Antiplatelet drugs market is estimated to be valued at USD 2,907 Mn in 2026 and is expected to reach USD 4,920 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.83% from 2026 to 2033.
The global antiplatelet drugs market is observing enormous growth due to the increase in cardiovascular diseases such as myocardial infarction, arterial thrombosis, and stroke. Rising adoption of guideline-based therapies for preventing platelet aggregation coupled with growing awareness about chronic heart conditions globally propels the demand for antiplatelet medications. Additionally, innovative drug formulation developments in various formulations such as oral and intravenous significantly enhance patient compliance and therapeutic outcomes.
|
Current Events |
Description and its impact |
|
Regulatory and Patent Landscape Shifts |
|
|
Geopolitical Tensions and Supply Chain Disruptions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on drug class, the ADP Receptor Inhibitors segment is anticipated to contribute the highest market share of 42% in 2026. These medications have commonly been prescribed to prevent platelet aggregation in individuals vulnerable to heart attacks or strokes. Their success in controlling thrombotic events has made these the go-to choice for physicians and healthcare practitioners across the globe.
For instance, in March 2025, SERB Pharmaceuticals announced the final results from the pivotal Phase 3 REVERSE‑IT trial demonstrating that bentracimab, a human monoclonal antibody designed to reverse the antiplatelet effects of the ADP receptor inhibitor ticagrelor.
In terms of mode of administration, the oral segment is estimated to account for the highest market share of 75% by 2026. Oral antiplatelet agents are preferred for chronic therapy as they are more patient-friendly for administration, thereby facilitating adherence to treatment regimens. Oral formulations account for a higher segment share due to their potential to be formulated as combinations to treat cardiac conditions.
For instance, in October 2025, Alembic Pharmaceuticals announced final approval from the US Food and Drug Administration (US FDA) for generic Ticagrelor Tablets, 60 mg, a critical oral antiplatelet drug indicated to lower the rate of thrombotic cardiovascular events including CV death, MI, and stroke, with acute coronary syndrome or history of cardiovascular events.
By application, the myocardial infarction segment is estimated to contribute the highest market share of 35% in 2026. Antiplatelet drugs play a vital role in the management and prevention of heart attacks by offering protective cover from blood clots. Increased prevalence of coronary artery diseases and treatment guidelines have fueled this segment of the application market.
For instance, in August 2025, the TARGET‑FIRST trial presented at ESC Congress 2025 showed that in low‑risk acute MI patients who underwent early complete revascularisation, transitioning to P2Y12 inhibitor monotherapy (without continued aspirin) after one month of DAPT was noninferior for cardiovascular outcomes and reduced clinically relevant bleeding.
To learn more about this report, Request Free Sample
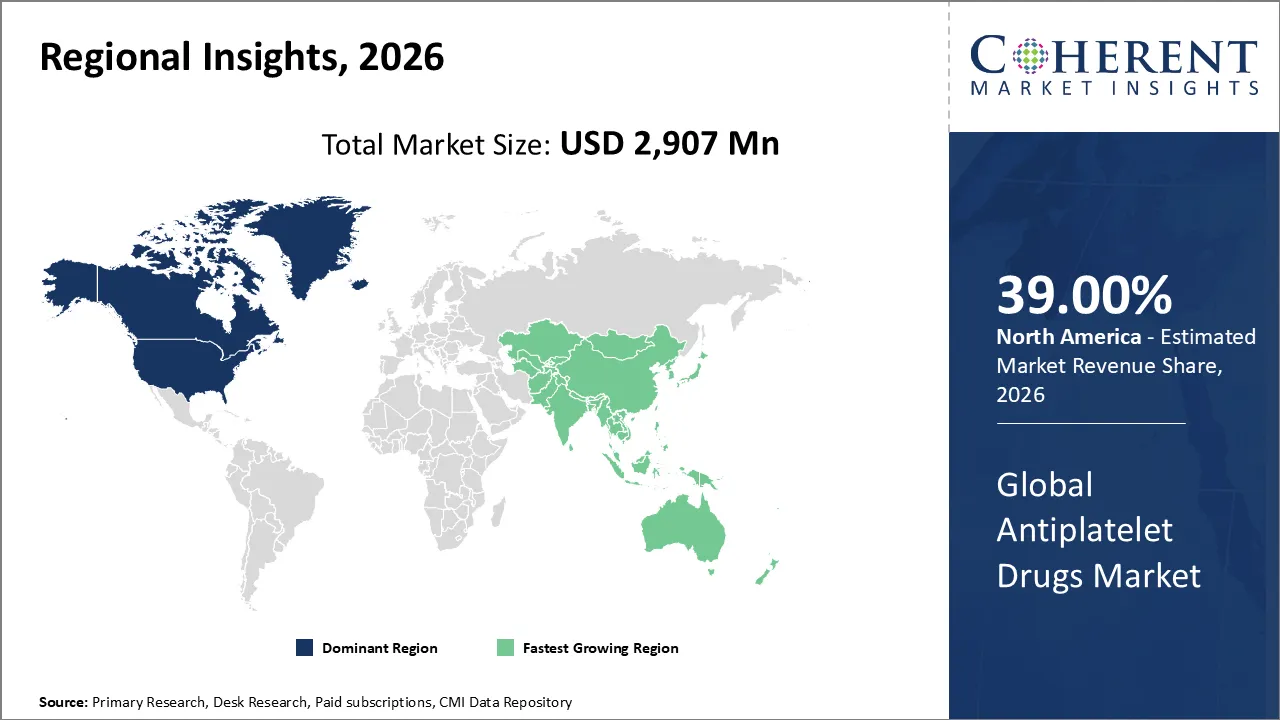
North America is expected to have a strong hold on the antiplatelet drugs market in 2026, with a projected share of 39%. The drivers of the market in the North American region are the well-established medical infrastructure, the increased rate of CVDs, guideline-based use of the drug, and the presence of major organizations in the country. The presence of established hospitals that offer proper heart care services supports the view that the market is expected to grow.
For instance, in March 2025, the U.S. Food and Drug Administration (FDA) approved the first generic version of Xarelto (rivaroxaban) 2.5 mg tablets, which are to be taken in combination with aspirin to lower the risk of major cardiovascular and thrombotic events in adults suffering from CAD and PAD.
The Asia Pacific region has registered the highest rate of growth for antiplatelet drugs due to increased prevalence of cardiovascular diseases, improvement in healthcare infrastructure, and increasing health awareness among people. China, India, and Japan are reporting a boost in antiplatelet drug adoption due to public health initiatives and an uptick in hospital infrastructure.
For instance, in October 2025, Alembic Pharmaceuticals received final US FDA approval for generic Ticagrelor Tablets 60 mg, a critical oral antiplatelet medication.
The U.S. antiplatelet drugs market is growing, especially due to the large prevalence of cardiovascular diseases, universal acceptance of guideline-based drug regimens, and strong healthcare infrastructure. Increasing interventions and preventive measures for myocardial infarction, arterial thrombosis, and percutaneous coronary interventions will boost demand for both brand and generic drugs.
For instance, in October 2025, Alembic Pharmaceuticals announced that it had received final approval from the U.S. Food and Drug Administration (USFDA) for its Abbreviated New Drug Application (ANDA) for Ticagrelor Tablets 60 mg, a generic equivalent to AstraZeneca’s Brilinta®.
The market for antiplatelet medications is expanding in China due to the country's growing emphasis on cardiovascular disease prevention and treatment. Patient uptake of antiplatelet medications is being aided by public health campaigns, better hospital facilities, and growing use of guideline-based treatments.
For instance, in June 2025, Pfizer China announced the launch of Brilinta® (ticagrelor) in hospital networks across major Chinese cities to strengthen acute coronary syndrome treatment. The launch aligns with China’s national strategies to reduce cardiovascular mortality and enhance patient outcomes.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,907 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.83% | 2033 Value Projection: | USD 4,920 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca Plc, Boehringer Ingelheim GmbH, Bristol-Myers Squibb Company, Daiichi Sankyo, Inc., Eli Lilly and Company, Merck & Co., Inc., Otsuka Pharmaceutical Company, Ltd., Sanofi, and The Medicines Company. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing occurrence of cardiovascular conditions such as myocardial infarction, arterial thrombosis, and cerebral thrombosis is one of the major factors influencing the market for antiplatelet agents. The rising number of patients undergoing percutaneous coronary interventions and other cardiac procedures contributes to the increasing need for oral and intravenous antiplatelet drugs. Increasing awareness of the benefits of antiplatelet therapy among healthcare professionals and patients contributes to market expansion.
The drug market presents considerable growth opportunities through the introduction of new antiplatelet drugs, reversals, and generic drugs. For instance, generic ticagrelor tablets and the development of reversal agents, such as bentracimab, result in safer and more affordable treatment options. New oral and intravenous drugs with focus on improved efficacy and reduced risk of bleeding can improve existing products, reaching new patient segments, extending treatment adherence, and providing opportunities for pharmaceutical companies to generate revenues.
The antiplatelet drugs market has a revolution going on, driven by clinical innovation for patient-centric therapies. Innovation such as reversal agents and P2Y12 inhibitor regimens tailored toward the optimization of bleeding risk and antithrombotic efficacy are transforming the way clinicians practice.
Geographically, North America remains the engine of innovation in advanced healthcare infrastructure and early adoption of specialized therapies, while Asia Pacific and other emerging regions experience rapid growth on the back of increasing awareness of cardiovascular health and growing preventive care practices.
Overall, the market develops into a place where scientific innovation, evidence-based medicine, and treatment for the patient intersect. Those companies that focus on next-generation antiplatelet drugs, reversal agents, and physician education will be in the leading positions, as providers more and more turn their attention to therapies that can provide efficacy with safety in the management of cardiovascular risks.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients