The global adult vaccines market is estimated to be valued at USD 32.80 Bn in 2026 and is expected to reach USD 51.60 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.6% from 2026 to 2033. This growth can be attributed to the rise in awareness regarding adult immunization, increasing incidence rate of chronic diseases, and the increase in government initiatives to encourage adult vaccination programs worldwide. Furthermore, developments in vaccine technologies are also likely to propel the market growth in the future.
|
Current Events |
Description and its Impact |
|
Strengthening Regulatory Frameworks for Adult Immunization |
|
|
Expansion of Adult Vaccination Programs and New Vaccine Approvals |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The influenza segment is anticipated to hold a market share of 38.0% in 2026, primarily due to the recurring impact of seasonal flu outbreaks. Influenza is a very contagious respiratory infection that results in high morbidity and mortality rates. As a result, the impact of this disease is felt throughout the year. Moreover, the fact that the virus is changing every year, requiring the formulation of the vaccine to change as well, ensures the steady market share of this segment in the adult vaccines market.
The influenza vaccine is further expected to gain from the increasing awareness of the importance of the flu vaccine, which is being promoted by various campaigns. In addition, the workplace wellness programs are also playing their role in promoting the use of the flu vaccine. As a result, the segment for the influenza vaccine is expected to hold a dominant position in the market for adult vaccines.
For instance, in September 2024, Centers for Disease Control and Prevention (CDC) launched its influenza vaccination campaign for the 2024-2025 season, emphasizing the need for annual vaccination in all adults, especially in high-risk groups. The campaign aids in the allocation of 148 million doses of influenza vaccines in the U.S., based on surveillance results for strain selection to increase the immunogenicity of the quadrivalent influenza vaccine.
The inactivated and subunit vaccines segment is projected to hold a market share of 55.0% in the global adult vaccines market, attributed to well-proven safety and efficacy profiles of the vaccine types. Inactivated and subunit vaccines are effective in inducing the immune response without causing the disease in the body, making them the most suitable option for adults suffering from weak immune conditions or chronic diseases.
In addition, the well-proven safety and efficacy profiles of the vaccine types have resulted in a high level of validation, making them the most accepted option in the development of vaccines against diseases such as influenza, hepatitis, and pneumococcal infections.
Furthermore, the vaccine types also provide a high level of tolerability, with mild side effects, making them the most suitable option for the adult population, including elderly patients suffering from weak immune conditions or chronic diseases.
The preventive segment is projected to hold a market share of 92.0% in 2026, attributed to the global focus on preventive healthcare and disease prevention. Vaccination is one of the most critical tools in public health practice for disease prevention and control by reducing the incidence and severity of infectious diseases. The importance of vaccination has increased in recent times because of the increasing aged population and chronic conditions, which enhance the severity of infectious diseases. Routine vaccination in adults for infectious diseases such as influenza, pneumonias, and hepatitis is one of the critical tools in healthcare practice.
The increasing vaccine-preventable diseases and strong support from governments and healthcare organizations have also fueled the growth of preventive vaccines. The increasing vaccine-preventable diseases in recent times have positively impacted preventive vaccines. The strong support from governments and healthcare organizations has also fueled the growth of preventive vaccines. The COVID-19 pandemic has positively impacted preventive vaccines in recent times by increasing vaccine acceptance and proving that vaccination is one of the critical tools in disease prevention.
|
Regulatory Authority / Framework |
Regulatory Focus |
Impact on Adult Vaccines Market |
|
U.S. Food and Drug Administration (FDA) |
Biologics License Application (BLA), clinical trials, safety & efficacy standards for vaccines |
Ensures high safety and accelerates approval of innovative vaccines (e.g., mRNA, RSV) |
|
European Medicines Agency (EMA) |
Centralized approval process, pharmacovigilance, and quality standards |
Promotes harmonized approvals across Europe and strengthens vaccine safety monitoring |
|
World Health Organization (WHO) |
Prequalification (PQ) program and global immunization guidelines |
Facilitates global vaccine access, especially in low- and middle-income countries |
|
Centers for Disease Control and Prevention (CDC) / ACIP |
Adult immunization schedules and vaccination recommendations |
Drives vaccine adoption through evidence-based guidelines and public health policies |
|
European Commission |
Vaccine procurement, public health policies, and regulatory directives |
Supports large-scale immunization programs and cross-country vaccine distribution |
|
National Medical Products Administration (NMPA) |
Vaccine approval, clinical evaluation, and post-market surveillance |
Expands market access in China and strengthens regulatory transparency |
|
Central Drugs Standard Control Organization (CDSCO) |
Vaccine approval and regulation under New Drugs & Clinical Trials Rules |
Encourages vaccine availability and growth in emerging markets |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
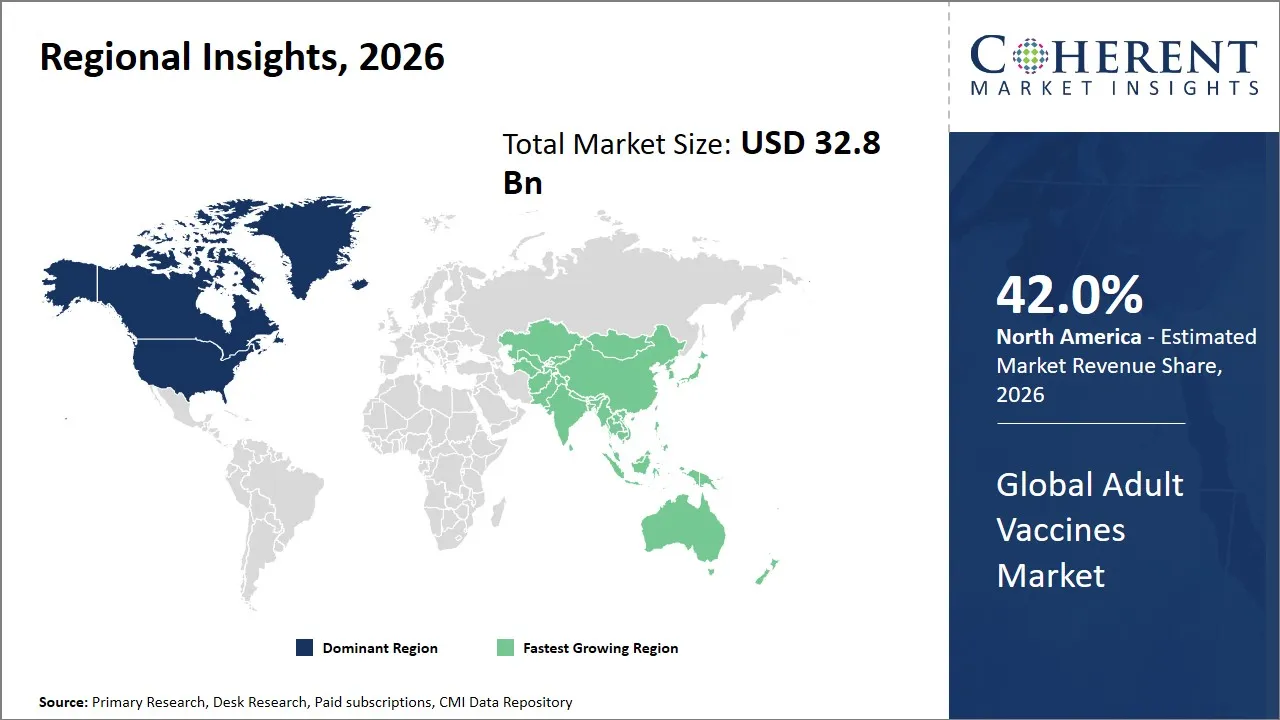
The North America region is projected to lead the market with a 42.0% share in 2026, attributed to a well-established healthcare infrastructure, high research standards, and government initiatives for adult immunization programs. Organizations like the Centers for Disease Control and Prevention, a U.S. government agency, and similar organizations actively support adult vaccination awareness and policy initiatives, resulting in high vaccination rates.
Furthermore, the presence of pharmaceutical and biotechnological companies like Pfizer, Merck & Co., and Moderna facilitates the rapid development and distribution of adult vaccines, including influenza, pneumococcal, HPV, and COVID-19 vaccines. Established healthcare infrastructure in terms of reimbursement policies and high supply chain standards is a significant contributor to market dominance. Trade agreements and high investment in vaccine research and development by both public and private entities are factors that contribute to North America's dominance in the market.
For instance, on March 27, 2026, a report published by the University of Minnesota emphasized the fact that after the pandemic, the Americas saw a surge in pertussis infections, which reflected the deficiency in routine immunization. The rise in vaccine-preventable diseases emphasizes the need for adult booster vaccinations, thereby increasing the demand for adult vaccines.
(Source: University of Minnesota)
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 25.0% in 2026, attributed to the expansion of healthcare infrastructure, increasing awareness of adult vaccines, and the government's increasing focus on preventive healthcare. Countries such as China, India, and South Korea are increasing their immunization policies to include adult vaccines in addition to the existing childhood vaccines. This is due to the increasing elderly population and the prevalence of chronic diseases in the region.
Improvements in logistics, increasing disposable incomes, and the expansion of pharmaceutical manufacturing bases also contribute to the growth of the adult vaccines market in the region. Increasing participation by multinational companies in the vaccine market, such as GlaxoSmithKline (GSK) and Sanofi, along with local players such as the Serum Institute of India, also contributes to the growth of the adult vaccines market by increasing competition in the region.
The adult vaccines market in the U.S. is characterized by strong government support through several schemes, including the Adult Immunization Action Plan and strong funding for research. Pfizer and Moderna are examples of companies that have changed the adult vaccines market through innovative mRNA vaccines, such as those used in dealing with the COVID-19 pandemic. There is a strong network of healthcare facilities and insurance schemes, resulting in high vaccination rates. Also, the country has a strong portfolio in conducting research, including trials, and in promoting public health through preventive care.
For instance, in August 2024, recommendations published by U.S. Centers For Disease Control and Prevention in Advisory Committee on Immunization Practices on the administration of the RSV vaccine in adults aged ≥60 years have emphasized the importance of the risk-based approach in the administration of the RSV vaccine in the adult population. In particular, the recommendations have emphasized the importance of vaccinating the high-risk populations who are suffering from chronic conditions and immunocompromising conditions.
(Source: U.S. Centers For Disease Control and Prevention)
Germany holds the top position in the adoption rate of adult vaccination in the European region due to the well-established healthcare system in the country and the high rate of public trust in the procedure. The German Standing Committee on Vaccination, also known as STIKO, has issued comprehensive recommendations on adult vaccination. Some of the prominent companies in this sector are BioNTech, which developed a COVID-19 vaccine in collaboration with Pfizer, and Bavarian Nordic, which has driven the development in vaccine technology. The government's reimbursement policies provide incentives for adult vaccination, and the well-established healthcare system in the country ensures accessibility to adult vaccines thus, driving the market growth.
The adult vaccines market in China has seen a tremendous growth rate, where the government has framed several policies that encourage adult vaccination in the country. This is mainly due to the rise in the elderly population and non-communicable diseases. The Chinese Center for Disease Control and Prevention has been actively promoting adult vaccination in China, aiming to increase vaccination rates in the country.
The local vaccine manufacturers in China, such as Sinovac and CanSino Biologics, hold a major share of the adult vaccines market in China, where multinationals are entering the market through joint ventures in China. There has been a significant rise in healthcare infrastructure and technology, resulting in a higher availability of vaccines in China.
The strategies adopted by the Chinese government in terms of easing the vaccine approval process in China and enhancing research and development in China have significantly contributed to the growth of the adult vaccines market in China.
The adult vaccines market in India is gaining momentum due to an increase in government initiatives aimed at adult immunization in addition to childhood immunization. Various organizations, like the Ministry of Health and Family Welfare, are working to spread more awareness regarding vaccines in the adult population to prevent preventable diseases.
The Serum Institute of India, which is the largest vaccine manufacturer in the world in terms of volume, plays an important role in providing vaccines at affordable prices both in the country and worldwide. An increase in healthcare spending, urbanization, and better cold chain infrastructure is also adding to the momentum driving the market growth in the country.
|
Region |
Reimbursement Scenario |
Key Programs / Systems |
Impact on Adult Vaccines Market |
|
North America |
Strong reimbursement coverage through public and private insurance for key adult vaccines (influenza, pneumococcal, COVID-19) |
Centers for Medicare & Medicaid Services (CMS), Private Insurers |
High vaccination rates and strong market dominance |
|
Europe |
Strong reimbursement: Universal healthcare systems provide broad reimbursement for recommended adult vaccines |
National Health Service (NHS), EU National Health Programs |
Ensures wide accessibility and stable market growth |
|
Asia Pacific |
Mixed reimbursement; strong in Japan and Australia, limited but improving in China and India |
National Immunization Programs, Government Subsidies |
Moderate adoption with high growth potential |
|
Latin America |
Partial reimbursement through public health programs; reliance on government-funded vaccination drives |
National Immunization Programs |
Moderate uptake with cost constraints |
|
Middle East & Africa |
Limited reimbursement; largely dependent on government initiatives and international support programs |
Gavi, the Vaccine Alliance, Public Health Systems |
Lower adoption but improving access in select countries |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report

To learn more about this report, Request Free Sample
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders emphasize research and development investments in next-generation vaccines that are more potent, safe, and offer a wider range of virus strains. They emphasize innovation in vaccine platforms, regulations, and large-scale commercialization by leveraging global distribution channels and collaborating strategically with governments and healthcare organizations. |
In May 2023, GlaxoSmithKline plc announced obtained U.S. FDA approval for its RSV vaccine (arexvy), which is the first RSV vaccine for adults and further establishes GlaxoSmithKline as a leader in adult respiratory vaccines and expands the Company’s portfolio of innovative vaccines. |
|
Mid-Level Players |
Mid-level market players emphasize cost-effective vaccine solutions by optimizing production processes and developing strategic partnerships. They emphasize regional market development, flexible production, and partnerships that improve supply chain efficiency and cater to regional, price-sensitive, and emerging market demands. |
In August 2023, Biological E Limited expanded its vaccine portfolio by forging strategic partnerships that would enhance the availability of vaccines at an affordable price in emerging markets and increase the availability of adult vaccines. |
|
Small-Scale Players |
Small-scale market players concentrate on the development of innovative specialized vaccines, targeting specific diseases, where advanced technology and associations with research institutions are used to develop and enter the markets. |
In June 2023, Valneva SE obtained U.S. FDA approval for its vaccine for adults to protect against chikungunya infection (IXCHIQ). |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 32.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.69% | 2033 Value Projection: | USD 51.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc, GlaxoSmithKline plc, Merck & Co Inc, Sanofi, Johnson and Johnson, Moderna, AstraZeneca plc, CSL Limited, Serum Institute of India, and Bharat Biotech |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
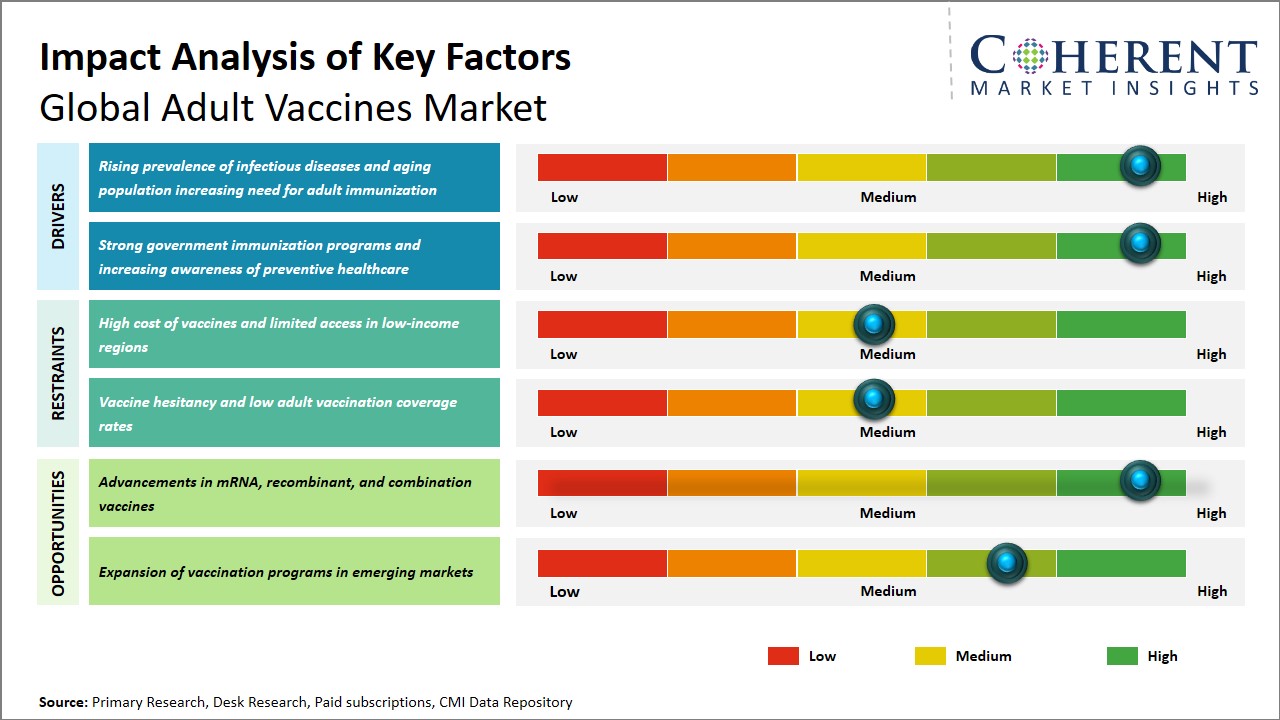
The rising rates of infectious diseases across the world have further increased the importance of adult vaccination programs. Adults are prone to various infectious diseases. On the other hand, the demographic shift in the world population towards an increasing number of elderly citizens has also increased the need for booster shots and adult vaccines. This segment of the population is usually prone to various infectious diseases like flu, pneumonia, and herpes zoster. Therefore, adult vaccines are extremely important in reducing morbidity and mortality rates.
Additionally, the rising rates of various infectious diseases that were previously eradicated due to weakening immunity and genetic changes in pathogens have also increased the importance of adult vaccines. Healthcare facilities across the world are increasingly recognizing the importance of adult vaccines. This is because adult vaccines can help reduce morbidity and mortality rates among this segment of the population.
For instance, according to the World Health Organization (WHO), immunization results in the prevention of approximately 4.4 million deaths globally. Furthermore, statistics provided by UNICEF and WHO (WUENIC estimates) reveal that an estimated 14.3 million children remained unimmunized in 2024, emphasizing the gap in immunization and consequently the spread of infectious diseases.
(Source: UNICEF, World Health Organization (WHO))
The global adult vaccines market is expected to gain significantly from recent developments in vaccine technology, especially in relation to mRNA, recombinant, and combination vaccines. For example, mRNA vaccines, which have entered the arena in recent times with much popularity owing to COVID-19, have one unique advantage in that they can be adapted quickly to counter new infectious diseases. This has opened a vast window of opportunity in targeting new diseases in adults, such as influenza, respiratory syncytial virus, and cytomegalovirus, thus expanding the vaccine portfolio for adults.
In addition, recombinant vaccine technology, which employs genetically engineered proteins, ensures a greater degree of safety and efficiency in vaccine development by targeting one particular antigen, thus eliminating side effects. This is especially true in adults, who might be suffering from various co-morbid conditions, such as compromised immunity.
Furthermore, combination vaccines, which ensure immunity against various infectious agents in one dose, ensure greater compliance in patients, thus reducing healthcare costs by limiting the number of clinic visits. These combination vaccines ensure greater efficiency in vaccination, thus ensuring greater vaccination rates, especially in adults.
For instance, in June 2024, Moderna, Inc. reported encouraging Phase 3 findings for its mRNA-1083 combination vaccine, designed to combat both influenza and COVID-19. The results showed that this new approach generated stronger immune responses than current vaccines given separately. This development underscores the increasing importance of mRNA-based combination vaccines in boosting immune system activity and streamlining vaccination plans for adults.
(Source: Moderna, Inc.)
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients