chain is also rising.
- Transition towards value-based healthcare models is increasing focus on generation to clinical utility of novel tracers. Regulatory emphasis on analytical and clinical validity of radiopharmaceuticals is growing. Payers are also demanding evidence of improved health outcomes for coverage. As a result, investments in conducting comparative effectiveness studies to support real-world performance increases.
Global Radioactive Tracer Market- Restraints
- Short half-life of radiopharmaceuticals: The short half-lives of commonly used radioisotopes used in radioactive tracers poses logistical challenges impacting clinical adoption. For instance, fluorine-18 used in FDG has a half-life of approximately 110 min, limiting its availability. Short half-lives necessitate specialized infrastructure for production and fast delivery to clinical sites. Centralized manufacturing leads to higher costs and reliability concerns. Decentralized supply chain models are still emerging.
- High costs associated with radioactive tracers: High costs related to radioactive tracers and. Radioisotope production in nuclear reactors and accelerators requires heavy capital investments. Compliance with strict regulatory standards for radiopharmaceutical manufacturing and handling also adds costs. Prices for single patient doses for many tracers range from US$ 1000-3000. Reimbursement restrictions can affect affordability.
- Stringent regulatory policies: Stringent regulatory policies for approval and manufacturing of radioactive tracers hampers rapid clinical translation of novel radiopharmaceuticals. Complex approval procedures due to safety and efficacy concerns deter investment. Adherence to cGMP, radiation safety guidelines and specialized facilities add time and expenses to development. Harmonization of global regulations will be needed to accelerate access.
- Limited reimbursements policies: Limited reimbursement for diagnostic radiotracers and emerging alpha/beta-emitting radiotherapies constrains the market penetration. Lack of reimbursement codes and coverage policies for novel PET tracers affects clinical adoption. Outpatient payment rules for therapeutic radiopharmaceuticals also limit revenue potential. Dialogue between stakeholders to align value demonstration with payer evidence needs is required to improve access.
Global Radioactive Tracer Market- Recent Developments:
New Product Launches:
- On July 28, 2023, Curium, a leader in nuclear medicine, announced that the European Commission had granted marketing authorization for PYLCLARI (INN: Piflufolastat (18F) formerly known as (18F)-DCFPyL) indicated for the detection of prostate-specific membrane antigen (PSMA) positive lesions with positron emission tomography (PET) in adults with prostate cancer (PCa)
- On May 27, 2021, Lantheus Holdings, Inc., a leader and fully integrated provider of innovative imaging diagnostics and targeted therapeutics, announced that the U.S. Food and Drug Administration (FDA) hasd approved PYLARIFY, an F 18-labeled prostate-specific membrane antigen (PSMA) targeted positron emission tomography (PET) imaging agent to identify suspected metastasis or recurrence of prostate cancer. PYLARIFY is the first and only commercially available approved PSMA PET imaging agent for prostate cancer.
- In September 2020, Telix Pharmaceuticals , a pharmaceutical company, and ANMI announced the first commercial production of TLX250-CDx, a 89Zr-labelled antibody for imaging multiple myeloma
Acquisition, Collaboration and Partnerships:
- On September 13, 2023, Telix Pharmaceuticals Limited., a pharmaceutical company, announced collaboration with Eckert & Ziegler, a radio tracer manufacturer, and RefleXion Medical, a pharmaceutical company to jointly develop a new concept – Satellite Hot Labs (SHLs). SHLs are designed specifically for radiolabeling, quality control and storage of molecular targeting radiotracers, referred to as Bio Guides that are used in biology-guided radiotherapy (BgRT) to direct the radiation beam to indicated solid tumor cancers.
- On September 7, 2023, Curium, a leader in nuclear medicine, announced that it had entered into an exclusive rights agreement with b.e. Imaging, a imaging center, for the distribution of PYLCLARI (INN: Piflufolastat (18F) formerly known as (18F)-DCFPyL), indicated for the detection of prostate-specific membrane antigen (PSMA) positive lesions with positron emission tomography in adults with prostate cancer. Under the terms of the agreement, b.e. Imaging will be responsible for obtaining marketing authorization from Swiss competent authority Swissmedic, and will have exclusive distribution rights for PYLCLARI across Switzerland.
- On June 27, 2023, NorthStar Medical Radioisotopes, LLC, a global innovator in the development, production and commercialization of radiopharmaceuticals used for therapeutic applications and medical imaging, and Nucleus RadioPharma, a full-service contract development and manufacturing organization (CDMO) dedicated to building robust and reliable clinical and commercial supply chains for targeted radiotherapies, announced the signing of a supply agreement for the therapeutic radioisotope actinium-225 (Ac-225). Under terms of the agreement, NorthStar will supply its high purity, non-carrier added (n.c.a.) Ac-225 to Nucleus. Nucleus will use NorthStar’s Ac-225 for their customers’ radioligand pharmaceutical programs.
- On June 21, 2023, Telix Pharmaceuticals Limited., a pharmaceutical company, announced acquisition of Lightpoint Medical, a medical device company specializing in the intra-operative detection of targeted radiopharmaceuticals, to expand into other urologic and non-urologic malignancies
- In June 2021, Curium , a biotechnology company, acquired IASON GmbH, a biotechnology company, to strengthen its position in the PET imaging market. This allowed Curium to expand its offerings for neurology, oncology, and cardiology applications.
- In November 2020, Telix Pharmaceuticals, a pharmaceutical company, partnered with China Grand Pharma, a pharmaceutical company, to distribute illuccix, a kidney cancer imaging agent, in China. This partnership expanded Telix's presence in the Chinese radiopharmaceuticals market.
Figure 2. Global Radioactive Tracer Market Share (%), By Type, 2026
To learn more about this report, Request Free Sample
Top companies in Global Radioactive Tracer Market:
- PerkinElmer Inc.
- Cardinal Health
- Bracco
- NTP Radioisotopes
- Nordion (Canada) Inc.
- Curium
- IRE
- Telix Pharmaceuticals Limited
- NorthStar Medical Radioisotopes, LLC
- Lantheus Holdings, Inc.
- Eckert & Ziegler
- Siemens Healthineers
- GE Healthcare
- Mallinckrodt Nuclear Medicine LLC
- Jubilant DraxImage Inc.
- Sumitomo Heavy Industries, Ltd.
Sources
Primary Research Interviews
- Radioactive tracer producers (e.g., Curium, GE Healthcare, Lantheus Medical Imaging, Cardinal Health, Bracco Imaging)
- Nuclear medicine physicians and PET imaging specialists
- Cyclotron and radiopharmacy operators
Databases
- U.S. FDA radiopharmaceutical approvals
- EMA EPAR
- IAEA radiopharmaceutical data
Magazines
- Applied Radiology
- Diagnostic Imaging
Journals
- Journal of Nuclear Medicine (SNMMI)
- European Journal of Nuclear Medicine and Molecular Imaging (EANM)
Newspapers
- Reuters Health
- Financial Times – MedTech
Associations
- SNMMI
- EANM
- IAEA
Public Domain Sources
- U.S. FDA radiopharmaceutical guidance
- IAEA publications
- ResearchGate publications on PET tracers
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
*Definition: Radioactive tracer market refers to the industry and scientific field involving the production, sales, and usage of radioactive tracers for diagnostic and therapeutic applications in medicine. Radioactive tracers are radioactive isotopes that are chemically bonded to other molecules and introduced into the body to map biological processes for imaging. These emit signals that are detected by specialized cameras or imaging equipment. The market comprises companies manufacturing radioactive tracers, distribution firms, nuclear pharmacies, hospitals, cancer research centers and other end users. Key applications include cancer diagnosis and staging, cardiac imaging, neurological disorders, thyroid disorders, and others. The market is driven by the rising prevalence of chronic diseases, adoption of nuclear imaging modalities, and investments in radioisotope production.
Few Other Promising Reports in Medical Devices Industry
Cardiovascular Monitoring and Diagnostic Devices Market
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Select a License Type
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients




