Cancer Supportive Care Drugs Market is estimated to be valued at USD 37,324.1 Mn in 2026 and is expected to reach USD 52,169.7 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of4.9% from 2026 to 2033.
The rising prevalence of cancer and the growing focus on improving patient quality of life are rapidly expanding the Cancer Supportive Care Drugs Market. Advances in therapies and increased awareness of managing side effects from cancer treatments are driving the demand for supportive care drugs. Hospitals and healthcare providers play a central role in delivering these medications, backed by strong research and development efforts. Moreover, growing investments and innovative drug formulations are actively enhancing treatment outcomes, making supportive care a vital part of comprehensive cancer management worldwide.
|
Current Events |
Description and its impact |
|
Regulatory Changes and Healthcare Policy Reforms
|
|
|
Public Health Trends
|
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Nonsteroidal Anti – Inflammatory Drugs hold the largest market share of 28.9% in 2026. Healthcare providers are pushing for Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) in cancer supportive care because they are good at treating pain and swelling induced by cancer and its treatments. As people worry more about using opioids, NSAIDs are becoming more popular as safer options. New ideas have made NSAID formulations better, which has made them more useful. Ongoing study focuses on their possible chemopreventive benefits. Adding NSAIDs to holistic cancer care also makes them more useful as supportive therapy.
For instance, in January 2024, GC Biopharma announced that it launched a 30-tablet version of its over-the-counter NSAID, Tak-Sen soft cap, used to relieve inflammation and common pain like headaches and muscle aches.
To learn more about this report, Request Free Sample
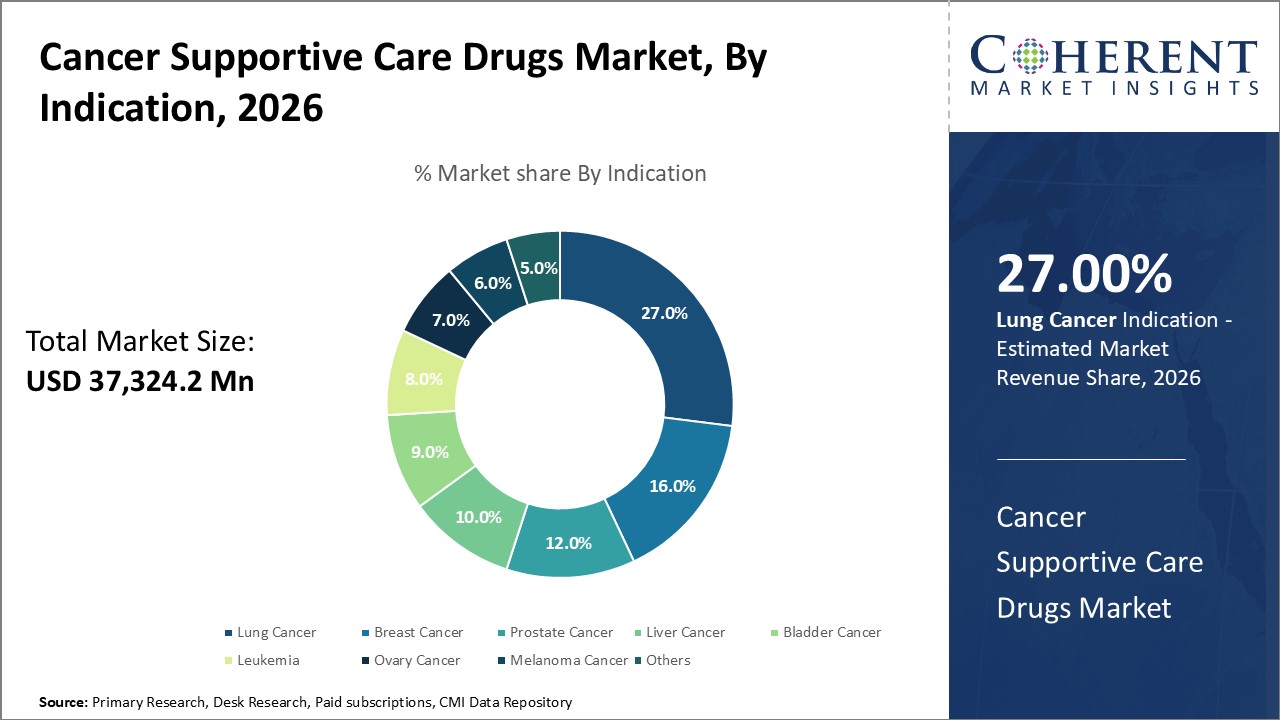
Lung cancer is a major player in the cancer supportive care medications industry since it has high rates of incidence and death. New targeted medicines and immunotherapy drugs have made more treatment options available, which has led to a growing need for supportive care to help with adverse effects. This expansion is also driven by the creation of tailored treatment plans and chemotherapy-free regimens. Also, an older population and more knowledge of the need of early detection are both making the demand for appropriate supportive care for lung cancer even greater.
For instance, in June 2025, Glenmark Pharmaceuticals Ltd. launched the lung cancer drug Tevimbra in India on Tuesday, after receiving approval from the Central Drugs Standard Control Organisation.
Hospital pharmacies make sure that people can get important medicines quickly, which is a very important part of the cancer supportive care drugs industry. They provide a consistent supply of drugs and manage the inventory well, which keeps treatment from being interrupted. Skilled pharmacists work closely with oncology teams to make sure that each patient gets the right medications and follows clinical recommendations. Additionally, hospitals use cutting-edge technologies to improve the safe delivery of supportive care medications, which is why hospital pharmacies are so important for providing full cancer care.
For instance, in April 2025, Sahyadri Hospital advanced cancer care by launching a holistic treatment approach that integrates advanced technology with patient-centered support. Such innovations are accelerating the cancer supportive care drugs market revenue.
To learn more about this report, Request Free Sample
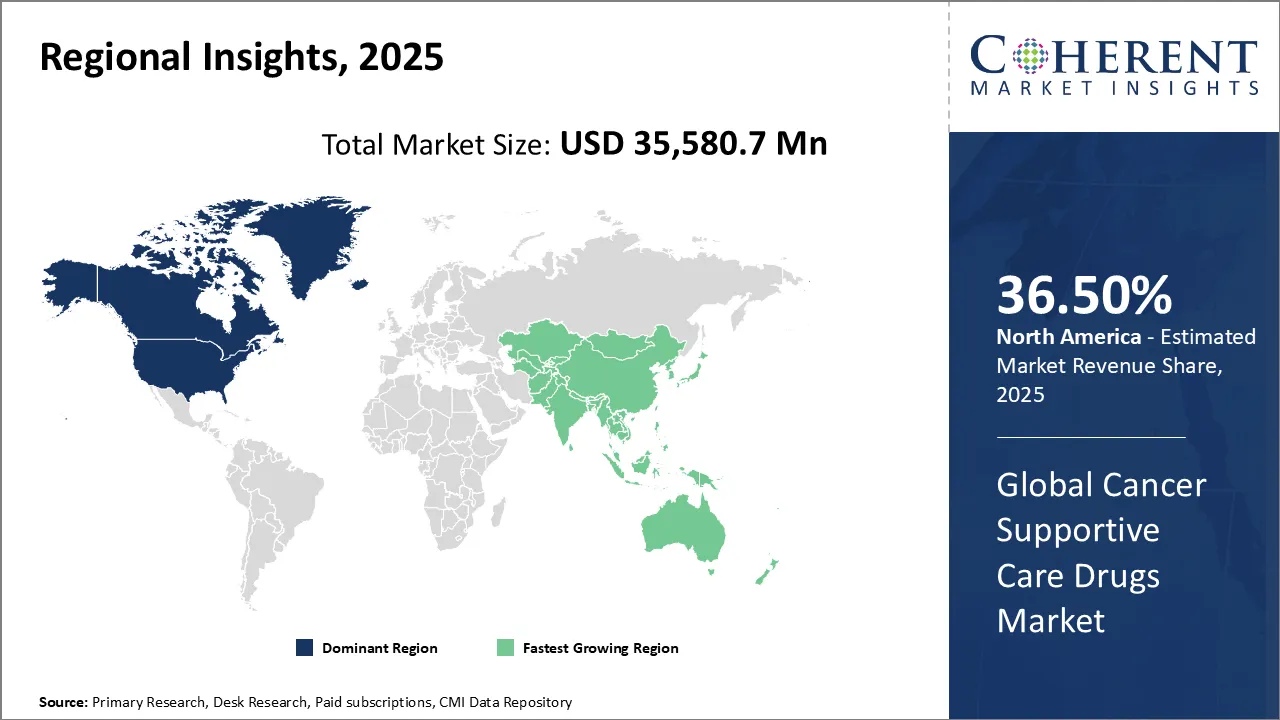
North America dominates the overall cancer supportive care drugs market revenue with an estimated share of 36.5% in 2026. Advanced drug development and increased adoption of biosimilars drive North America’s leadership in the cancer supportive care drugs market. The region’s strong healthcare infrastructure and substantial investments in oncology research support this growth. Growing awareness of managing treatment side effects and government initiatives help patients access supportive care therapies. Additionally, healthcare providers’ shift toward personalized medicine and value-based care models fuels demand for innovative, cost-effective supportive care drugs, strengthening North America’s role as a key player in comprehensive cancer management.
For instance, in April 2024, Accord BioPharma, the U.S. specialty division of Intas Pharmaceuticals, announced FDA approval of HERCESSI™ (trastuzumab-strf), a biosimilar to Herceptin®, to treat HER2-positive breast and gastric cancers.
Increasing cancer incidence and improving healthcare infrastructure are driving rapid growth in the Asia Pacific cancer supportive care drugs market demand. Governments are expanding initiatives, and rising awareness is helping patients access these medications more easily. The region is adopting advanced therapies and biosimilars at a growing pace, supported by continuous investments in research and development. Economic growth and broader healthcare coverage are also boosting demand for effective supportive care drugs, establishing Asia Pacific as a key emerging market in cancer management. For instance, in July 2026, Six Southeast Asian countries launched Project ACTIVE 2.0 in Malaysia to improve cervical cancer treatment and support patient recovery across the region.
Advancements in innovative therapies and a strong focus on personalized medicine shape the United States cancer supportive care drugs market. Healthcare providers integrate supportive care early in treatment to improve patient outcomes and quality of life. Increased funding for oncology research and favorable reimbursement policies drive market growth. Additionally, the growing use of biosimilars and adoption of value-based care models promote cost-effective treatment options, strengthening the country’s leadership in developing and adopting supportive care drugs. For instance, in March 2026, Shorla Oncology, a U.S.-Ireland specialty pharmaceutical company, launched dedicated sales and market access teams and made IMKELDI, the first oral liquid form of imatinib for certain leukemias and cancers, commercially available.
Rising cancer cases and expanding healthcare access are steadily growing India’s cancer supportive care drugs market. Governments are increasing initiatives and awareness programs to improve patient education and access to supportive therapies. Growing investments in healthcare infrastructure and pharmaceutical manufacturing strengthen the market. Additionally, healthcare providers adopt affordable biosimilars and generic drugs to ensure wider availability. Efforts to promote early diagnosis and integrate supportive care into treatment protocols actively boost demand, establishing India as an emerging force in the cancer supportive care landscape. For instance, in June 2026, Glenmark Pharmaceuticals Ltd. received DCGI approval to launch BRUKINSA® (zanubrutinib) in India, making it the country's first BTK inhibitor approved for five B-cell cancers and advancing oncology care.
The cancer supportive care drugs market is witnessing a significant shift toward biosimilars and generic drugs as healthcare systems strive to contain rising costs. These alternatives provide comparable efficacy and safety to branded drugs but at a fraction of the price, making supportive care more affordable and accessible globally. Additionally, improved regulatory frameworks and growing physician confidence in biosimilars further accelerate their uptake. As a result, biosimilars and generics are playing a pivotal role in expanding treatment options and improving adherence in cancer supportive care.
Recent advances in targeted supportive therapies focus on mitigating specific side effects of cancer treatments such as chemotherapy-induced nausea, pain, and neutropenia. These therapies use precise mechanisms to address symptoms without interfering with the primary cancer treatment, leading to better patient tolerance and fewer complications. The development of novel drug delivery systems and combination therapies enhances treatment effectiveness and reduces toxicity. Consequently, targeted supportive therapies are becoming integral to comprehensive cancer care, driving demand for specialized supportive drugs.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 37,324.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.9% | 2033 Value Projection: | USD 52,169.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Amgen Inc., Johnson & Johnson Private Ltd., Merck, Novartis AG, Baxter International, Hoffmann-La Roche Ltd., Fagron, Teva Pharmaceutical Industries Ltd., G1 Therapeutics Inc., APR Applied Pharma Research s.a., Acacia Pharma, EMD Serono, and Kyowa Hakko Kirin Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients