When anticoagulant therapy needs to be reversed or neutralised due to bleeding problems, an overdose of anticoagulant therapy, or unexpected surgery, anticoagulant reversal drugs are required. Anticoagulant reversal is a key step in the treatment of patients who are taking an anticoagulant and are experiencing life-threatening bleeding. The reversal medications recommended by doctors are determined by the patient's anticoagulant of choice. Direct oral anticoagulants (DOACs) consists of dabigatran etexilate, a direct thrombin inhibitor, and specific inhibitors of stimulated coagulation factor X (apixaban, betrixaban, edoxaban, and rivaroxaban). DOACs are linked with lower rates of main and serious bleeding events compared with warfarin. In an emergency situation, clinicians need to attain rapid reversal of anticoagulation effects of the DOACs. Idarucizumab and andexanet alfa, which reverse the anticoagulant effects of dabigatran and FXa inhibitors are DOAC reversal drugs.
The global anticoagulant reversal drugs market is estimated to be valued at US$ 861.6 million in 2021 and is expected to exhibit a CAGR of 14.9% during the forecast period (2021-2028)
Figure 1. Global Anticoagulant Reversal Drugs Market Share (%) in Terms of Value, By Product Type, 2021
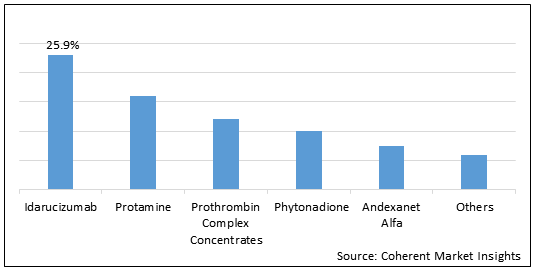
To learn more about this report, Request Free Sample
Key players operating in the global anticoagulant reversal drugs market are focusing on acquisitions and mergers, which is expected to drive the market growth.
Increasing acquisition provides the opportunity to expand commercial range of anticoagulant reversal drugs, which is expected to drive the market growth during the forecast period. For instance, in May 2020, Alexion Pharmaceuticals, Inc. announced acquisition of Portola Pharmaceuticals, Inc. The acquisition includes Andexx [coagulation factor Xa (recombinant), inactivated-zhzo], branded as Ondexxya, a Factor Xa inhibitor reversal medication. Alexion Pharmaceuticals, Inc. acquired Portola through a tender offer and subsequent merger with Odyssey Merger Sub Inc. Near-term diversification will be added by this acquisition to Alexion’s commercial portfolio.
Anticoagulant Reversal Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 861.6 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 14.9% | 2028 Value Projection: | US$ 2,282.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Portola Pharmaceuticals, Boehringer Ingelheim, CSL Behring, Bausch Health Companies Inc., Octapharma AG, Dr. Reddy's Laboratories, AMAG Pharmaceuticals, Inc., Pfizer, Inc., SGPharma Pvt. Ltd., Alps Pharmaceutical Ind. Co., Ltd., Fresenius Kabi AG, Mundipharma International Limited, and Hisamitsu Pharmaceutical Co., Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Anticoagulant Reversal Drugs Market Share (%), By Distribution Channel, 2021
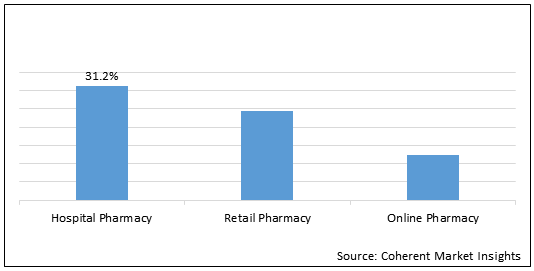
To learn more about this report, Request Free Sample
Increasing requirement of anticoagulants for blood-related disorders boost the demand for anticoagulant reversal drugs in case of uncontrolled bleeding, which is anticipated to drive the market growth.
Increasing demand for anticoagulant reversal drugs in case of uncontrolled bleeding is expected to drive the market growth during the forecast period. Some of the common bleeding disorders include blood clots, Von Willebrand disease, and hemophilia A and B. One out of 100 individuals are affected by Von Willebrand disease as it is a very common bleeding disorder. Bleeding disorders can cause undesirable bleeding from the patient’s body as well as affect blood clotting. For instance, there are around 30,000 to 33,000 people in the U.S. who are suffering from hemophilia and approximately 60% of them are suffering from acute hemophilia, according to the National Hemophilia Foundation (NHF).
Global Anticoagulant Reversal Drugs Market– Impact of Coronavirus (COVID-19) Pandemic
The unanticipated COVID-19 epidemic had a significant beneficial influence on anticoagulant reversal medicines, with an increase in the number of research and development efforts focused primarily on COVID-19 infection. For instance, about ten clinical trials are now underway to assess the efficacy of factor Xa inhibitors in COVID-19 patients, as revealed by a paper published in the American Journal of Cardiovascular Drugs, in 2020. The development of methods for the parenteral delivery of these medicines that are useful in the treatment of severe COVID-19 patients. As a result, the COVID-19 pandemic will drive the global anticoagulant medication market throughout the projection period.
Global Anticoagulant Reversal Drugs Market: Restraint
The high cost of anticoagulant reversal drugs is one of the restraining factor that is expected to hinder the market growth during the forecast period. 4-F PCC (Kcentra) supports in treating bleeding cases due to apixaban and rivaroxaban and it is administered at 50 units/k, according to the American College of Cardiology. Its average wholesale price is US$ 2.90 per unit, thus treating a patient whose weight is 100 kg that cost about US$ 14,500.
Key Players
Major players operating in the global anticoagulant reversal drugs market include Portola Pharmaceuticals, Boehringer Ingelheim, CSL Behring, Bausch Health Companies Inc., Octapharma AG, Dr. Reddy's Laboratories, AMAG Pharmaceuticals, Inc., Pfizer, Inc., SGPharma Pvt. Ltd., Alps Pharmaceutical Ind. Co., Ltd., Fresenius Kabi AG, Mundipharma International Limited, and Hisamitsu Pharmaceutical Co., Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients