Blood Ketone Meter Market is estimated to be valued at USD 448.0 Mn in 2026 and is expected to reach USD 724.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.1% from 2026 to 2033.
Analysts’ Views on Global Blood Ketone Meter Market:
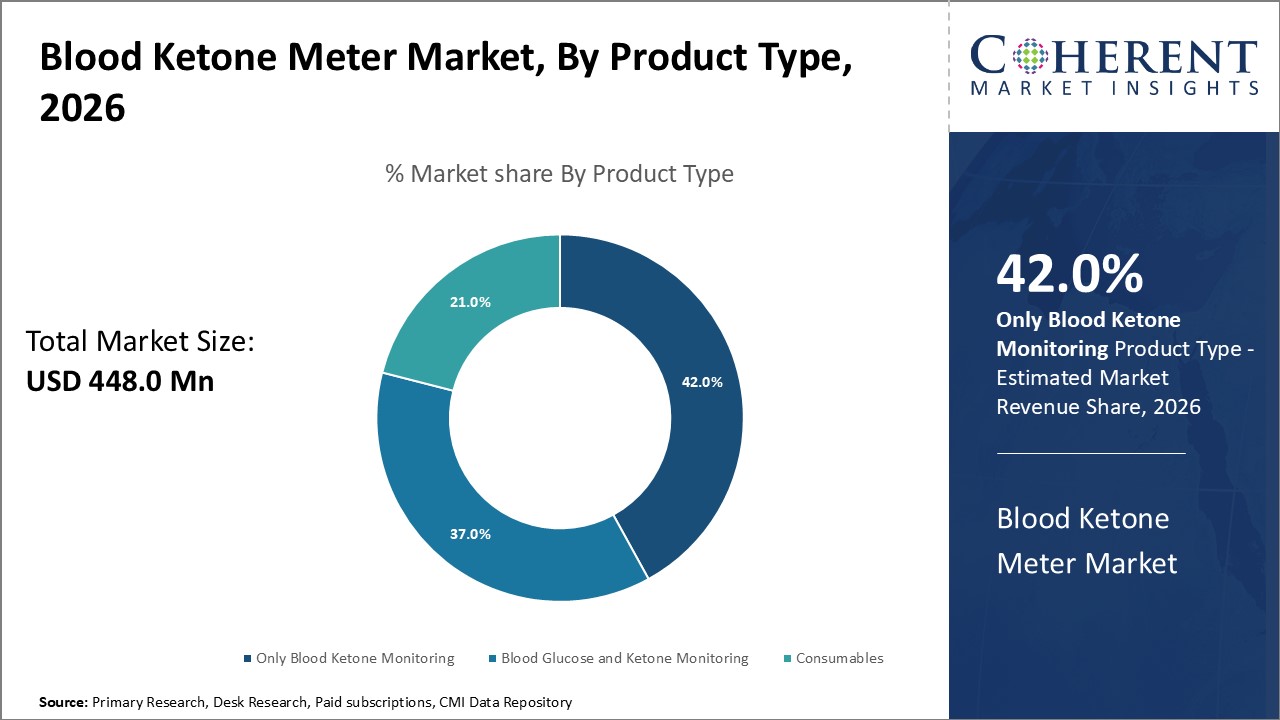
The blood ketone monitoring segment holds the largest revenue share in the global market due to its ability to detect the early generation of ketones and alert the onset of ketoacidosis, which can reduce the complications of diabetic ketoacidosis. The increasing incidence of diabetes-related complications is expected to drive the market demand worldwide. Additionally, the hospital segments are expected to exhibit the highest growth rate in the coming years. These devices are specifically designed for diabetic patients and are also widely used by dairy farmers and veterinarians to test ketones in cattle. Therefore, the growing adoption of blood glucose and ketone monitoring in both humans and animals is expected to gain popularity in the future. For instance, in September 2022, Novo Nordisk announced the upcoming availability of an unbranded biologic of Tresiba (insulin degludec) injection, a once-daily long-acting basal insulin indicated to control high blood sugar in patients 1 year of age and older with diabetes. The unbranded biologic, insulin degludec, was approved by the US Food and Drug Administration (FDA).
Figure 1. Global Blood Ketone Meter Market Share (%), by Product Type, 2026
To learn more about this report, Request Free Sample
Global Blood Ketone Meter Market – Drivers
Launch of new products by key market players to expand product portfolio
Increasing incidence of diabetes coupled with rising cases of diabetic ketoacidosis are expected to drive the blood ketone meter market growth. Market players are focused on launching new products in the market, which is expected to boost the growth of the global blood ketone meter market. For instance, in November 2022, Eli Lilly and Company , The Tempo Personalised Diabetes Management Platform, which includes the Tempo Smart Button, TempoSmart, compatible software, and TempoPen, a prefilled insulin pen, has started to be rolled out by the company to provide personalised assistance for adults with diabetes.
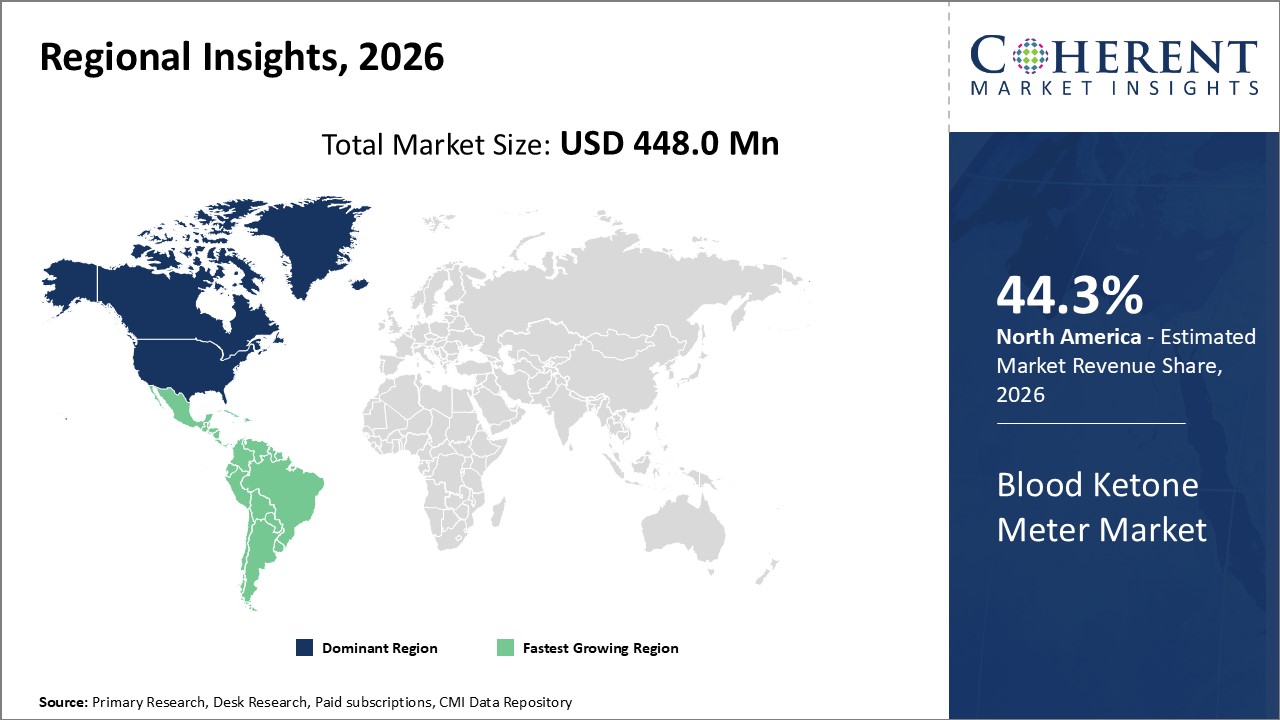
Figure 2. Global Blood Ketone Meter Market Value (US$ Million), by Region, 2023
To learn more about this report, Request Free Sample
Global Blood Ketone Meter Market- Regional Analysis
Among regions, North America is expected to be the dominant region in the blood ketone meter market, owing to the increasing launch of products. For instance, According to the National Diabetes Prevention Programme amended by the Centres for Disease Control in August 2021, the US government enhanced awareness programmes like the National Diabetes Prevention Programme to combat the rising burden of pre-diabetes and type 2 diabetes. Partnerships between public and private organisations were established as part of this effort in order to deliver affordable medicines and evidence-based treatments that help in the prevention of type 2 diabetes across the United States.
Its rapid spread had a significant impact on the global economy, social behavior, and healthcare systems. The virus affected production and demand, disrupted distribution channels, and had a financial impact on firms and markets. Generally speaking, when exposed to any virus, people with diabetes are more likely to experience more severe symptoms and problems. Infections with viruses can also cause internal edoema or inflammation in diabetics. Blood sugar levels that are above target can also cause this, and the inflammation that results could lead to more serious consequences.
Blood Ketone Meter Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 448.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.1% | 2033 Value Projection: | USD 724.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, ACON Laboratories Inc., Nipro Corporation, Keto-Mojo, PortaCheck Inc., Nova Biomedical, GlucoRx Limited, EKF Diagnostics Holdings Plc., Taidoc Technology Corporation, ForaCare Inc., Bruno MD, and Apex Biotechnology Corporation. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Blood Ketone Meter Market Segmentation:
The global blood ketone meter market report is segmented into by product type, by application, by end user, and by region.
By Product, the market is segmented into Blood Ketone Monitoring, Blood Glucose Ketone Monitoring, and Consumables. Out of which, the Consumables segment is expected to hold a dominant position in the global blood ketone meter market during the forecast period. For instances, in April 2026, Ascensia Diabetes Care, a global company dedicated to improving the lives of people with diabetes and helping empower launched the Eversense E3 CGM System for diabetes patients under 18 years of age continually measuring glucose levels for up to 180 days in persons with diabetes in the U.S. which was approved by the U.S. Food and Drug Administration (FDA). The Eversense E3 CGM System is intended to measure glucose levels in people 18 years and older who have diabetes. The sensor can be worn under the skin continually for up to 180 days. It is intended to replace the fingerstick blood glucose measurements that are traditionally used to help make treatment decisions for diabetes.
By End User, the market is segmented into Hospitals, Diagnostic Centers, Ambulatory Surgical Centers, Home Care Settings, Veterinary Hospitals, and Clinics. Among end User, the hospital segmented is expected to dominant over the forecast period.
Among Application, the Human segment is expected to grow in the blood ketone meter market over the forecast period. For instance, in December 2020, Xeris Pharmaceuticals, Inc., a specialty pharmaceutical company leveraging its novel formulation technology platforms to develop and commercialize ready-to-use injectable and infusible drug formulations, announced that it had entered into an exclusive distribution agreement with Megapharm Ltd., a leading Israel-based pharmaceutical company, for the commercialization in Israel and the Palestinian Authority of Xeris’ Gvoke (glucagon injection) for the treatment of severe hypoglycemia in adults and children with diabetes ages 2 years and above. Gvoke is the world’s first and only ready-to-use liquid glucagon.
Global Blood Ketone Meter Market Cross Sectional Analysis:
A cross-sectional study of the market for blood ketone metres revealed that in 2022, the market would be worth US$ 342.8 million.The market is anticipated to expand at a compound annual growth rate (CAGR) of 8.2% during the forecast period. This has led to market expansion. The hospital segment currently makes up the majority of the market, although the consumables segment is currently the largest section due to an increase in instances of insulin-dependent diabetes. The market segment for blood ketone monitoring holds the biggest proportion against the treatment of diabetes. The market size was estimated to be USD 342.8 million in 2022 and is anticipated to increase by 7.9% CAGR to reach US$ 585.6 million by 2030.
Global Blood Ketone Meter Market: Key Developments
For instance, in February 2023, Assure Tech, a high-tech biotechnology company specializing in research and development, production, and sale of diagnostic reagents, point-of-care testing, and biological materials launched a new platform for comprehensive health solutions for pets in China. This platform includes a glucose and blood ketone monitoring device that offers a novel technique for diagnosing blood ketone and glucose levels in pets.
In August 2021, Fitbit and LifeScan, a world leader in blood glucose monitoring serving more than 20 million people with diabetes, have teamed up to provide people with diabetes with a more thorough understanding of how lifestyle factors can affect the blood glucose levels. Life style factors like regular activity, diet, and sleep can affect blood glucose levels, as well as tools that can help lead to healthy lifestyle changes.
For instance, in February 14 2023, Insulet Corporation, is an innovative medical device company, announced it had acquired the assets of Automated Glucose Control LLC (AGC), a company in Palo Alto, California focused on developing and commercializing best in class automated insulin delivery technology.
Global Blood Ketone Meter Market: Key Trends
Better healthcare infrastructure, effective government policies, and Investment from key players in R&D can drive the growth of market.
Effective government policies
The blood ketone meter market is experiencing significant growth due to various factors, including the increasing support and initiatives from governments for glucose monitoring adoption. For example, campaigns introduced by organizations such as the American Medical Association, Centre of Disease Control, and ADA, the American Diabetes Association have helped raise awareness about pre-diabetes and encouraged people to undertake tests for diabetic conditions.
Better healthcare infrastructure
Additionally, the National Diabetes Awareness Month in November is aimed at promoting diabetes prevention and control. The market is also benefiting from technological advancements, such as the introduction of multi-functional devices and different body function monitoring devices.
Investment from key players in R&D can drive the growth of market:
Key players are investing in R&D activities to propose multi-functional devices and accelerate the proliferation rate of innovative healthcare diagnostic tools. For Instance, in May 2021 Roche Diabetes Care, a global leader for diabetes management systems and services announced that it has signed a collaboration agreement with Eli Lilly and Company, a global leader in diabetes care delivering innovative solutions including medicines and technologies, to work towards optimising the management of insulin pen therapy. By collaborating with Lilly, Roche follows its ambition to create an open ecosystem including its own and partner devices, solutions and services, across the continuum of care in diabetes
Global Blood Ketone Meter Market: Restraint
Inaccuracy in Blood Glucose Monitoring
Blood ketone meter sales are expected to be constrained by a growing number of issues, including technical flaws and inconsistent readings in ketone meters. As blood ketone meters are highly advanced technological equipment, they can experience technical problems, inconsistent findings, and other problems that could cause product recalls. For instance, in September 2022, KetoSens Test Strips batch UJ03PBXNG, manufactured by I-SENS, Inc., have been recalled because they give false readings.The test results showed that the patient had higher ketone levels than their actual levels. For instance, in February 2020, Abbott Laboratories, a medical devices and health care company recalls its Class 2 Device Precision Xtra Blood Glucose and ketone Monitoring system.
Global Blood Ketone Meter Market - Key Players
Major players operating in the blood ketone meter market include Abbott Laboratories, ACON Laboratories Inc., Keto-Mojo, EKF Diagnostics, Taidoc Technology Corporation, Apex Biotechnology Corporation, Bruno MD, Nova Biomedical, Nipro Corporation, ForaCare Inc., GlucoRx Limited, and PortaCheck Inc.
*Definition: Ketone testing is an essential part of type 1 diabetes treatment since it helps to prevent diabetic ketoacidosis, a short-term disease that can be lethal. Glucose provides the majority of the body's energy. The body begins to use lipids as fuel when its supply of glucose runs low. Thus, ketones are produced as a result. For insulin-dependent diabetics, measuring ketones is crucial, particularly when the body is not making enough insulin. Thus, as the need for ketone testing is increasingly acknowledged, there will be an increase in the market demand for blood ketone test meters. The Global Blood Ketone Meter Market is anticipated to develop as a result of rising diabetic ketoacidosis instances and diabetes incidence. Blood glucose and ketone monitoring play an important role in controlling and maintaining blood ketone levels. Therefore, the key players are investing in the R&D of the blood ketone meter market.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients