U.S. Cystoscopy Market Size and Forecast – 2026 to 2033
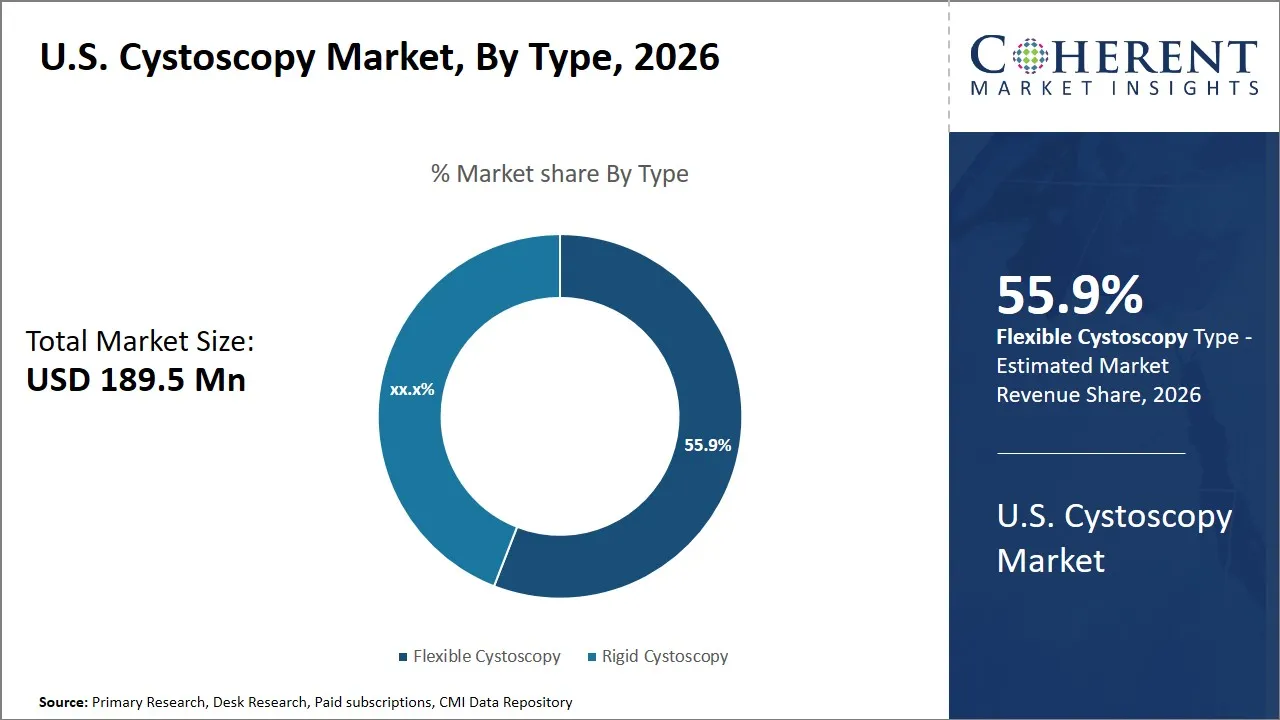
The U.S. cystoscopy market is estimated to be valued at USD 189.5 Mn in 2026 and is expected to reach USD 280.6 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033. This steady growth reflects increasing demand for minimally invasive diagnostic procedures and rising prevalence of urological disorders, driving the adoption of advanced cystoscopy technologies across healthcare facilities in the U.S.
Key Takeaways of the U.S. Cystoscopy Market
- Flexible cystoscopy segment is expected to lead the U.S. cystoscopy market, capturing 55.9% share in 2026.
- Hospitals segment is estimated to represent 35.6% of the U.S. cystoscopy market share in 2026.
Market Overview
- The U.S. cystoscopy market remains fundamentally strong because cystoscopy is still a core diagnostic and surveillance tool for bladder cancer, hematuria, and other lower urinary tract disorders, giving the procedure a stable clinical base rather than a niche role.
- Demand is being supported by the continued burden of bladder cancer in the U.S. and the need for repeated follow-up in many patients, which keeps cystoscopy volumes recurring rather than one-time.
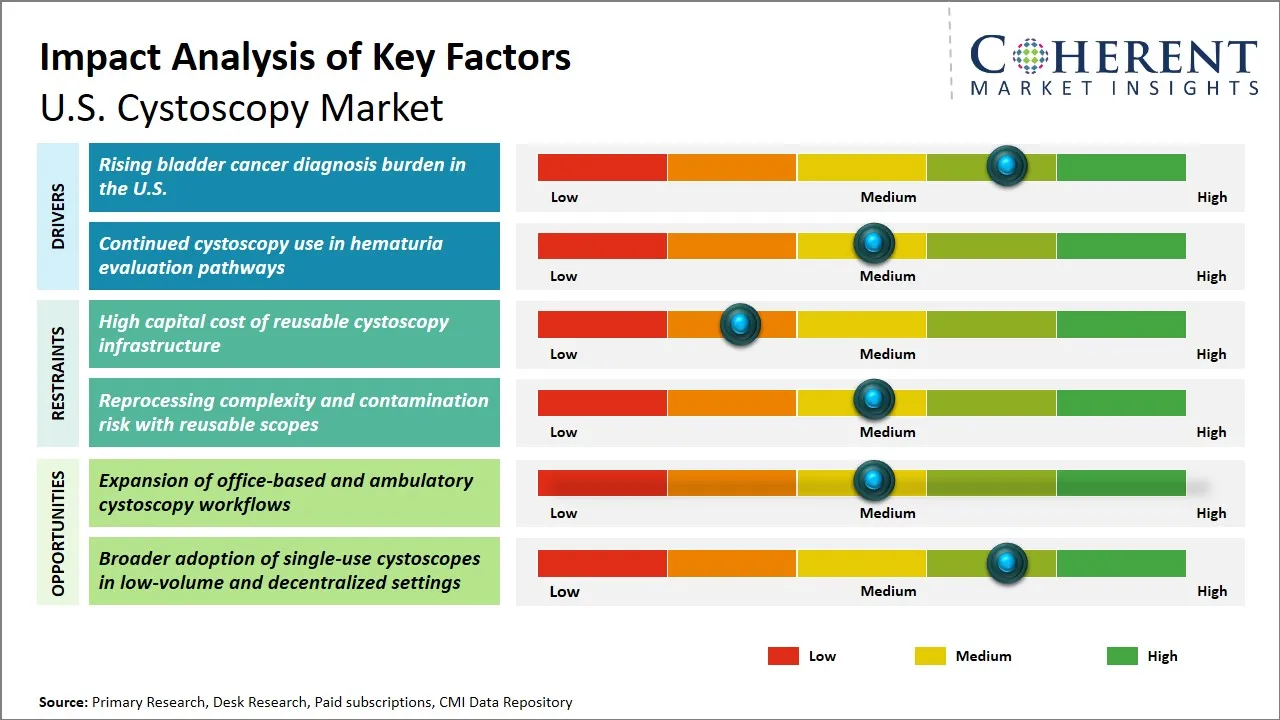
- The market is shifting from conventional reusable systems toward single-use cystoscopes, driven by infection-control concerns, reprocessing burden, and the need for faster procedure readiness in outpatient and office-based settings.
- Technology competition is intensifying as manufacturers focus on HD imaging, digital visualization, portability, and workflow efficiency, making product differentiation increasingly dependent on usability and operational value rather than only optics hardware.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Rising adoption of single-use cystoscopes |
|
|
Growing burden of bladder cancer and urological disorders |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Flexible Cystoscopy Segment Dominate the U.S. Cystoscopy Market in 2026?
The flexible cystoscopy segment is expected to hold the largest share of 55.9% in 2026, owing to its advanced technological advantages and enhanced patient-centric features. The flexible cystoscopes also provide more maneuverability compared to the rigid cystoscopes and this enables the urologists to move around the urinary tract with ease and precision. This flexibility greatly minimizes patient discomfort in the procedure that makes it a favorite procedure by patients and the health care practitioners who seek to increase their patient compliance and satisfaction.
For instance, in March 2025, Photocure announced that abstracts presented at the EAU 2025 Congress highlighted the benefits of Blue Light Cystoscopy in bladder cancer management, including its effect on recurrence risk and diagnostic decision-making. This is relevant to flexible cystoscopy because it strengthens the clinical case for enhanced-visualization cystoscopy workflows in bladder cancer surveillance and diagnosis.
Why Does the Hospitals Segment Dominate the U.S. Cystoscopy Market in 2026?
The hospitals segment is projected to capture 35.6% share in 2026, driven primarily by their expansive capabilities in delivering comprehensive urological care. Hospitals have multidisciplinary departments and sophisticated facilities, which make it possible to administer a vast scope of diagnostic and treatment cystoscopy processes. The complexity of the cases that they could provide special care on makes them the best place to carry out cystoscopy. Access to latest medical equipment and well-trained medical specialists, such as urologist and anesthesiologist, is among the major determinant factors in hospital dominance.
For instance, in June 2025, Nashville General Hospital began using Blue Light Cystoscopy with Cysview in its urology service. The hospital positioned the technology as a more advanced approach for identifying additional lesions not easily seen with white light alone, showing how public and community hospital systems are adopting advanced cystoscopy tools to improve oncology care.
High-Burden U.S. Regions for Bladder Cancer and their Implications for Cystoscopy Demand
- Northern New England- The burden of bladder cancer is significantly higher in the states of Maine, New Hampshire and Vermont, which have experienced an incidence rate nearly 20 percent higher than the U.S. as a whole in a long-term regional trend. Northern New England is a high-risk region which should be of significance in cystoscopy demand, particularly in diagnosis and surveillance.
- Appalachian region- Appalachian region is still experiencing wider limits of cancer disparity and rural Appalachian residents exhibit an elevated burden of cancer incidence and mortality than those who are not in the Appalachian region. In the case of bladder cancer, rural environments are also worse in results and access disparities, and so this area would be commercially feasible in terms of hospital-based and community urology cystoscopy care.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In September 2025, Zenflow secured U.S. FDA 510(k) clearance for the Spring Scope, a sterile single-use flexible cystoscope intended for visualization of the lower urinary tract. The approval strengthens Zenflow’s position in and reflects growing momentum behind disposable cystoscopy systems as healthcare providers prioritize infection control and operational efficiency.
- In June 2025, Ambu received U.S. FDA 510(k) clearance for its aScope 5 Cysto HD integrated with aBox 2, marking a key expansion of its single-use urology portfolio. The clearance supports broader adoption of high-definition, disposable cystoscopy solutions in the U.S., where hospitals and urology centers are increasingly prioritizing workflow efficiency, sterility, and reduced reprocessing burden.
- In June 2025, the U.S. FDA approved UroGgen’s Zusduri (mitomycin intravesical solution) for adults with recurrent low-grade intermediate-risk non-muscle invasive bladder cancer. The approval is significant for the U.S. cystoscopy market because, in the FDA-cited ENVISION trial, tumor status was evaluated every three months using cystoscopy, reinforcing the procedure’s importance in ongoing bladder cancer assessment and follow-up care.
Reusable vs Disposable Cystoscope Adoption Split in the U.S. Cystoscopy Market
|
Segment/Setting |
Estimated Adoption Split |
|
Overall U.S. Cystoscopy Market |
Single-use: ~10–20% | Reusable: ~80–90% |
|
Hospitals / High-Volume Centers |
Single-use: ~5–15% | Reusable: ~85–95% |
|
Ambulatory / Office-Based / Lower-Volume Settings |
Single-use: ~15–30% | Reusable: ~70–85% |
|
Short-Term Market Direction |
Single-use share increasing | Reusable share gradually declining but still dominant |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Top Strategies Followed by U.S. Cystoscopy Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading companies in the U.S. cystoscopy market focus on R&D to improve imaging quality, scope flexibility, handling, and patient comfort. They also use partnerships, OEM collaborations, and wider distribution expansion to strengthen market presence and reach more healthcare providers globally. |
Olympus continues to strengthen its cystoscopy portfolio through advanced endoscopy innovation and its broad hospital distribution network, helping it maintain a strong position in urology visualization systems. |
|
Mid-Level Players |
Mid-level players in the U.S. cystoscopy market usually compete on value rather than premium positioning. They focus on cost-effective systems that offer acceptable imaging quality, ease of use, and lower operating burden, making them attractive to hospitals, outpatient centers, and office-based urology practices with tighter budgets. They also rely more on partnerships and targeted commercialization than on heavy in-house R&D. |
UroViu is a good example for this profile. The company markets portable single-use cystoscopy solutions around lower capital needs, no reprocessing, workflow efficiency, and office-based use, which aligns well with price-sensitive and operationally focused buyers in the market in the U.S. |
|
Small-Scale Players |
Small-scale players in the U.S. cystoscopy market usually compete by focusing on niche innovation rather than scale. They target specific unmet needs such as single-use cystoscopy, simplified workflow, portability, or specialty imaging, and often grow through local partnerships, distributor tie-ups, or OEM collaborations instead of building large direct sales networks. |
OTU Medical fits this profile well. It focuses on single-use cystoscopes and expanded its reach through a partnership with KARL STORZ for worldwide commercialization, showing how a smaller player can use innovation plus strategic alliances to strengthen market presence. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
U.S. Cystoscopy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 189.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 280.6 Mn |
| Segments covered: |
|
||
| Companies covered: |
Olympus Corporation, KARL STORZ, Richard Wolf Medical Instruments, Stryker, Boston Scientific, Ambu, Becton Dickinson, Coloplast, UroViu, OTU Medical, Zhuhai Pusen Medical, Hunan Vathin Medical, MacroLux Medical Technology, Strauss Surgical, and Henke-Sass Wolf |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Cystoscopy Market Dynamics
To learn more about this report, Request Free Sample
U.S. Cystoscopy Market Driver - Rising Bladder Cancer Diagnosis Burden in the U.S.
The rising cases of bladder cancer in the U.S. have a major role to play in the U.S. cystoscopy market development since early diagnosis and regular screening of the bladder is important in the management of the condition. Bladder cancer is one of the most prevalent malignancies and its high recurrence rate requires repeated diagnostic testing which is mainly done through cystoscopy tests. With the rising aging population, the rise in the exposure to risk factors, which include smoking and environmental carcinogens, the number of new cases has increased as well as the number of stringent screening procedures. Moreover, the development of cystoscopy technology with better imaging options has made these diagnostic tests more precise and comfortable, which has promoted the increased usage of the cystoscopy among the healthcare providers.
For instance, in September 2025, the U.S. Food and Drug Administration approved Johnson & Johnson’s Inlexzo (gemcitabine intravesical system) for adults with BCG-unresponsive non-muscle invasive bladder cancer with carcinoma in situ, with or without papillary tumors. The approval highlights continued treatment innovation in bladder cancer and reinforces the significant ongoing disease burden in the U.S., supporting sustained need for diagnosis, monitoring, and cystoscopy-based follow-up.
U.S. Cystoscopy Market Opportunity - Expansion of Office-Based and Ambulatory Cystoscopy Workflows
The U.S. cystoscopy market has an opportunity to enlarge substantially due to the increasing growth in office-based and ambulatory cystoscopy workflow. Historically limited to hospitals, cystoscopy examinations are moving more frequently to outpatient clinics and ambulatory surgery centers (ASCs) as a result of the development of minimally invasive technology and the emergence of a more cost-oriented way of healthcare delivery. This enables the urologists carry out diagnostic and therapeutic cystoscopy procedures in convenient and friendly settings that are patient friendly and thus avoiding hospital admissions thus lowering the costs of healthcare in totality.
For instance, in November 2025, UroViu announced CE certification for its Wi-Fi-enabled cystoscope portfolio and said its platform can transform any room into a procedure room, enabling schedule expansion and workflow efficiencies. While this was not a U.S. regulatory update, it is highly relevant commercially because it reflects the same market direction favoring portable cystoscopy systems for decentralized and office-based care delivery.
Analyst Opinion (Expert Opinion)
- The U.S. cystoscopy market is still remaining strong since the clinical need will not fade away. Cystoscopy has continued to be a fundamental part of workup hematuria work up and overseeing of bladder cancer and the 2025 AUA/SUFU microhematuria update retained it as significant in risk-based appraisal. A practical change in the purchasing behavior is also helping it grow. The providers are not only considering image quality but also heed more to faster workflow, less reprocessing load and simpler operation at office based and ambulatory location. This is why newer flexible and single use platforms are receiving increased interest despite the fact that cost justification between reusable and disposable systems is a real issue.
- What is even more interesting about this market now is that all trends of clinical evidence, introduction of products and industry happenings are following one direction. The AUA 2025 in Las Vegas was still a major guideline and practice discussion point and EAU 2025 offered new information on blue light cystoscopy and management of bladder cancer. On the company front, the recent U.S. FDA clearances of the aScope5 Cysto HD by the company of Ambu demonstrate the attempts of the manufacturers to push cystoscopy into more flexible workflow-based uses rather than depending on the traditional reusable systems. Practically, the market is moving to replacement of pure equipment to a model that is based on convenience, outpatient effectiveness and procedure preparedness.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Flexible Cystoscopy
- Rigid Cystoscopy
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Cancer Centers/ Diagnostic Centers
- Ambulatory Surgical Centers
- Specialty Clinics
- Key Players Insights
- Olympus Corporation
- KARL STORZ
- Richard Wolf Medical Instruments
- Stryker
- Boston Scientific
- Ambu
- Becton Dickinson
- Coloplast
- UroViu
- OTU Medical
- Zhuhai Pusen Medical
- Hunan Vathin Medical
- MacroLux Medical Technology
- Strauss Surgical
- Henke-Sass Wolf
Sources
Primary Research Interviews
Industry Stakeholders list
- Urologists
- Urologic oncologists
- Hospital urology department heads
- Ambulatory surgery center administrators
- Endoscopy nurse managers
- Hospital procurement and supply chain managers
End-users list
- Hospitals
- Ambulatory surgery centers
- Office-based urology clinics
- Academic medical centers
- Integrated delivery networks
- Veterans’ Health Administration facilities
Government and International Databases
- SEER Program
- U.S. Cancer Statistics Data Visualizations Tool
- FDA 510(k) Premarket Notification Database
- HCUP Nationwide Databases
- Global Cancer Observatory
- ClinicalTrials.gov
Trade Publications
- Urology Times
- Renal & Urology News
- MedTech Dive
- MD+DI
- Medical Design & Outsourcing
- Becker’s ASC Review
Academic Journals
- The Journal of Urology
- Urology
- BJU International
- Urologic Oncology: Seminars and Original Investigations
- World Journal of Urology
- Bladder Cancer
Reputable Newspapers
- Associated Press
- The Wall Street Journal
- Financial Times
- The Washington Post
- The New York Times
Industry Associations
- American Urological Association
- Society of Urologic Oncology
- Society of Urodynamics, Female Pelvic Medicine & Urogenital Reconstruction
- Endourological Society
- American Association of Clinical Urology
- Society of Urologic Nurses and Associates
Public Domain Resources
- NCBI Bookshelf
- MedlinePlus
- Open Payments
- NIH RePORTER
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients