Ataxia is a group of degenerative disorders associated with the cerebellum in the brain, which is responsible for regulating balance and coordination of the body. Ataxia does not have a cure and only its symptoms such as weakness, difficulty in movement and others can be treated. Ataxia can be of several types such as spinocerebellar ataxia, Friedreich’s ataxia, ataxia-telangiectasia, multiple system atrophy (MSA), episodic ataxia, and others.
Spinocerebellar ataxia (SCA) is caused due to genetic defects, which lead to impairment of nerve fibers carrying neural signals in the brain, thus resulting in degeneration of cerebellum. People with this condition tend to face difficulties in movement and balance, develop weakness in body, difficulty in eye movement, and others. SCA is a progressive type of ataxia and can occur at any age. Friedreich’s ataxia is a type of genetic ataxia wherein in addition to common movement related symptoms, people also experience stiffness and loss of sensation in muscles, weakness in the body, heart condition (hypertrophic cardiomyopathy), and others.
Ataxia-telangiectasia, also known as louis-bar syndrome is a genetic ataxia, which is seen mostly in new born babies and young children. Some of the characteristic symptoms of ataxia-telangiectasia include appearance of enlarged blood vessels in the eyes and on the facial skin. Multiple system atrophy is an acquired type of ataxia and is a form of Parkinsonism, which affects the cerebellum and autonomic nervous system (ANS). ANS is responsible for controlling involuntary functions in the body such as blood pressure, urination, and others. Common symptoms associated with multiple system atrophy include erectile dysfunction, rapid blood pressure drop when a person stands up, difficulty coordinating movements, and others. MSA usually develops in adults of age 30 and above. Episodic ataxia is a genetic ataxia characterized by recurring troubles with movement and balance which can happen multiple times a day or just once or twice a year.
The global ataxia market is estimated to be valued at US$ 30,880.5 million in 2021 and is expected to exhibit a CAGR of 9.4% over the forecast period (2021-2028).
Figure 1. Global Ataxia Market Share (%), By Region, 2021
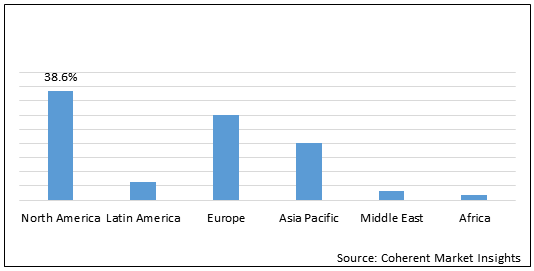
To learn more about this report, Request Free Sample
The increasing incidence of ataxia is expected to drive the growth of the global ataxia market over the forecast period.
For instance, according to an article published in the Journal of Neurology, in October 2018, the global prevalence of spinocerebellar ataxia (SCA) is estimated to three 3 in 100,000 people as of 2018, wherein SCA3 subtype of spinocerebellar ataxia was found to be the most common type around the globe. The same source also stated that SCA6 subtype of spinocerebellar ataxia is one of the most common autosomal dominant cerebellar ataxia in the North of England, with a global prevalence of 5.2 per 100,000 persons.
Ataxia Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 30,880.5 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 9.4% | 2028 Value Projection: | US$ 47,360.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., CRISPR Therapeutics, Acorda Therapeutics, Capsida Biotherapeutics Inc., Larimar Therapeutics, Inc., Healx, Intellia Therapeutics, Inc., Editas Medicine, bluebird bio, Inc., Sanofi, Merck & Co., Inc., H. Lundbeck A/S, Cellectis SA, Bio-Techne, GlaxoSmithKline plc, and Allergan |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Ataxia Market Share (%), By Treatment Type, 2021
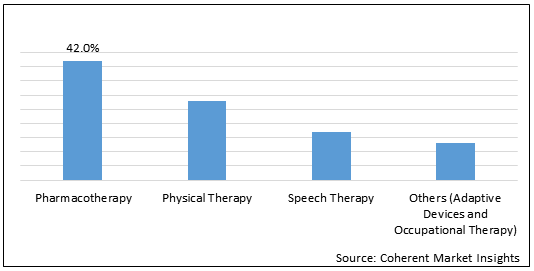
To learn more about this report, Request Free Sample
The increasing research and development activities for the development of novel therapeutics for the treatment of ataxia is expected to drive the market growth over the forecast period.
Key players operating in the market are focusing on submitting application for approval of drugs therapy for the treatment of ataxia, which is expected to drive growth of the global ataxia market over the forecast period.
For instance, in March 2019, Biohaven Pharmaceuticals, a U.S. based clinical stage biopharmaceutical company initiated its Phase III clinical trial of its third-generation prodrug troriluzole indicated for the treatment of spinocerebellar ataxia.
Moreover, according to an article published in the Neurobiology of Disease journal, in January 2021, an oral drug called leriglitazone has shown benefits in treating Friedrich's ataxia in cellular and animal models. Leriglitazone is manufactured by Minoryx Therapeutics, a Spain based clinical stage Biotechnology Company and is currently in advanced Phase II clinical trials for treatment of Friedrich's ataxia.
Global Ataxia Market – Impact of Coronavirus (Covid-19) Pandemic
Coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19, as pandemic on March 11, 2020. According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 234,553,539 cases and 4,796,222 deaths due to coronavirus (COVID-19) were reported till October 3, 2021, across the globe.
The COVID-19 pandemic and lockdown in various countries across the globe has negatively impacted the financial status of businesses across all sectors. The COVID-19 pandemic has impacted the entire supply chain of the healthcare industry mainly due to strict lockdown in several regions. However, COVID-19 pandemic is expected to have a positive impact on the global ataxia market as recent research studies have established a link between ataxia and COVID-19 infection. For instance, according to an article published by the BMC (biomedcentral) Infectious Diseases journal in March 2021, SARS-CoV-2 infection can be a potential cause of post-infectious neurological diseases such as ataxia, thus high-dose corticosteroid treatment must be considered if a post-infectious immune-mediated mechanism is suspected in a COVID-19 positive patient.
Global Ataxia Market Restraint
The rarity of ataxia disease is the major hurdle in conducting clinical trials for its treatment and this is expected to hinder growth of the global ataxia market over the forecast period. For instance, according to an article published by the Annals of Clinical and Translational Neurology journal, in May 2021, the rarity of spinocerebellar ataxia (SCA) is the biggest challenge faced while conducting clinical trials for treatment of SCA as there are insufficient number of people suffering from SCA which could be recruited for a given clinical trial.
Key Players
Major players operating in the global ataxia market include Pfizer, Inc., CRISPR Therapeutics, Acorda Therapeutics, Capsida Biotherapeutics Inc., Larimar Therapeutics, Inc., Healx, Intellia Therapeutics, Inc., Editas Medicine, bluebird bio, Inc., Sanofi, Merck & Co., Inc., H. Lundbeck A/S, Cellectis SA, Bio-Techne, GlaxoSmithKline plc, and Allergan
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients