Alzheimer's Disease Diagnostics and Therapeutics Market is estimated to be valued at USD 7,704.5 Mn in 2026 and is expected to reach USD 11,282.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.6% from 2026 to 2033. It is a form of dementia that progresses over time, causing a person's capacity for independent functioning to be disrupted by a steady decline in thinking, behavioural, and social abilities. Alzheimer's disease has no known cure, but there are treatments that may slow down the disease's progression as well as medication and non-medication options that could help manage symptoms.
Figure 1. Global Alzheimer’s disease Diagnostics and Therapeutics Market Share (%), By Product, 2026
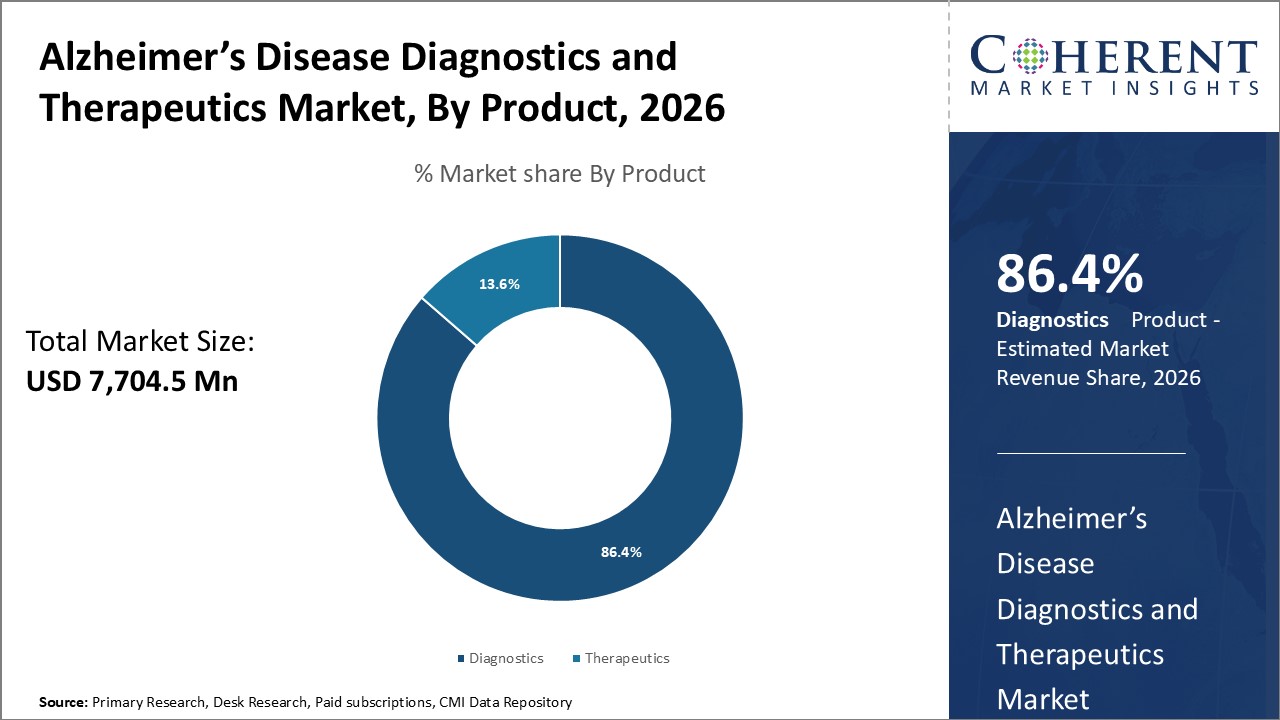
To learn more about this report, Request Free Sample
Global Alzheimer’s disease Diagnostics and Therapeutics Market- Drivers
Increase in the prevalence of Alzheimer’s disease in the old age population is expected to drive the market for the forecast period.
According to Alzheimer's Association, April 2022, an estimated 6.5 million people age 65 and older in 2021 were living with Alzheimer's in the U.S. among which 73% were aged 75 or older.
Key players are focusing on providing new diagnostic tests which will drive the market growth during the forecast period. For instance, Attyloid GmbH, a biotechnology company, offers a biomarker-based laboratory test sFIDA (surface-based fluorescence intensity distribution analysis), for diagnosing Alzheimer’s disease. sFIDA can identify and quantify Aβ oligomers. These small protein aggregates play a significant role in the development and prognosis of Alzheimer’s disease. By sFIDA, differentiation between control samples and AD patient is easily possible.
Alzheimer’s disease Diagnostics and Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7,704.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.6% | 2033 Value Projection: | USD 11,282.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Luye Pharma Group, Vigil Neuroscience, Inc., Grifols, S.A., AbbVie Inc., Zydus Lifesciences Ltd., Merck KGaA, F. Hoffmann-La Roche Ltd., Novartis AG, Johnson & Johnson, Eli Lilly and Company, Biogen Inc., Siemens Healthcare GmbH, Merz Pharma, Eisai Co. Ltd., Lupin Limited, Teva Pharmaceutical Industries Ltd., Apotex Inc., Corium, Inc., Supernus Pharmaceuticals, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Alzheimer’s disease Diagnostics and Therapeutics Market Share (%), by Region, 2026
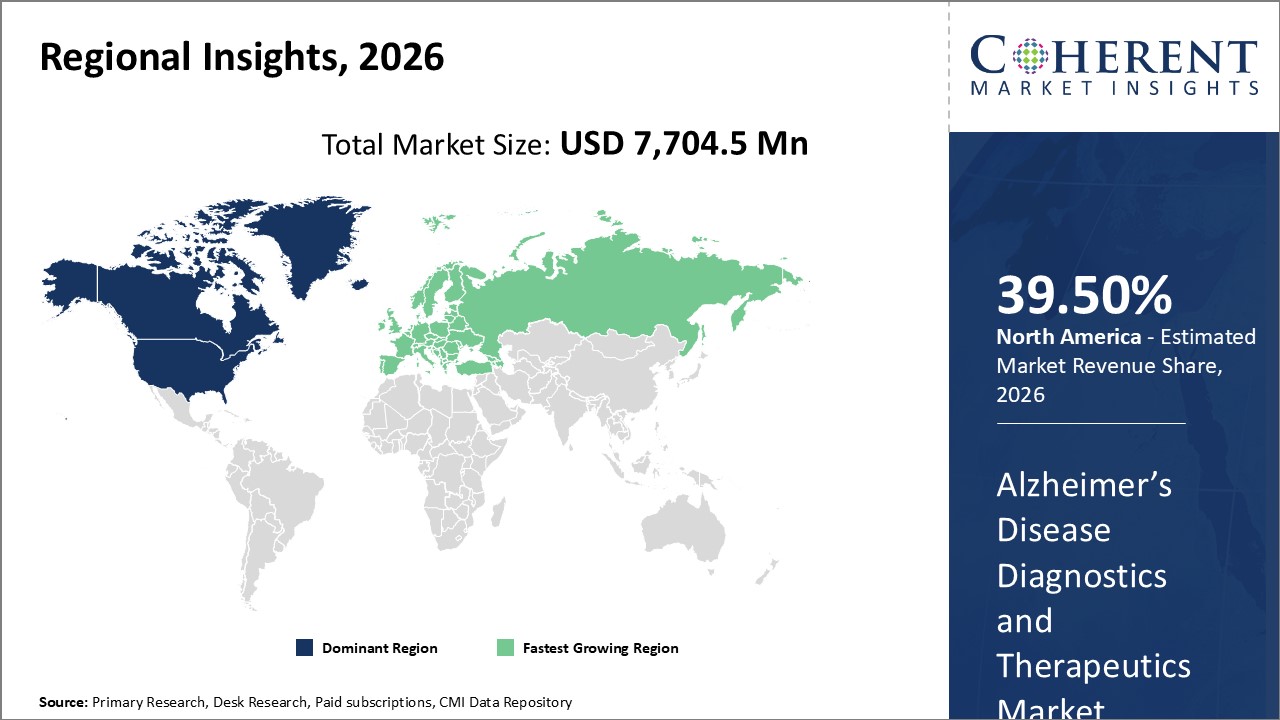
To learn more about this report, Request Free Sample
Global Alzheimer’s disease Diagnostics and Therapeutics Market: Key Developments
In October 2021, Salk Institute, a research institute in California, U.S., initiated a phase 1 clinical trial of the investigational Alzheimer’s drug CMS121 to evaluate its safety in humans.
Moreover, In June, 2021, Biogen, a multinational biotechnology company, and Eisai Inc., a U.S. subsidiary of Eisai Co., Ltd., announced a range of programs/initiatives intended to support access for all qualified patients, including traditionally underserved populations
Global Alzheimer’s disease Diagnostics and Therapeutics Market: Restraint
The uncertainty, failure, and discontinuation of the clinical trials of Alzheimer's drugs lead to the availability of limited treatment alternatives. This increases the costs related to research and development. Hence, it is expected to restrain the market growth. For instance, Amgen Inc. and Novartis AG discontinued their Phase 2 clinical trial for CNP520 (umibecestat), a BACE1 inhibitor for Alzheimer's disease, in July 2019, due to the lack of desired results.
Global Alzheimer’s disease Diagnostics and Therapeutics Market- Key Players
Major players operating in the global Alzheimer’s disease diagnostics and therapeutics market include Luye Pharma Group, Vigil Neuroscience, Inc., Grifols, S.A., AbbVie Inc., Zydus Lifesciences Ltd., Merck KGaA, F. Hoffmann-La Roche Ltd., Novartis AG, Johnson & Johnson, Eli Lilly and Company, Biogen Inc., Siemens Healthcare GmbH, Merz Pharma, Eisai Co. Ltd., Lupin Limited, Teva Pharmaceutical Industries Ltd., Apotex Inc., Corium, Inc., Supernus Pharmaceuticals, Inc.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients