The global biodefense market is estimated to be valued at USD 19.12 Bn in 2026 and is expected to reach USD 32.13 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 7.7% from 2026 to 2033.
To learn more about this report, Request Free Sample
The market is experiencing strong growth due to rising government investments in biological threat preparedness and increasing awareness about bioterrorism risks. Moreover, the growing demand for vaccines, diagnostics, and protective equipment is expected to boost the Biodefense Market value. However, factors such as high development costs, ethical concerns surrounding dual-use research, and stringent regulatory hurdles are expected to hamper the growth of the global biodefense market.
|
Event |
Description and Impact |
|
Russia-Ukraine Conflict and Geopolitical Tensions |
|
|
U.S. BARDA and Strategic National Stockpile Modernization |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
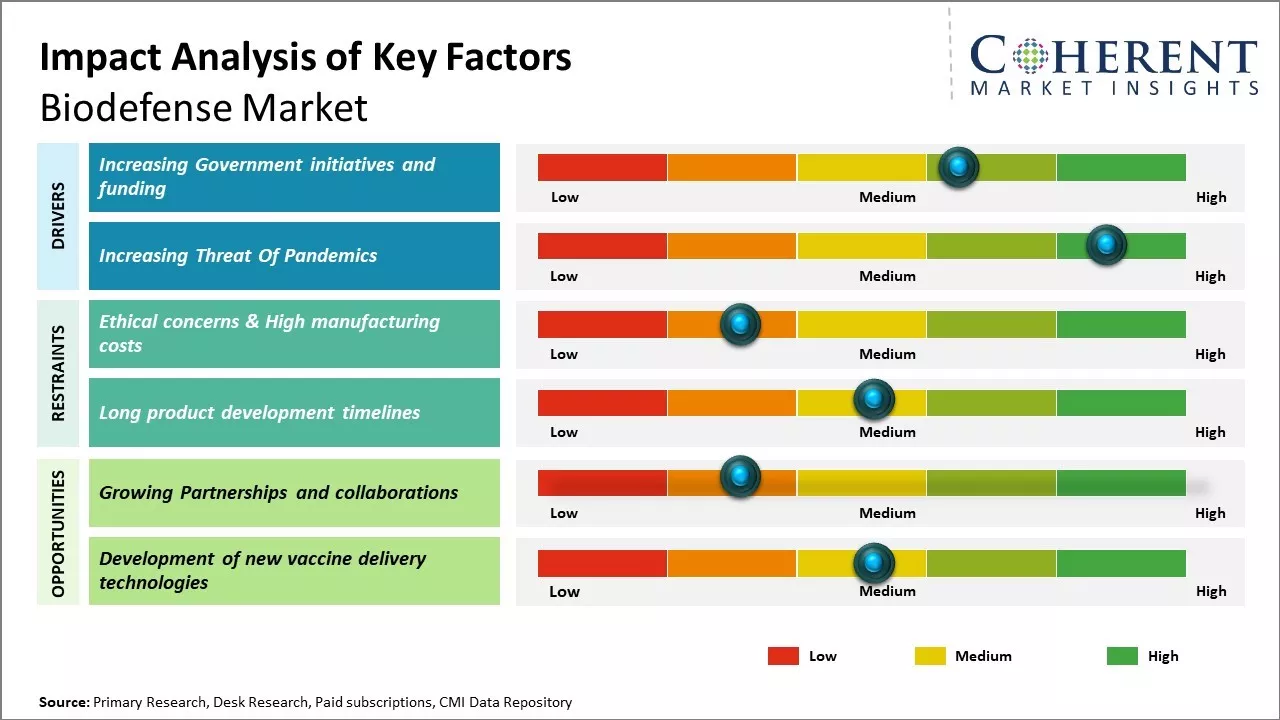
Government initiatives and funding have played a key role in spurring innovation and development in the biodefense sector. In recent years, many developed nations have acknowledged biodefense and pandemic preparedness as strategically important objectives. For example, the U.S. allocated US$9 billion towards biodefense between 2021 and 2022 according to data from the U.S. Department of Health and Human Services.
These funds are being utilized to bolster stockpiles of critical medical countermeasures like vaccines and therapeutics. Several projects focused on developing rapid response capabilities, such as new vaccine platforms allowing production within months, have received major government grants.
Ethical concerns surrounding some biodefense technologies have slowed market adoption and created regulatory hurdles. As biotechnologies advance the ability to genetically modify or create harmful pathogens, difficult questions arise around dual-use risks. Governments and oversight bodies struggle to balance security needs with preventing misuse that could endanger public health.
Lengthy review and licensing procedures aim to ensure technologies only target legitimate threats, but this scrutiny increases development timelines and costs. Some promising areas like gene drives that could eliminate wild virus carriers face fierce opposition from those worried about environmental or socially unequal impacts. Addressing these societal anxietals in a transparent, inclusive manner is critical to gaining broad acceptance for biodefense solutions.
High manufacturing costs also pose a barrier, as developing and producing medical countermeasures for rare and emerging diseases has little initial commercial viability. According to the WHO, between 2017 and 2019 fewer than 5% of global health R&D funding was allocated for diseases that account for almost 90% of the global disease burden.
Partnerships and collaborations could provide significant opportunities for growth in the biodefense market research by facilitating research and development. Working together, public and private entities have better potential to address today's complex biosecurity challenges. No single organization possesses all the necessary resources and expertise to tackle threats such as pandemics or biological attacks on their own.
Through cooperation and shared knowledge, partners can achieve more advanced scientific understanding and technological solutions than any could alone. For example, the International Centre for Genetic Engineering and Biotechnology has brought together over 30 international scientific institutions to work on rapid detection kits, vaccines, and therapeutics against potential biological weapons agents.
Collaboration also allows partners to capitalize on their respective strengths. Government research agencies can provide long-term funding support for basic research while private firms contribute applied science, manufacturing capabilities, and marketing networks.
Anthrax is expected to contribute the highest share of the biodefense market, accounting for 29.6% of the market share in 2026, among the product segment due to its widespread potential as a biological weapon. As a select agent with the ability to sicken or kill on inhalation, anthrax poses grave threats that drive defense spending.
Its spores form and survive in infected hosts, allowing the disease to lie dormant for decades before potential release and spread. Government stockpiling aims to treat mass exposures from possible aerosol attacks. Since the 2001 anthrax letter attacks that killed five, defenses have expanded against this lethal threat.
Research progresses on improved vaccines to bolster immune responses for high-risk groups. Approaches target both the lethal toxins anthrax produces and the bacteria itself. Natural outbreaks also occur, keeping anthrax a public health concern. Livestock vaccination programs help control accidental spread from infected animals to humans. However, bioterrorists could potentially weaponize anthrax stocks from such outbreak zones, further necessitating robust defenses. Detection technologies improve for early identification of anthrax in the environment.
To learn more about this report, Request Free Sample
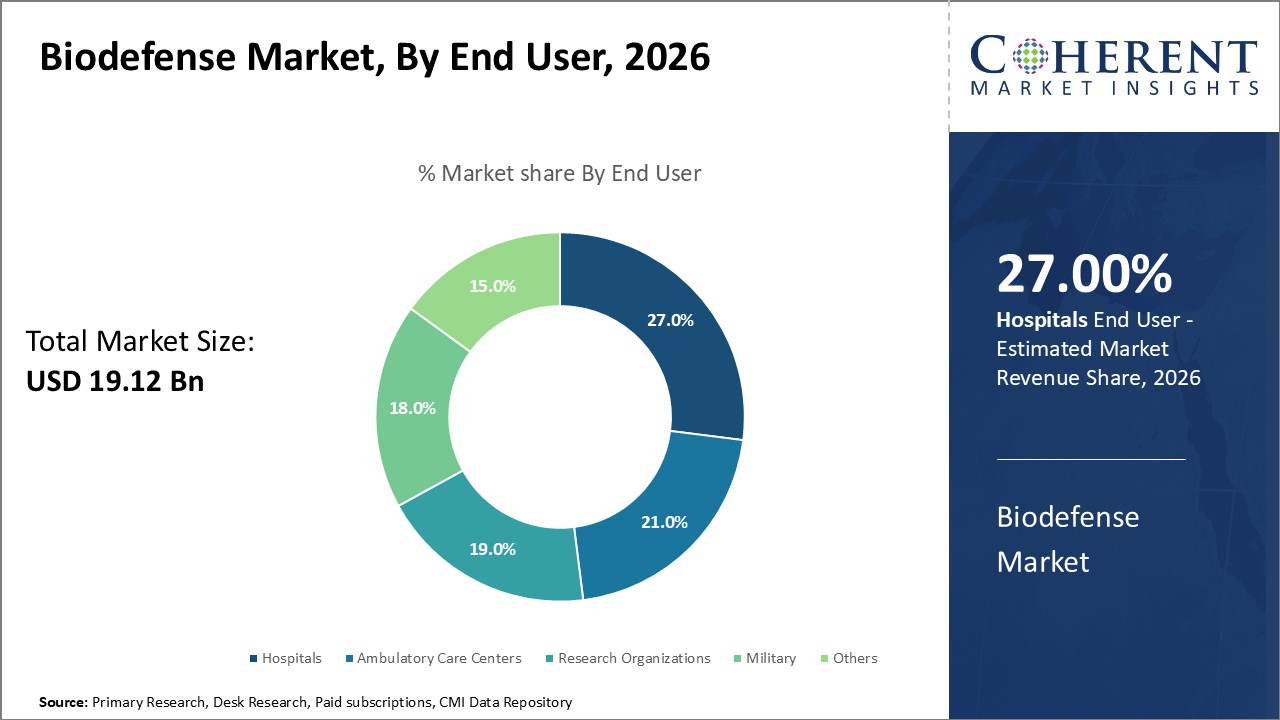
Among the end user segment, hospitals are expected to contribute the highest share to the biodefense market, accounting for 27% of the market share in 2026.This stems from hospitals playing a key role in acute treatment scenarios during biological attacks or outbreaks. In such emergencies, hospitals bear the primary responsibility of triaging and caring for mass casualties. Their infrastructure and resources must support surge capacities for patients presenting with unknown infectious diseases or exposure to toxic agents.
Emergency departments and intensive care units would see sharp rises in patient volumes during biodefense incidents and require specialized biodefense outbreak containment and decontamination capabilities. Personal protective equipment, quarantine spaces, and diagnostic testing equipment tailored for rare pathogens help hospitals effectively respond. Surging stockpiles of biodefense drugs and vaccines target hospitals to allow rapid prophylaxis of healthcare workers and at-risk groups.
Retrofitting current hospital facilities with biosafety levels aids in safely isolating and diagnosing infected patients. Negative pressure rooms help contain dangerous pathogens. Resources expand for providing longer hospital stays as required for severe cases of biothreat agents. Research into rapid point-of-care testing permits quicker diagnoses and exposure assessments in emergency settings.
To learn more about this report, Request Free Sample
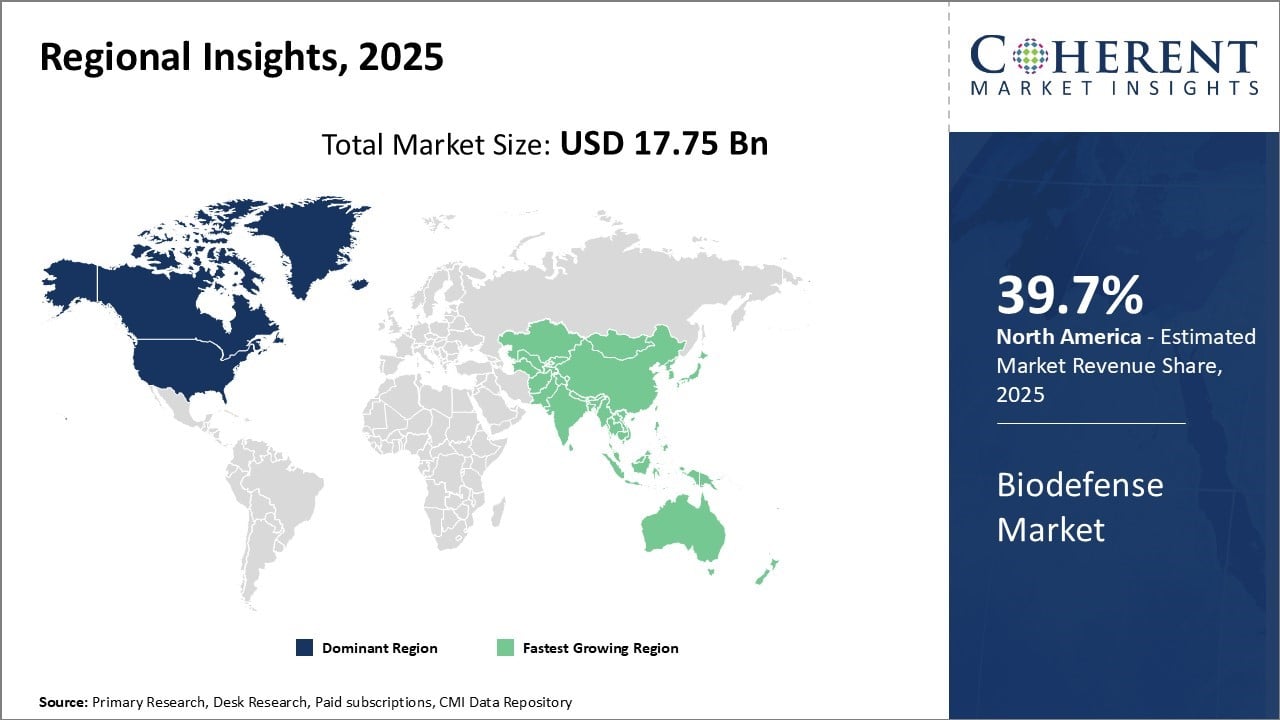
North America has established itself as the dominant region in the global biodefense market, accounting for a market share of 39.7% in 2026. The U.S. has the most robust biodefense programs and makes significant investments to counter biological threats through its government agencies like the Centers for Disease Control and Prevention and National Institutes of Health. It has also undertaken large initiatives like the BioShield Act that has created guaranteed markets for biodefense products.
Leading pharmaceutical companies and research institutions based in the U.S. have extensive expertise and resources devoted to developing drugs and vaccines for biological threats. Due to the strong government support and presence of major industry players, North America accounts for the largest share of the global market.
The Asia Pacific region has emerged as the fastest growing market for biodefense. Countries like China, India, South Korea, and Japan are increasing their focus and spending on public health preparedness against biological disasters and pandemics. With some of the largest populations globally and history of disease outbreaks, the threat perception is high. Furthermore, rising standards of living and healthcare expenditures have enabled greater investments towards biodefense.
The region is also a significant producer and consumer of biodefense products, accounting for sizeable import/export volumes. Manufacturers view Asia Pacific as an attractive market given the large population base and growing procurement power of government and healthcare systems.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 19.12 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.7% | 2033 Value Projection: | USD 32.13 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
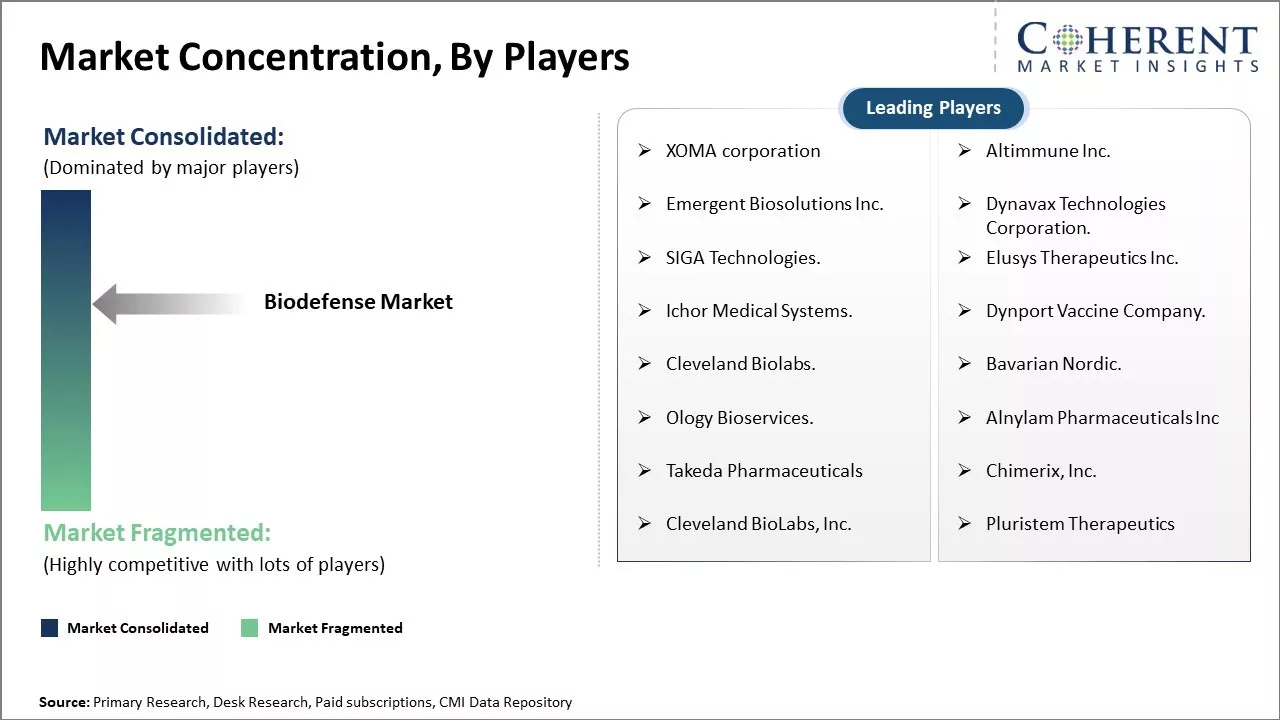
XOMA corporation, Altimmune Inc., Emergent Biosolutions Inc., Dynavax Technologies Corporation., SIGA Technologies., Elusys Therapeutics Inc., Ichor Medical Systems., Dynport Vaccine Company., Cleveland Biolabs., Bavarian Nordic., Ology Bioservices., Alnylam Pharmaceuticals Inc, Takeda Pharmaceuticals, Chimerix, Inc., Cleveland BioLabs, Inc., and Pluristem Therapeutics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients