Anti-Neprilysin Market is estimated to be valued at USD 1,448.2 Mn in 2026 and is expected to reach USD 2,010.8 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.8% from 2026 to 2033.
Analysts’ Views on Global Anti-Neprilysin Market :
The expansion of new manufacturing and production facilities is expected to boost growth of the global anti-neprilysin market over the forecast period. For instance, on June 2, 2022, Cipla Inc., a India-based biopharmaceutical and medical company, announced the commercial operation of additional capacity of captive renewable energy power plants in Maharashtra and Karnataka, India. The project will support the company’s green energy requirements for its manufacturing units at Kurkumbh and Patalganga (villages in India) and R&D center in Maharashtra, India, replacing around 70% of the total consumption for these units with green energy.
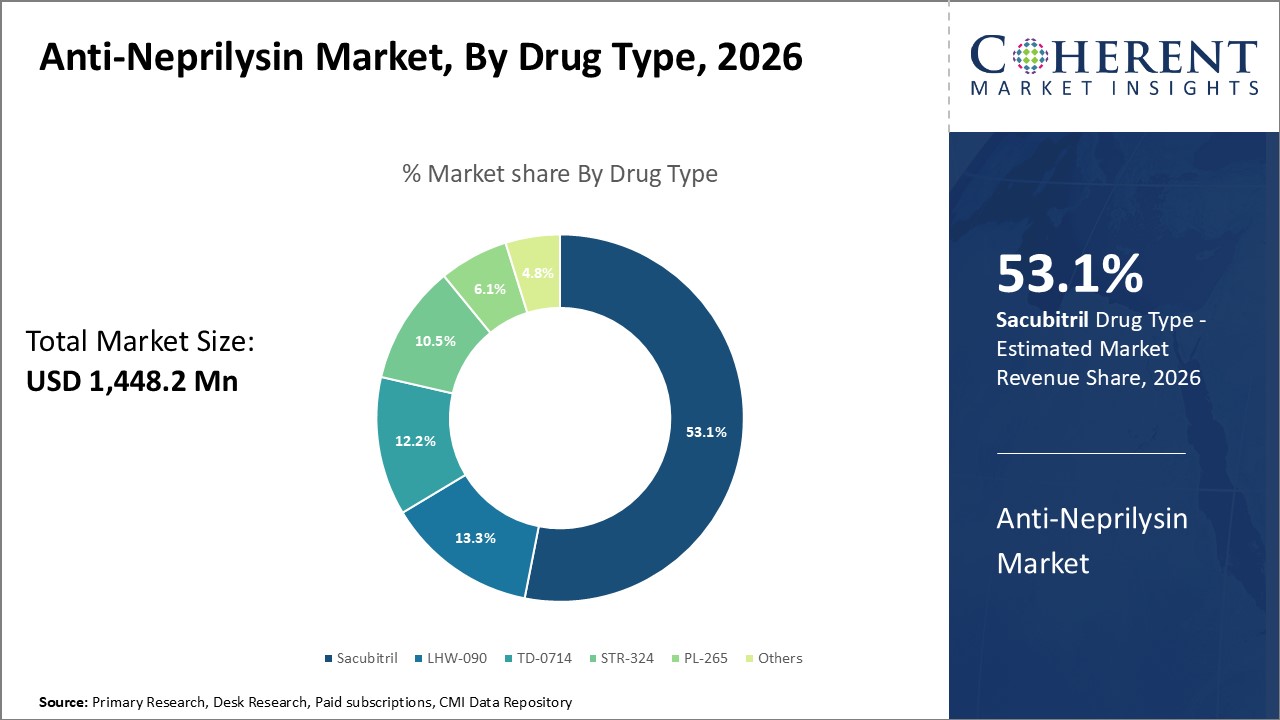
Figure 1. Global Anti-Neprilysin Market Share (%),By Drug Type, 2026
To learn more about this report, Request Free Sample
Global Anti-Neprilysin Market – Drivers
Increasing Research and Development Activities Regarding Anti-Neprilysin drugs
Growing research and development activities around the world is a major factor that boosts demand for anti-neprilysin drugs. For instance, PubMed, a free search engine for biological databases, published a research studies in September 2020, about a randomized, double-blind, placebo-controlled, single ascending dose (50–600 mg TD-0714) and multiple ascending dose (10–200 mg TD-0714 q.d. for 14 days) trial of TD-0714, an orally active, potent, and selective inhibitor of human neprilysin (NEP) in development for the treatment of chronic heart failure. The study was conducted on healthy volunteers. It was concluded that TD-0714 was generally well tolerated, and no serious adverse events or clinically significant effects on vital signs or electrocardiogram parameters were observed. The drug exhibited dose-proportional pharmacokinetics (PKs) with high oral bioavailability, minimal accumulation after once-daily dosing, and negligible renal elimination.
Introduction of New Anti-Neprilysin Drugs Products
Increasing product launches by key market players can drive growth of the global anti-neprilysin market. For instance, on January 21, 2023, MSN Laboratories, a a India-based research-based, integrated pharmaceutical company, announced the relaunch of Sacubitril/Valsartan tablets, 50 mg, 100 mg, and 200 mg, for heart failure with reduced ejection fraction (HFrEF) under the brand name Sacutan, at lower prices.
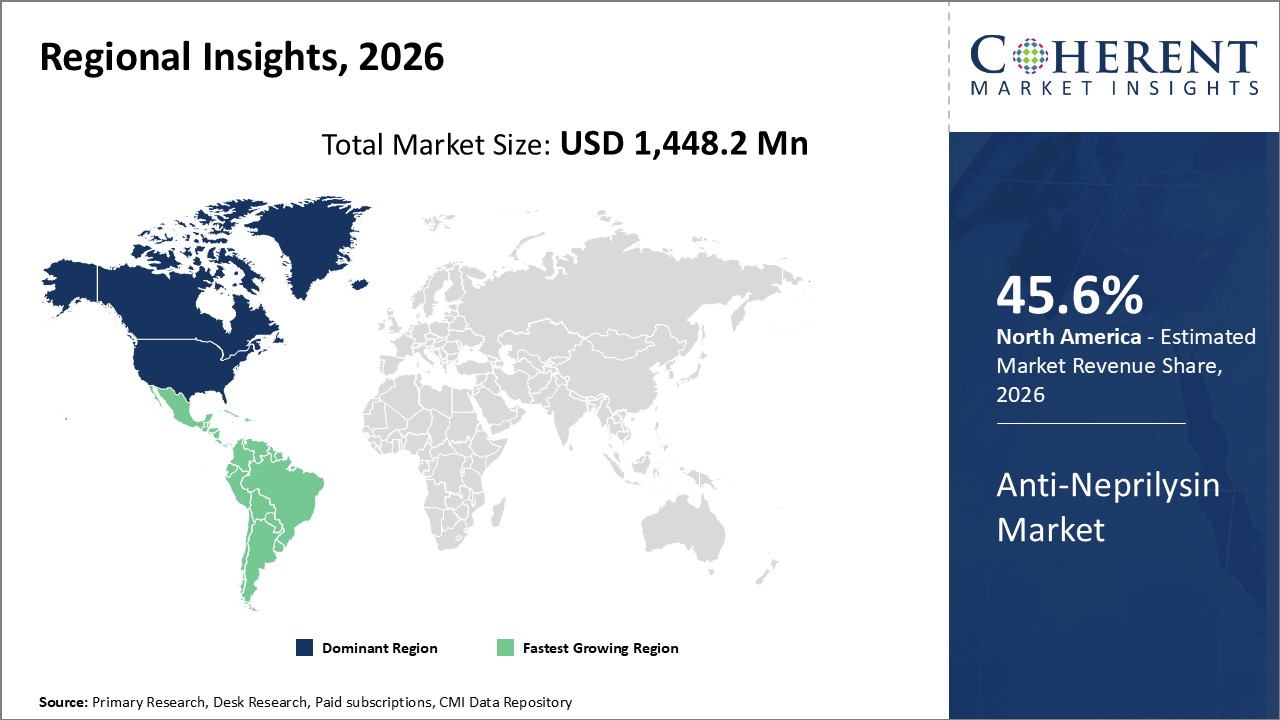
Figure 2. Global Anti-Neprilysin Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Anti-Neprilysin Market - Regional Analysis
Among region, North America is estimated to hold a dominant position in the global anti-neprilysin market over the forecast perioddue to increasing number of R&D activities in the region. For instance, according to a report published in June 2020, by American Journal of Managed Care, an independent, peer-reviewed publication dedicated to disseminating clinical information to managed care physicians, clinical decision makers, and other healthcare professionals, inflammation and fibrosis seen in heart failure with reduced ejection fraction (HFrEF) appeared subdued following treatment with anti-neprilysin agents.
Global Anti-Neprilysin Market Segmentation:
The global anti-neprilysin market report is segmented into product drug type, indication, distribution channel, and region.
By Drug Type, the market is segmented into sacubitril, TD-0714, STR-324, PL-265, and LHW-090. Out of which, the sacubitril segment is expected to hold a dominant position in the global anti-neprilysin market during the forecast period due to increasing product launches related to sacubitril
By Indication, the market is segmented into acute heart failure, cancer pain, hypertension, Alzheimer’s disease, and others. Out of which, acute heart failure segment is expected to hold a dominant position in the global anti-neprilysin market during the forecast period due to growing prevalence of the disease.
By Distribution Channel , the market is segmented into hospitals pharmacies, retail pharmacies, and online pharmacies. Out of which, the hospital pharmacies segment is expected to dominate the market over the forecast period due to\ increased hospitalizations across the world due to various diseases and disorders.
Among all the segments, the drug type segment has the highest potential due to the increasing product approvals around the world over the forecast period. For instance, on March 31, 2023, Novartis AG, a Switzerland-based biopharmaceutical company, announced that the European Medicines Agency’s (EMA), the central drug regulatory authority of the European Union (EU), Committee for Medicinal Products for Human Use (CHMP) hadadopted a positive opinion recommending approval of Entresto (sacubitril/valsartan) for a new indication to treat symptomatic chronic heart failure with left ventricular systolic dysfunction in pediatric patients aged from 1 to <18 years.
Anti-Neprilysin Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,448.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 2,010.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BIOPROJET, Novartis AG, Pharmaleads SA, Theravance Biopharm, Cipla Inc, Oceanic Pharmachem Pvt. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Anti-Neprilysin Market- Cross Sectional Analysis:
Among drug type segment, sacubitril is expected to held a dominant position in Europe region. On July 7, 2023, Novartis AG, a Switzerland-based biopharmaceutical company, announced that the U.S. District Court for the District of Delaware had issued a negative decision regarding the validity of a patent covering Entresto and combinations of sacubitril and valsartan, which expires in 2026 with its pediatric exclusivity. Novartis AG subsequently appealed to the U.S. Court of Appeals for the Federal Circuit (CAFC) to reverse the District Court’s decision.
Global Anti-Neprilysin Market : Key Developments
In December 2020, The U.S. Food and Drug Administration, the central drug regulatory authority in the U.S., announced that an expanded indication was warranted for sacubitril/valsartan that allow it as a treatment for certain patients with HF with preserved ejection fraction
In March 2021, Stragen Pharma SA, a Switzerland-based biopharmaceutical company, started enrollment of healthy volunteers in a phase II trial of STR-324 for postoperative pain. STR-324 is an anti-nephrilysin investigational new drug (IND) candidate. .
On January 18, 2023, Cipla Inc., a India-based biopharmaceutical and medical company, announced the launch of Cippoint, a point-of-care testing device. The device offers a wide range of testing parameters including cardiac markers, diabetes, infectious diseases, fertility, thyroid function, inflammation, metabolic markers, and coagulation markers. The device had been approved in August 2020, by the European In-Vitro Diagnostic Device (IVD) Directive, thus ensuring reliable testing solutions. The IVD sets out the essential safety, health, design, and manufacturing requirements that in vitro diagnostic medical devices and their accessories must meet. This ensures universally high safety standards, providing public confidence in the system. It enables the products to be used in any European Union country.
Global Anti-Neprilysin Market: Key Trends
Key market players are adopting organic strategies like the expansion of manufacturing facilities that is expected to drive the global anti-neprilysin market growth. On May 9, 2023, Sandoz, a global leader in off-patent (generic and biosimilar) medicines, announced a multi-year partnership with Just Evotec Biologics, a U.S.-based subsidiary of Evotec SE. The agreement includes the development and manufacturing of multiple biosimilar medicines with an option for expansion, and is the most recent step towards strengthening the Sandoz foundation as a stand-alone off-patent medicine company.
In October 2020, according to an study published in PubMed, a free search engine for biological databases and medical reports, that evaluated the potency obtained after integrating a neprilysin inhibitor into a comprehensive multi-drug regimen, Sacubitril/valsartan is an efficacious, safe, and cost-effective therapy that improves quality of life and longevity in patients with chronic HFrEF, as well as reduces hospital admission. An in-hospital initiation strategy offers a potentially new avenue to improve the clinical uptake of sacubitril or valsartan.
Global Anti-Neprilysin Market: Restraint
Side-effects of Anti-Neprilysin Drugs
The side effects of anti-neprilysin drugs can restrain growth of the global anti-neprilysin market. Sacubitril/valsartan was associated with a higher incidence of hypotension, symptomatic hypotension and angioedema .
This restrain can be overcome by the taking the right dose of the drug, as recommended by the physician.
Product Recalls
Manufacturing, production, and design flaws in anti-nepriysin drugs can restrain the growth of the market. In August 2020, the U.S. Food and Drug Administration (FDA) announced the product recall of valsartan tablets manufactured by Hetero Labs Ltd., aIndia-based biopharmaceutical company, for containing a probable cancer-causing chemical.
This can be overcome by strict compliance with regulatory guidelines and the adoption of good manufacturing practices.
Global Anti-Neprilysin Market - Key Players
Major players operating in the global anti-neprilysin market include BIOPROJET, Novartis AG, Pharmaleads SA, Theravance Biopharm, Cipla Inc, and Oceanic Pharmachem Pvt. Ltd.
*Definition: Neprilysin inhibitors are a new class of drugs that are used to treat high blood pressure and heart failure. These work by blocking the action of neprilysin, and thus, preventing the breakdown of natriuretic peptides. Neprilysin enzyme, also called neutral endopeptidase, plays a role in the degradation of natriuretic peptides and other vasoactive peptides including bradykinin. Natriuretic peptides remove sodium from the blood and excrete it in the urine. In the absence of natriuretic peptides, sodium levels increases in the blood that leads toincreased blood pressure.Bradykinin is a vasodilator that relaxes and widens the walls of blood vessels. This facilitates the free flow of blood in the vessels. In the absence of bradykinin, the blood vessels may not relax and may cause an increase in blood pressure. Neprilysin inhibitor increases the availability of natriuretic peptides, helps bradykinin to achieve vasodilation and natriuresis (excretion of sodium), and decreases blood pressure.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients