Alagille Syndrome Treatment Market is estimated to be valued at USD 108.1 Mn in 2026 and is expected to reach USD 171.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 6.8% from 2026 to 2033.
Analysts’ Views on the Global Alagille Syndrome Treatment Market:
Alagille syndrome is one of the rarest forms of liver disease, and most patients go untreated due to lack of effective treatment. The lack of effective treatment leaves patients with limited options for managing their symptoms and improving their quality of life. Patients with Alagille syndrome often experience a range of symptoms including liver damage, heart problems, and developmental issues. The absence of effective treatment not only hampers their ability to alleviate these symptoms but also makes it challenging for healthcare professionals to provide comprehensive care and support for these patients. Without effective treatment, patients with Alagille syndrome may face a lifetime of complications and difficulties. The absence of options to manage their symptoms not only impacts their physical health but also takes a toll on their emotional well-being, highlighting the urgent need for further research and development in this field.
Figure 1. Global Alagille Syndrome Treatment Market Share (%), By Drug, 2026
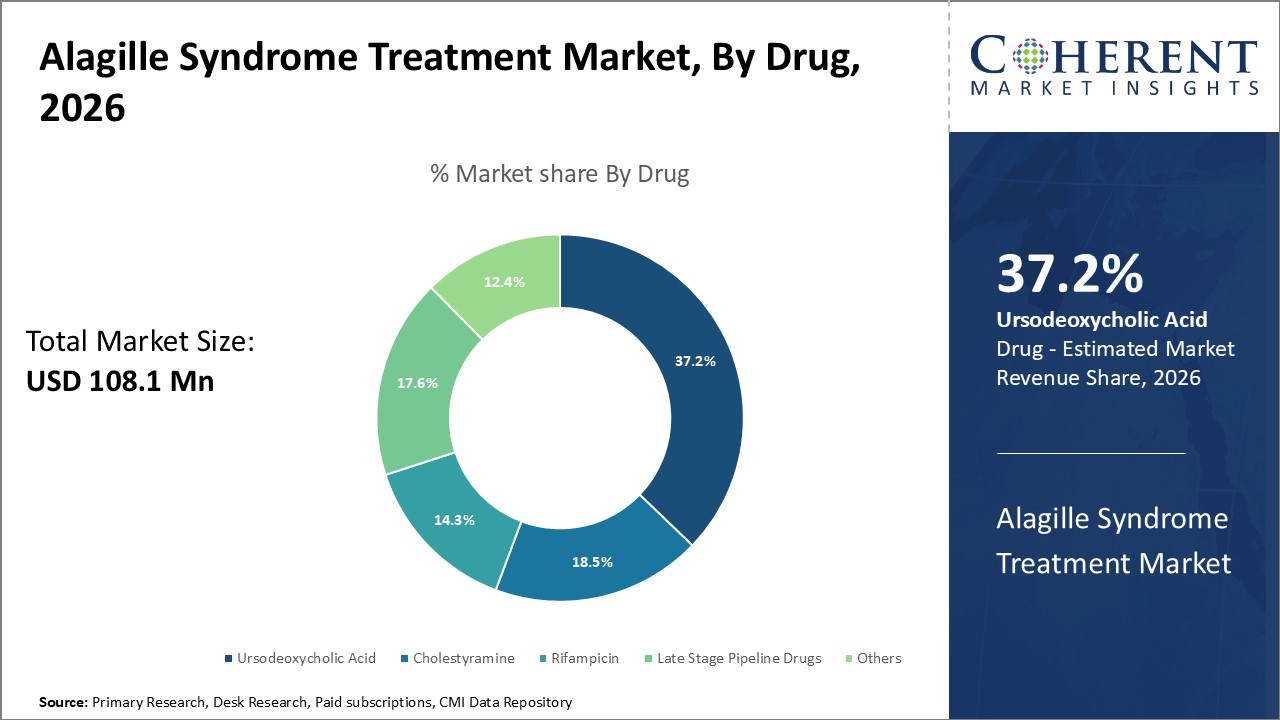
To learn more about this report, Request Free Sample
Global Alagille Syndrome Treatment Market - Drivers
Figure 2. Global Alagille Syndrome Treatment Market Share (%), By Region, 2026
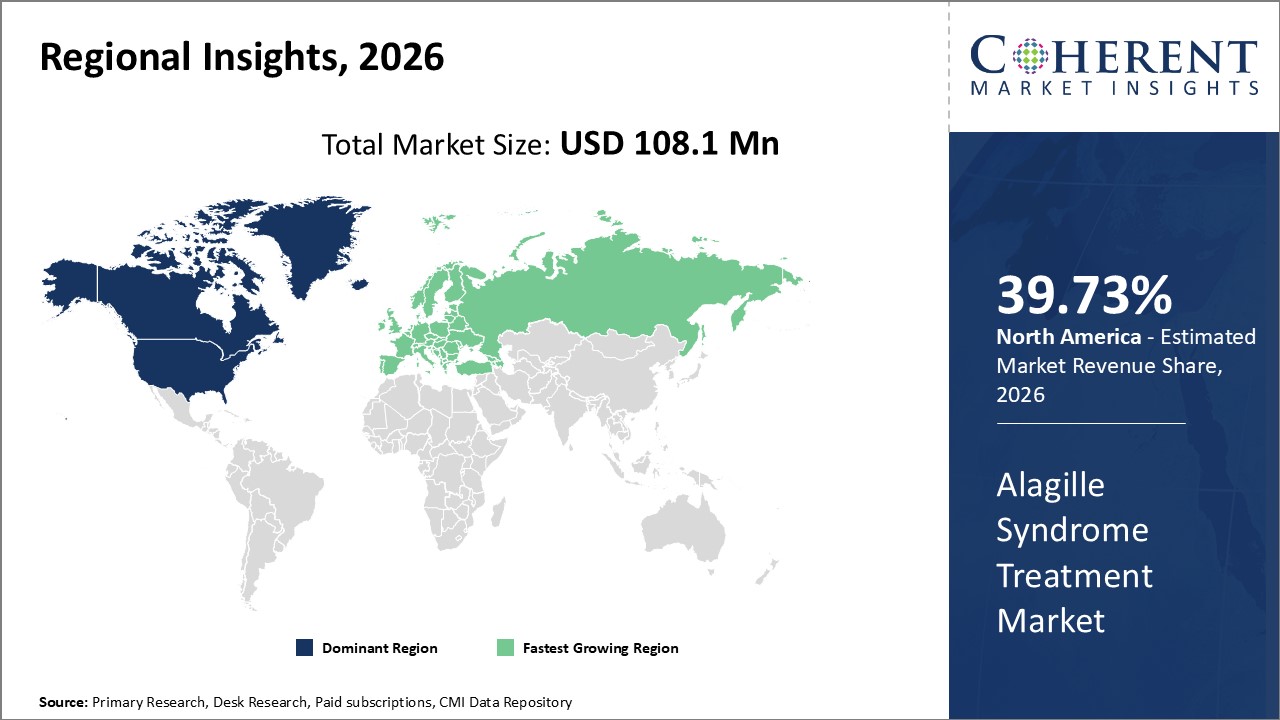
To learn more about this report, Request Free Sample
Global Alagille Syndrome Treatment Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global alagille syndrome treatment market over the forecast period. North America is estimated to hold 39.73% of the market share in 2026. The global alagille syndrome treatment market is expected to witness significant growth in the coming years, driven by the high prevalence of Alagille syndrome. The exact prevalence rate of Alagille syndrome in North America is not known, but researchers are working to determine it. Alagille syndrome is considered a rare genetic disorder that affects multiple organ systems, making it challenging to accurately estimate its prevalence. However, ongoing research efforts are focused on gathering data and conducting comprehensive studies to determine the exact prevalence rate of Alagille syndrome in North America. These efforts involve collaboration between medical professionals, researchers, and patient advocacy groups to ensure a thorough understanding of the disorder's occurrence. By obtaining accurate prevalence data, healthcare providers can better allocate resources and develop targeted interventions for individuals with Alagille syndrome in North America.
Global Alagille Syndrome Treatment Market Segmentation:
The global alagille syndrome treatment market report is segmented into drug, distribution channel, and region.
Alagille Syndrome Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 108.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.8% | 2033 Value Projection: | USD 171.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd., AbbVie, Inc., Glenmark Pharmaceuticals Limited, Par Pharmaceuticals, Inc., Mylan Pharmaceuticals, Inc., Sanofi S.A., Mylan N.V., Novartis International AG, Akorn, Inc., Albireo Pharma, Inc., Mirum Pharmaceuticals, Ipsen Pharma, and CANbridge Pharmaceuticals, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Alagille Syndrome Treatment Market - Cross Sectional Analysis:
Key players are making new drugs for the treatment of Alagille syndrome with advanced technology, which is expected to boost the demand for the alagille syndrome treatment market in the North America region. Alagille syndrome is a rare genetic disorder that affects the liver and other organs, causing various complications. The development of new drugs using advanced technology signifies a significant breakthrough in the treatment of this complex condition, offering hope for improved outcomes and a better quality of life for patients in North America. These new drugs have the potential to target specific genetic mutations associated with Alagille syndrome, providing more personalized and effective treatment options. Additionally, the advancements in technology have also led to improved diagnostic tools, allowing for earlier detection and intervention of Alagille syndrome in North America.
Global Alagille Syndrome Treatment Market: Key Developments
Global Alagille Syndrome Treatment Market: Key Trends
Global Alagille Syndrome Treatment Market: Restraint
Global Alagille Syndrome Treatment Market - Key Players
The major players operating in the global alagille syndrome treatment market include Teva Pharmaceutical Industries Ltd., AbbVie, Inc., Glenmark Pharmaceuticals Limited, Par Pharmaceuticals, Inc., Mylan Pharmaceuticals, Inc., Sanofi S.A., Mylan N.V., Novartis International AG, Akorn, Inc., Albireo Pharma, Inc., Mirum Pharmaceuticals, Ipsen Pharma, and CANbridge Pharmaceuticals, Inc.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients