Boyulinum Toxin Market Size and Forecast – 2026 – 2033
The Global Botulinum Toxin Market size is estimated to be valued at USD 6.2 billion in 2026 and is expected to reach USD 11.8 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 9.2% from 2026 to 2033.
Global Boyulinum Toxin Market Overview
The botulinum toxin market is driven by the growing demand for minimally invasive cosmetic procedures and increasing applications in therapeutic treatments. Rising consumer preference for facial aesthetics, anti-aging treatments, and non-surgical procedures continues to boost product adoption globally. Expanding medical applications in chronic migraine, muscle spasticity, cervical dystonia, hyperhidrosis, and neurological disorders are further accelerating market growth. Increasing disposable income, growing medical tourism, and greater awareness of aesthetic treatments also contribute to rising demand. Additionally, continuous product innovations, regulatory approvals for new indications, and expanding dermatology and cosmetic clinic networks are strengthening the global botulinum toxin market across both cosmetic and therapeutic segments.
Key Takeaways
Therapeutic Use segment dominates the Botulinum Toxin market with over 53% share, driven by expanding clinical indications and increasing reimbursement opportunities.
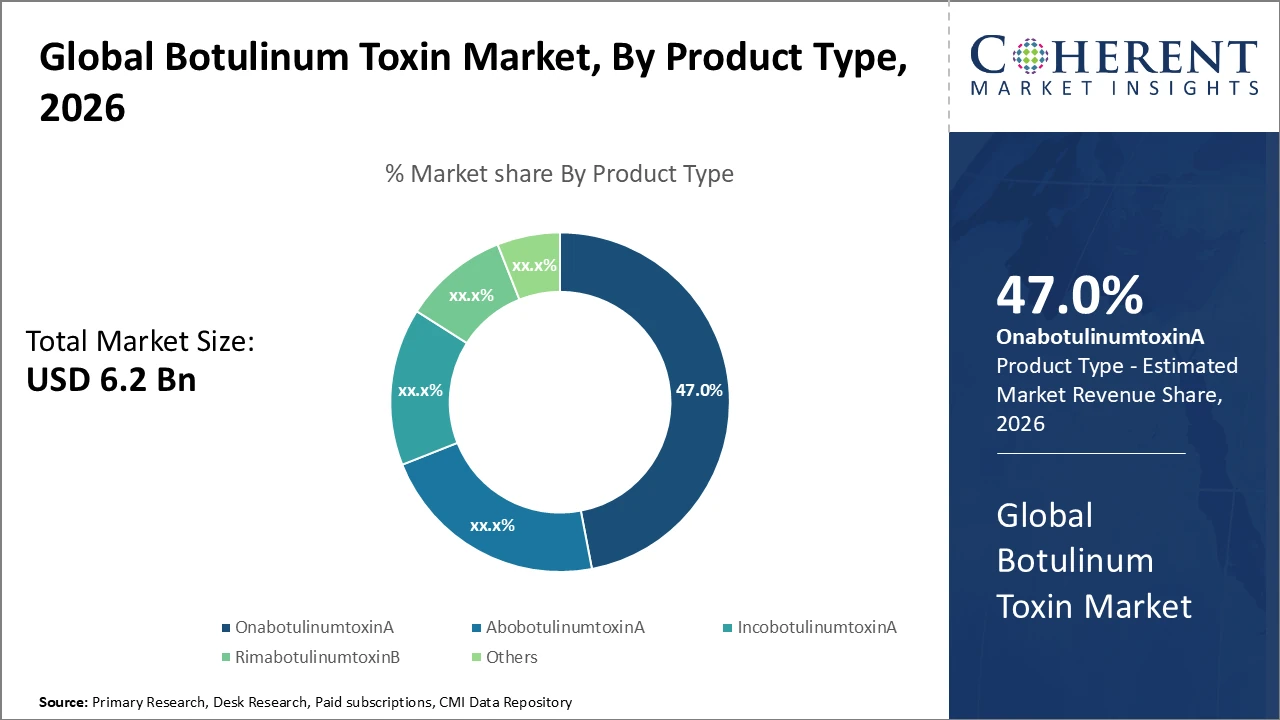
OnabotulinumtoxinA holds a leading position among product types, supported by established efficacy and extensive clinical data.
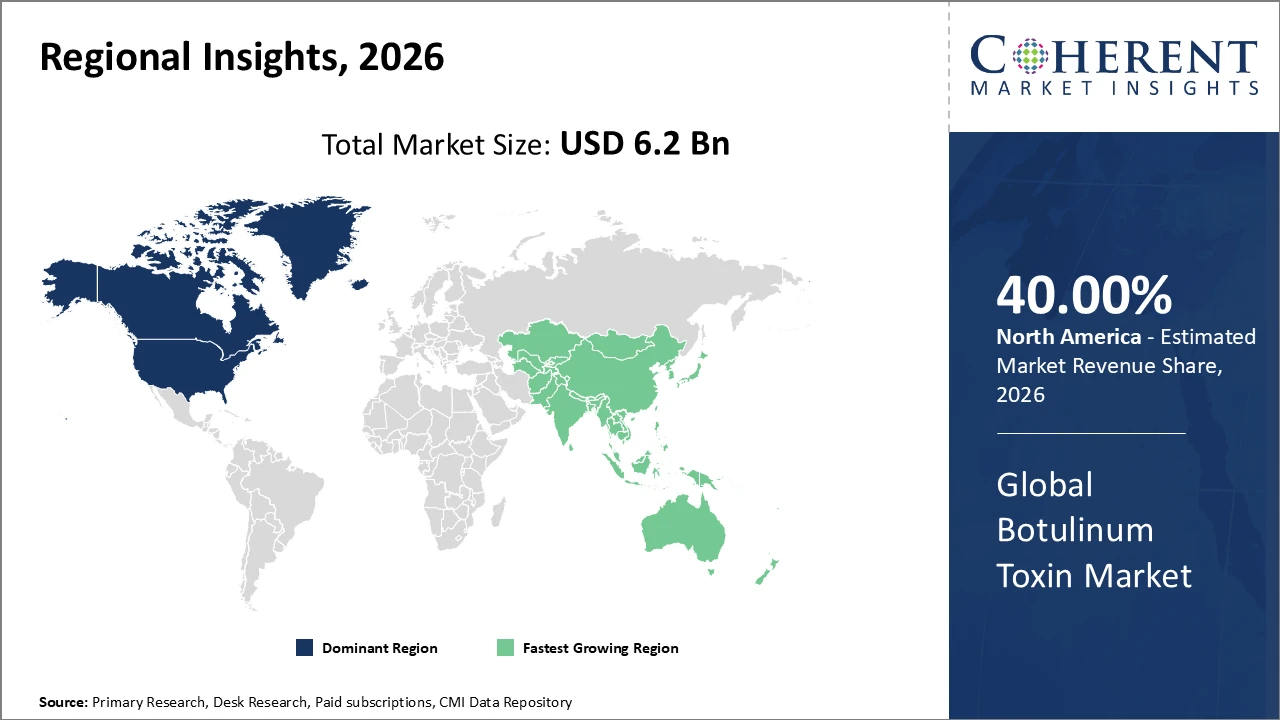
North America commands a market share above 40%, bolstered by advanced healthcare infrastructure and a regulatory environment that encourages innovation.
Asia Pacific exhibits the highest CAGR, fueled by growing adoption in emerging economies, rising aesthetic awareness, and local manufacturing capabilities that enhance market revenue potential.
Boyulinum Toxin Market Segmentation Analysis
To learn more about this report, Request Free Sample
Boyulinum Toxin Market Insights, By Product Type
OnabotulinumtoxinA leads the market with a 47% share, driven by its proven efficacy and extensive FDA approvals for both therapeutic and cosmetic uses. The fastest-growing subsegment, IncobotulinumtoxinA, benefits from a favorable safety profile and recent regulatory approvals for innovative neuromuscular disorders, broadening its clinical applications and market reach. AbobotulinumtoxinA maintains steady demand, particularly in aesthetic procedures, due to its cost-effectiveness. RimabotulinumtoxinB is primarily used for niche therapeutic indications such as cervical dystonia. The ‘Others’ category includes emerging biosimilars and new molecular variants, reflecting ongoing innovation and potential future growth opportunities in the botulinum toxin market.
Boyulinum Toxin Market Insights, By End User
Hospitals lead the Botulinum Toxin market with a 42% share, driven by extensive treatment capabilities, large patient volumes, and infrastructure supporting clinical trials that facilitate therapeutic application scaling. Clinics represent the fastest-growing segment, fueled by the rise of specialized aesthetic and dermatology centers offering cosmetic toxin procedures, reflecting demographic trends favoring outpatient care. Specialty centers target niche therapeutic applications, including neurological and muscular disorders, while Home Care Settings are emerging as convenient channels for patient-administered treatments, supported by advancements in self-administration technologies. These dynamics highlight a diversified end-user landscape, balancing established institutional demand with rapidly expanding outpatient and home-based adoption.
Boyulinum Toxin Market Insights, By Application
Therapeutic Use dominates the Botulinum Toxin market with a 53% share, driven by expanding indications such as chronic migraine, muscle spasticity, and hyperhidrosis. Growth is supported by broader insurance reimbursements and incorporation into clinical guidelines, enhancing revenue penetration. Cosmetic Use is the fastest-growing segment, fueled by rising aesthetic awareness, particularly among individuals aged 30-50 seeking minimally invasive treatments for wrinkle reduction and facial rejuvenation. Research Applications, though smaller in market size, show strong future potential, with increasing clinical trials investigating novel toxin applications, innovative delivery methods, and advanced formulations. These trends indicate continued diversification and long-term growth opportunities across multiple segments.
Boyulinum Toxin Market Trends
The Botulinum Toxin market is undergoing transformation driven by formulation innovations and geographic diversification.
Longer-acting toxin variants are reshaping treatment protocols, exemplified by the 2026 FDA approval of extended-duration formulations that enhance patient compliance.
Integration with digital health platforms for personalized treatment monitoring is an emerging trend, supporting better outcomes and driving market growth.
Strategic collaborations between pharmaceutical and medical device companies are accelerating innovation pipelines, enabling the development of advanced therapies that meet evolving end-user needs.
These trends collectively contribute to broader adoption and expansion of the botulinum toxin market globally.
Boyulinum Toxin Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Boyulinum Toxin Market Analysis and Trends
In North America, the Botulinum Toxin market is dominated by advanced healthcare infrastructure, high adoption of innovative therapies, and supportive regulatory frameworks. Leading companies, including AbbVie and Revance, have strengthened market revenue through targeted product innovations, enhanced therapeutic applications, and strategic collaborations with healthcare providers. These initiatives have reinforced trends toward both therapeutic diversification and aesthetic procedure growth. Strong clinical adoption, combined with favorable reimbursement policies, ensures North America remains a key and influential region in the global botulinum toxin market.
Asia Pacific Boyulinum Toxin Market Analysis and Trends
The Asia Pacific region is experiencing the fastest growth in the Botulinum Toxin market, with a CAGR exceeding 11%, driven by expanding healthcare access, increasing demand for aesthetic procedures, and a growing middle-class population. China and India lead the region, supported by government initiatives aimed at enhancing healthcare infrastructure and the development of local manufacturing hubs, which reduce costs and broaden market reach. Companies such as Medytox and Hugel have leveraged these opportunities by scaling local production and customizing market strategies to regional preferences. These factors collectively position Asia Pacific as a key growth engine in the global botulinum toxin market.
Boyulinum Toxin Market Outlook for Key Countries
USA Boyulinum Toxin Market Analysis and Trends
The USA continues to lead the global Botulinum Toxin market, supported by a robust ecosystem of established pharmaceutical companies, stringent regulatory frameworks, and comprehensive insurance coverage. In 2025, the launch of advanced toxin formulations with extended duration expanded therapeutic applications, driving market share growth. Key players actively invest in clinical trials, product innovation, and direct-to-consumer marketing, reinforcing business development and revenue generation. A strong network of cosmetic clinics and hospitals provides a substantial end-user base, supporting both aesthetic and therapeutic adoption. These factors collectively ensure sustained market expansion and maintain the U.S. as a dominant force in the global botulinum toxin market.
Germany Boyulinum Toxin Market Analysis and Trends
Germany holds a significant position in the European Botulinum Toxin market, driven by advanced healthcare infrastructure, high clinical adoption, and strong regulatory support. Hospitals and specialized clinics increasingly implement botulinum toxin treatments for therapeutic applications such as chronic migraine, spasticity, and hyperhidrosis, alongside growing demand in aesthetic procedures. The market benefits from reimbursement frameworks, well-established distribution networks, and strategic partnerships between global manufacturers and local providers. Emerging trends include longer-acting formulations, precision delivery systems, and integration with digital health platforms for personalized monitoring. Continuous clinical research and innovation position Germany as a key growth market within Europe’s botulinum toxin sector.
Analyst Opinion
Pricing Dynamics and Market Revenue Growth: Leading players use tiered pricing strategies to serve both premium and cost-sensitive segments. In 2024, a manufacturer’s volume-based pricing increased revenue by 14%, highlighting the role of pricing flexibility in expanding market share and improving treatment accessibility in emerging regions.
Production Capacity and Supply Chain Resilience: Manufacturing expansions in North America and Asia-Pacific enhance scalable production. In 2025, a major hub increased output by over 25%, addressing global supply bottlenecks and ensuring rapid fulfillment for therapeutic and cosmetic demand, supporting long-term market growth.
Therapeutic Applications and Industry Trends: The market is shifting from cosmetic-only uses to therapeutic indications like chronic migraines, hyperhidrosis, and muscular spasticity. In 2023, off-label uses rose 12%, especially in neurology and dermatology, driving segment diversification and revenue growth.
Regulatory Landscape and Market Access: Evolving approvals in the USA, Europe, and Japan accelerate product launches and foster competitive dynamics. A 2026 milestone approval of an extended-duration formulation enhanced market share, enabling innovation while maintaining regulatory compliance.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.2 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.2% | 2033 Value Projection: | USD 11.8 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Ipsen Pharma, AbbVie Inc., Hugel, Inc., Siax, Inc., Medytox, Inc., HKBK Group, Medisave Pharma, Evolus, Inc., Huadong Medicine, Revance Therapeutics, Inc. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Boyulinum Toxin Market Growth Factors
The expanding therapeutic indications beyond traditional cosmetic applications remain the primary driver of the Botulinum Toxin market, contributing to significant demand growth, particularly in neurology and dermatology. Advancements in formulation technology, including longer-lasting effects and improved safety profiles, have been reinforced by multiple FDA approvals between 2024 and 2026, supporting sustained market expansion. The rising adoption of minimally invasive aesthetic procedures further fuels growth, especially in regions with increasing disposable incomes and aging populations. Additionally, enhanced healthcare infrastructure and evolving regulatory approvals in emerging economies have improved market accessibility, creating attractive opportunities for companies focused on regional and global expansion.
Boyulinum Toxin Market Development
Hugel Inc. has launched its botulinum toxin product, Letybo, in Malaysia, strengthening its presence in the rapidly growing Southeast Asian medical aesthetics market.
Key Players
Leading Companies of the Market
Ipsen Pharma
AbbVie Inc.
Hugel, Inc.
Siax, Inc.
Medytox, Inc.
HKBK Group
Medisave Pharma
Evolus Inc.
Huadong Medicine
Revance Therapeutics, Inc.
Ipsen Pharma strengthened its market position in 2025 through strategic alliances with biotech firms, enabling the launch of innovative therapeutic formulations and driving a 19% increase in market share within the therapeutic segment. AbbVie focused on vertical integration by acquiring manufacturing sites in 2024, enhancing production efficiency by 22%, reducing operational costs, and supporting aggressive market penetration. Meanwhile, Revance Therapeutics pursued targeted geographic expansion, particularly in the Asia-Pacific region, resulting in a 15% growth in international market revenue in 2025. These competitive strategies underscore the emphasis on innovation, operational efficiency, and regional diversification to capture market opportunities.
Boyulinum Toxin Market Future Outlook
The future of the Botulinum Toxin market is poised for significant growth, driven by rising demand in both therapeutic and aesthetic applications. Increasing prevalence of chronic conditions such as migraines, spasticity, and hyperhidrosis supports clinical adoption, while growing interest in minimally invasive cosmetic procedures fuels the aesthetic segment. Technological advancements, including longer-acting formulations, precision delivery systems, and combination therapies, are enhancing efficacy and patient convenience. Expanding access in emerging markets, coupled with rising healthcare expenditure and awareness, is expected to accelerate adoption. Strategic partnerships, product innovations, and regulatory approvals will further strengthen market expansion over the next decade.
Boyulinum Toxin Market Historical Analysis
The Botulinum Toxin market has experienced steady growth over the past decade, driven by both therapeutic and cosmetic applications. Initially dominated by aesthetic uses such as wrinkle reduction, the market expanded into medical indications including chronic migraines, spasticity, hyperhidrosis, and cervical dystonia. Advancements in formulation, dosing precision, and delivery techniques enhanced safety and efficacy, boosting clinical adoption. Leading companies invested heavily in research, strategic partnerships, and regional expansion to strengthen market presence. Growing awareness, increased disposable income, and broader insurance coverage in developed markets also contributed to historical growth, while emerging markets gradually adopted these therapies, laying the foundation for future expansion.
Sources
Primary Research Interviews:
Dermatologists, neurologists, and pain management specialists administering botulinum toxin for aesthetic and therapeutic purposes.
Hospital administrators, procurement managers, and supply chain professionals involved in sourcing botulinum toxin products.
Product managers, R&D specialists, and regulatory experts from companies developing botulinum toxin formulations and delivery systems.
Magazines:
Medical Device & Diagnostic Industry (MD+DI) – Coverage of product innovations, market trends, and technological developments in therapeutic and aesthetic devices.
Aesthetic Surgery Journal – Insights on cosmetic procedures and botulinum toxin applications in dermatology and aesthetics.
Pharmaceutical Technology – Focus on formulation, delivery systems, and manufacturing trends in biologics and therapeutic toxins.
Journals:
Journal of Cosmetic Dermatology – Clinical studies and research on aesthetic uses of botulinum toxin.
Neurology – Research on therapeutic applications, including migraines, spasticity, and neuromuscular disorders.
Toxins – Scientific studies on botulinum toxin biology, safety, and efficacy in medical and cosmetic applications.
Newspapers:
The Financial Times – Coverage of global pharmaceutical trends, mergers, and industry investments.
Reuters – Updates on approvals, clinical trials, and product launches in the botulinum toxin market.
Bloomberg – Market performance, corporate strategies, and developments in the pharmaceutical and aesthetics sector.
Associations:
American Academy of Dermatology (AAD) – Guidelines, research, and clinical standards for cosmetic dermatology procedures.
American Academy of Neurology (AAN) – Standards and research for therapeutic botulinum toxin applications.
International Organization for Standardization (ISO) – Standards for biologics manufacturing, safety, and quality compliance.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients