The Automated Endoscopy Reprocessor market is anticipated to grow at a CAGR of 5.10% with USD 767.67 Mn in 2026 and is expected to reach USD 1,087.9 Mn in 2033. The increasing number of gastrointestinal (GI) diseases demanding endoscopic procedures are expected to drive the automated endoscope reprocessor market growth. The U.S. records more than 370,000 new GI cancer cases and nearly 200,000 related deaths each year, including colorectal and pancreatic cancers. Increasing adoption rate of endoscope reprocessors in hospitals for infection prevention and control is expected to fuel the global automated endoscopy reprocessor market revenue. AERs can enhance reliability and consistency of endoscope reprocessing by standardizing several important reprocessing steps, thereby reducing the possibility of human error.
High-level disinfection technologies are improving the way hospitals clean and disinfect endoscopes. Advanced chemical disinfection systems use solutions such as peracetic acid, OPA, and glutaraldehyde with automated monitoring to ensure safe and effective cleaning. The Olympus ENDOCAVITY system, used by hospitals like Mayo Clinic, Johns Hopkins Hospital, etc., helped minimize reprocessing time by 40% as well as cut procedure turnaround time from 45 minutes to 25 minutes while discarding 99.9% of pathogens.
Similarly, the STERIS System 1E has been adopted by leading healthcare providers including Cleveland Clinic, Massachusetts General Hospital, etc. This system automated chemical handling, lower staff exposure to disinfectants by 95% and improving workplace safety. Hospitals using the technology also reported zero chemical-related staff incidents after implementation.
|
Current Event |
Description and its Impact |
|
Stricter FDA Guidelines for Endoscope Reprocessing |
|
|
Growing Focus on Infection Prevention in Hospitals |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Double Basin segment is projected to account for the largest share of product type in 2026, representing approximately 60% of the total volume. The superior operational efficiency along with enhanced workflow capabilities in high-volume healthcare facilities is one of the factors responsible for the growth of the segment.
By 2037, the U.S. is projected to face a shortfall of approximately 1,390 gastroenterologists, according to the U.S. Health Resources & Services Administration. As demand for GI care continues to outpace the supply of physicians, health systems are facing escalating challenges in sustaining timely as well as equitable access to care.
Their ability to simultaneously process multiple endoscopes, which significantly reduces turnaround times and increases throughput capacity in busy endoscopy departments is further stimulating the segmental growth.
This simultaneous processing capability is particularly crucial in large hospitals and specialized gastroenterology centers where multiple procedures are conducted daily. The guidelines by the World Health Organization on healthcare facility efficiency target the importance of streamlined reprocessing workflows, which double basin systems inherently provide through their dual-chamber design.
To learn more about this report, Request Free Sample
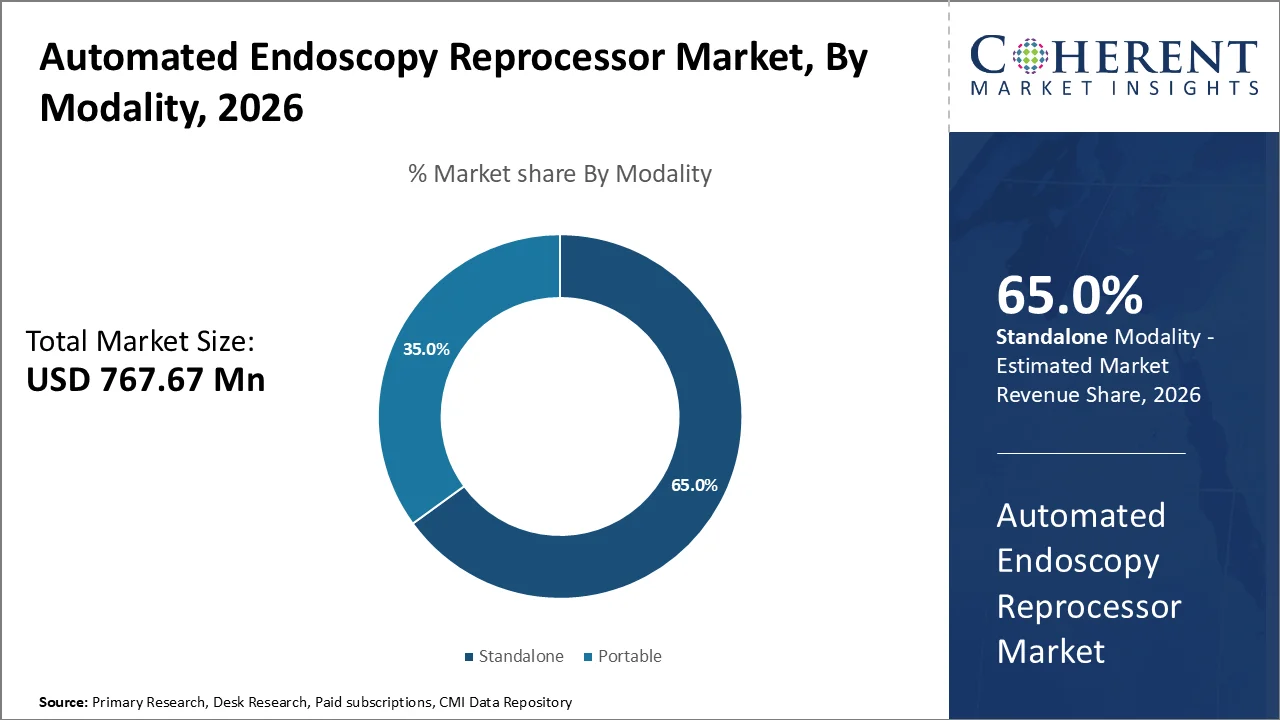
Based on modality, Standalone dominates the market, accounting for a significant 65% share in 2026, their superior processing capabilities, overall cleaning protocols, along with robust infrastructure requirements that align with the operational demands is augmenting growth.
Their ability to accommodate multiple endoscopes concurrently, with most systems capable of processing between 2-12 endoscopes per cycle depending on the model as well as manufacturer specifications is propelling the segmental growth. This high-capacity processing capability becomes crucial in busy medical facilities where endoscopic procedures including colonoscopies, upper endoscopies, bronchoscopies, etc., are performed in rapid succession.
Hospital dominates the market, accounting for a significant 64% share in 2026, the extensive endoscopic procedure volumes, comprehensive infrastructure requirements, as well as strict infection control protocols mandated by healthcare regulatory bodies.
Hospitals represent the backbone of healthcare delivery systems globally, performing the high volume of complex endoscopic procedures including colonoscopies, upper gastrointestinal endoscopies, Bronchoscopy specialized therapeutic interventions, etc. Bronchoscopy was one of the most prevalent in Asia compared to Europe (25% versus 5%).
The guidelines by World Health Organization on decontamination and reprocessing of medical devices emphasize the critical importance of automated reprocessing systems in hospital settings, particularly given the high patient turnover as well as diverse case complexity that characterizes hospital environments.
To learn more about this report, Request Free Sample
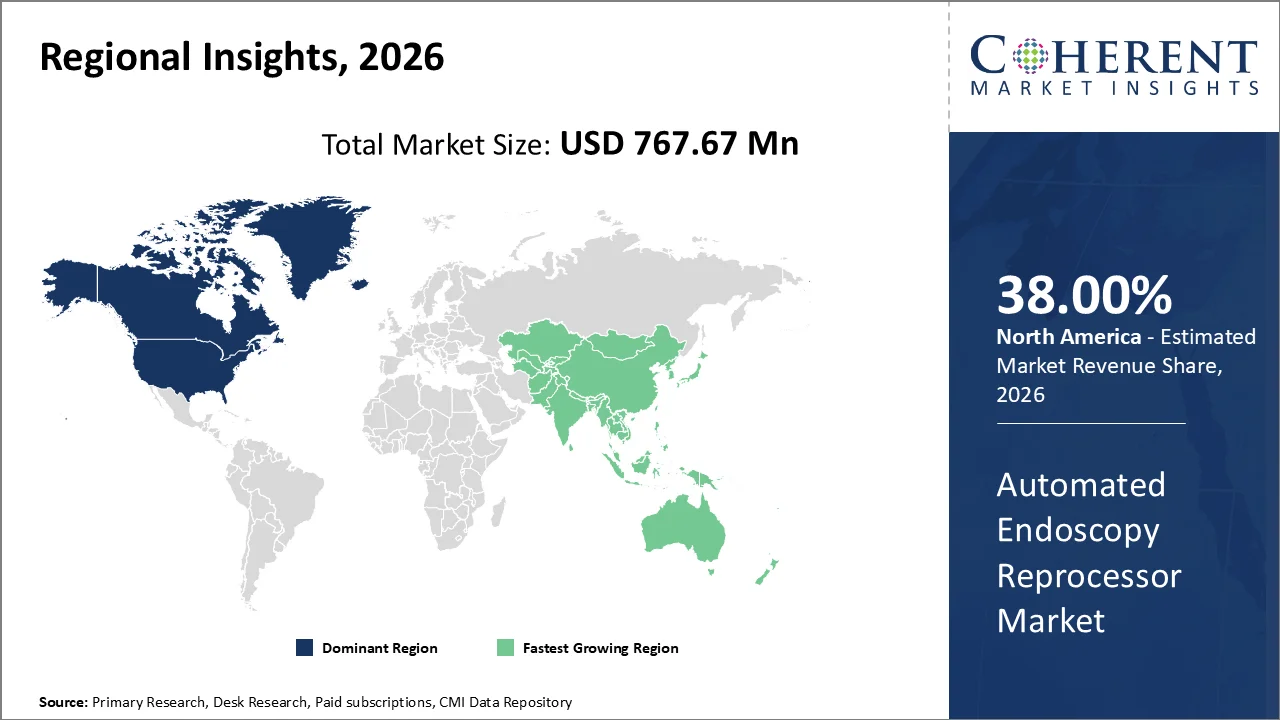
North America account 38% market share in 2026, the region's well-established healthcare infrastructure, stringent regulatory standards, as well as widespread adoption of advanced medical technologies. There was 871 Medical Device Manufacturing in the US businesses as of 2025, an increase of 1.2% from 2024.
The robust healthcare spending patterns, with institutions continuously investing in state-of-the-art medical equipment to ensure patient safety along with operational efficiency is fueling the market. The presence of major medical device manufacturers as well as healthcare technology companies in the United States and Canada has supported an ecosystem where innovation thrives and new technologies are rapidly integrated into clinical practice.
The Asia Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 6.2%. The sophisticated healthcare infrastructure and advanced medical technology adoption patterns that have been replicated across Asia-Pacific healthcare facilities. As per 2025 OECD report, as of 2021, Singapore had the most physicians per person among SEA countries (2.6 per 1 000 people).
For example, the Joint Commission International (JCI) accreditation standards, which originated in North America, are now implemented across over 1,000 healthcare facilities in the Asia Pacific region, mandating specific endoscopy reprocessing protocols that favor automated systems.
The U.S. contributes the highest share in the Automated Endoscopy Reprocessor (AER) market in North America owing to its robust healthcare infrastructure, extensive hospital network, as well as strict regulatory frameworks governing medical device sterilization.
The United States houses over 6,000 hospitals and numerous ambulatory surgical centers, making a high demand for automated endoscopy reprocessing equipment. The Centers for Disease Control and Prevention (CDC) has made a comprehensive guidelines for endoscope reprocessing, mandating high-level disinfection protocols that drive adoption of automated systems.
For example, the CDC's surveillance report documented healthcare-associated infections linked to inadequately processed duodenoscopes, prompting hospitals nationwide to invest in advanced AER systems. Major healthcare systems like Kaiser Permanente, Cleveland Clinic, etc., have implemented standardized AER protocols across their facilities, show growth of the market.
Japan contributes the highest share in the Automated Endoscopy Reprocessor market in APAC owing to its sophisticated healthcare infrastructure, stringent infection control protocols, and advanced medical technology adoption. The emphasis by the Japanese healthcare system on patient safety and quality assurance has driven high implementation of automated endoscopy reprocessing solutions across hospitals as well as ambulatory surgical centers.
The Japan Gastroenterological Endoscopy Society (JGES) has established comprehensive guidelines for endoscope reprocessing that mandate the use of automated systems in major medical facilities, majorly boosting market penetration. For example, leading Japanese hospitals like Tokyo University Hospital and Osaka Medical Center have fully integrated automated endoscopy reprocessors into their gastroenterology departments, with some facilities processing over 200 endoscopic procedures daily using these automated systems.
Some of the major key players in Automated Endoscopy Reprocessor market include, Cantel Medical Corp., Johnson & Johnson, Laboratoires Anios, Olympus Corporation, Steelco SpA, Steris Plc., Getinge Group, Hoya Group, and Metall Zug AG
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 767.67 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.10% | 2033 Value Projection: | USD 1,087.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Cantel Medical Corp., Johnson & Johnson, Laboratoires Anios, Olympus Corporation, Steelco SpA, Steris Plc., Getinge Group, Hoya Group, and Metall Zug AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients