Cardiac Pacemaker Market is estimated to be valued at USD 5,431.4 Mn in 2026 and is expected to reach USD 7,796.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
To learn more about this report, Request Free Sample
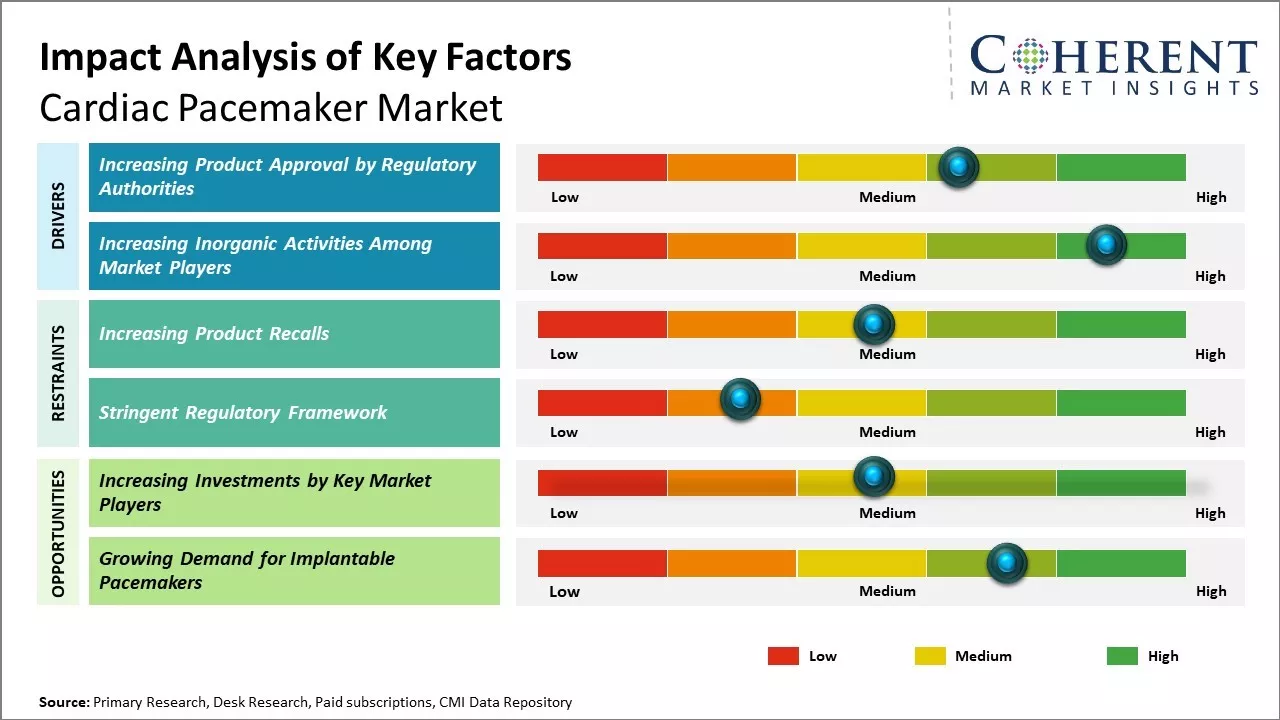
The cardiac pacemaker market Size is steadily expanding as a result of aging populations, rising cardiovascular disease rates, and continuous technological developments like leadless pacemakers and MRI compatibility. Cardiac pacemaker market growth is driven by rising geriatric population worldwide and the subsequent increase in the prevalence of cardiovascular and heart diseases. However, the availability of alternative therapies for cardiac arrhythmia monitoring device and the risks and complications associated with pacemaker implants may hamper the cardiac pacemaker market growth during the forecast period.
For instance, in November 2024, Abbott, the global healthcare company announced the launch of AVEIR VR single-chamber ventricular leadless pacemaker for the treatment of patients in India with slow heart rhythms.
|
Event |
Description and Impact |
|
Regulatory clearances for leadless, miniature pacemakers |
|
|
Surge in product recalls and device malfunctions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Product Type, the Single-chamber pacemaker segment is estimated to hold 46.3% of the market share in 2026, owing to their affordable price point. Single-chamber pacemakers offer a more basic yet effective solution at a fraction of the cost of dual-chamber or biventricular models, thus, making them desirable for price-sensitive customers.
Their simple design with only one lead wire also reduces manufacturing complexities and costs. Given the large population without health coverage, single-chamber pacemakers remain the go-to option for those seeking an affordable treatment option. This has ensured their continued dominance in the global cardiac pacemaker market over the years.
To learn more about this report, Request Free Sample
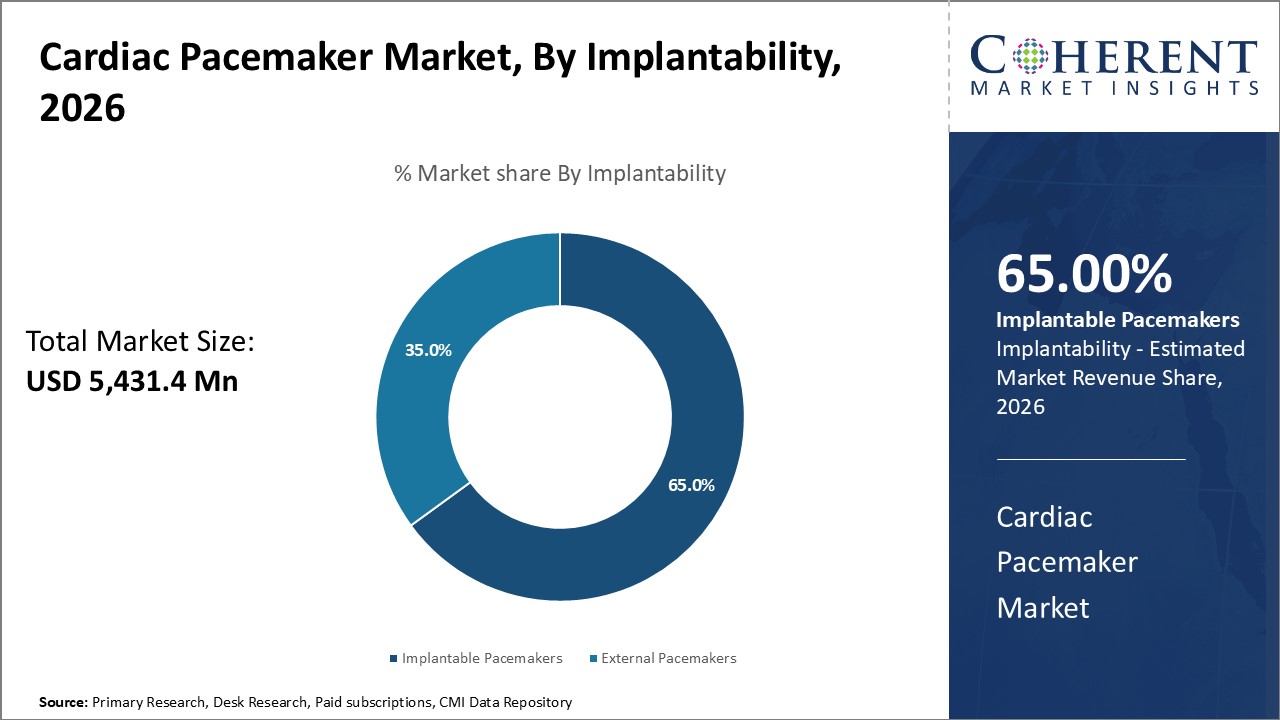
Implantability segment is sub-segmented into implantable pacemakers and external pacemakers. Implantable pacemakers’ segment is estimated to hold 65% of the market share in 2026, owing to their enhanced clinical suitability and acceptance. Unlike external pacemakers used temporarily, implantable devices offer permanence once surgically placed inside the body. This allows for superior management of cardiac arrhythmias without restrictions on patient movement or lifestyle.
Implantable cardiac pacemakers are also more seamlessly integrated with the body's physiology. Advancements in miniaturization have increased comfort levels while reducing risks of inflection and rejection. Their long-term reliability negates the need for frequent replacement. Widespread physician recommendation and training have boosted user familiarity and preference for implantable pacemakers over other options. This has established them as the gold standard treatment in most healthcare settings.
Hospitals segment is estimated to hold 34.6% of the market share in 2026 as most patients prefer dedicated cardiac facilities for pacemaker implantation and management. Hospitals employ skilled cardiac specialists, equip themselves with advanced diagnostic technologies and operate high-level intensive care units required for pacemaker procedures.
These also offer post-operative care and monitoring at convenient locations. This provides patients assurance of comprehensive care. Many countries also lack widespread access to cardiac clinics or surgery centers in rural areas. Hospitals remain the default option for a majority, driving their sales volumes. Governments and insurance providers also prefer recognized hospital infrastructures for reimbursing cardiac device costs.
To learn more about this report, Request Free Sample
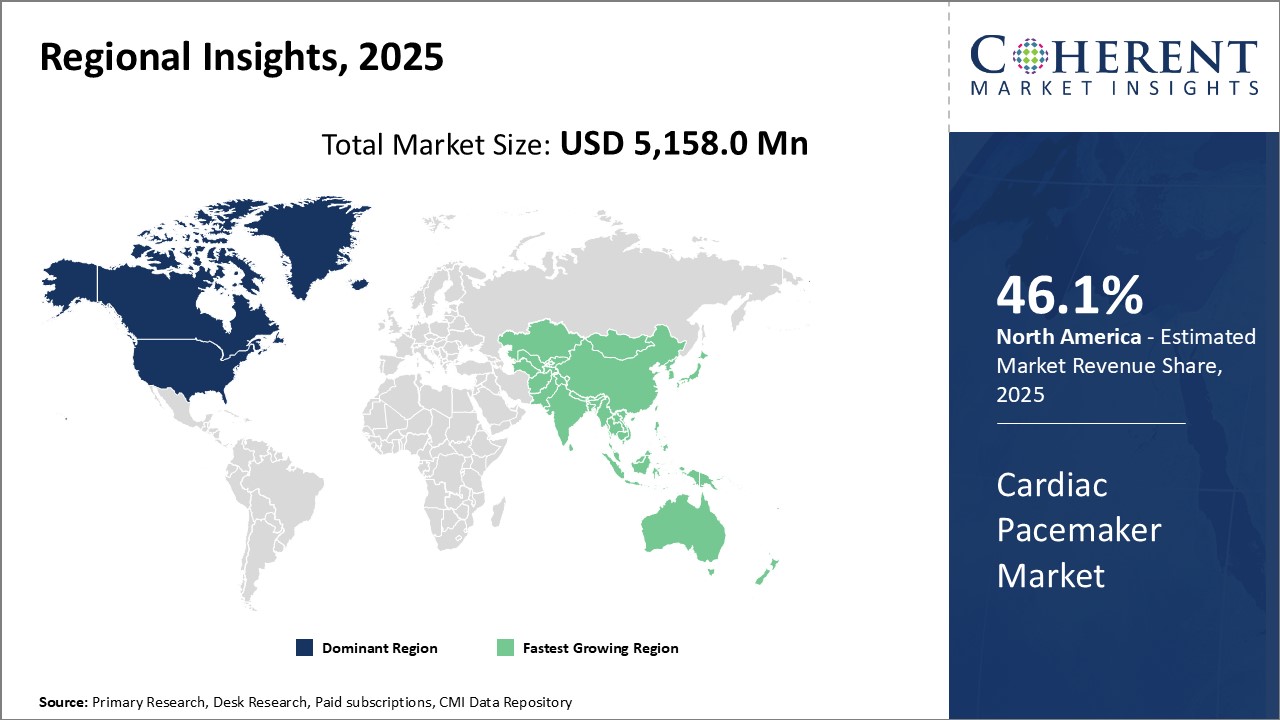
North America remains the dominant region in the global cardiac pacemaker market and is estimated to hold 46.1% of the market share in 2026. The U.S. leads the regional market and accounts for the major share due to the strong presence of leading pacemaker manufacturers in the country. Factors such as rising geriatric population suffering from cardiovascular diseases, favorable reimbursement policies, and higher healthcare spending have boosted the sales of pacemakers in the US.
The region is an early adopter of advanced devices due to the high acceptance of new technologies among physicians and patients. Leading global players such as Medtronic, Abbott, and Boston Scientific have their headquarters in the U.S., allowing them to cater to the regional demands efficiently and maintain their stronghold.
Asia Pacific has shown potential to become the fastest-growing region in the global market. China dominates the Asia Pacific cardiac pacemaker market due to rising incidence of cardiovascular diseases and growing healthcare expenditure. Moreover, the presence of a large patient pool makes the country highly lucrative for pacemaker manufacturers. With improving medical infrastructure and affordability, the adoption of pacemakers is increasing at a rapid pace in China.
The U.S. benefits from advanced healthcare infrastructure, early adoption of cutting-edge technologies such as leadless and MRI-compatible pacemakers, and favorable reimbursement policies that improve patient access to these devices.
India is emerging as a significant market for cardiac pacemakers within the Asia Pacific region, propelled by a large and growing patient population suffering from cardiovascular diseases. Factors such as increasing healthcare awareness, rising disposable incomes, and improving access to advanced medical technologies are contributing to market expansion.
China dominates the Asia Pacific cardiac pacemaker market, driven by its vast population, escalating incidence of cardiovascular diseases, and rising healthcare expenditure. The country’s rapid urbanization and lifestyle changes have contributed to a higher prevalence of heart rhythm disorders, making it a lucrative market for pacemaker manufacturers.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,431.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 7,796.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic, Boston Scientific Corporation, Abbott, BIOTRONIK SE & Co. KG, Pacetronix , Lepu Medical Technology (Beijing)Co., Ltd., LIVANOVA PLC, OSCOR Inc., MEDICO S.R.L., MicroPort Scientific Corporation, OSYPKA MEDICAL, Vitatron Holding B.V. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasing product approval by regulatory authorities is expected to drive the global cardiac pacemaker market growth over the forecast period. For instance, in April 2025, Medtronic has received FDA approval for its OmniaSecure™ defibrillation lead, the world’s smallest-diameter lumenless lead at 4.7 French. Designed for right ventricular placement, it aims to reduce complications. Clinical trials also showed 100% defibrillation success when implanted in the left bundle branch area, highlighting its potential for physiologic pacing.
Increasing inorganic activities such as agreements, partnerships, and collaborations among market players is expected to drive the global cardiac pacemaker market growth over the forecast period.
Pacemakers with remote monitoring capabilities allow healthcare professionals to track device performance and patient heart function in real time, often via smartphone apps. This trend supports better patient outcomes and reduces the need for frequent in-person check-ups.
Increasing adoption of inorganic growth strategies such as investments is expected to drive growth of the global cardiac pacemaker market. For instance, in July 2025, Ligand Pharmaceuticals and Medtronic have jointly committed $70 million in strategic capital to support Orchestra BioMed’s growth. The investment aims to accelerate development of innovative therapies, including the BackBeat Cardiac Neuromodulation Therapy for hypertension. This collaboration strengthens Orchestra BioMed’s financial position and advances its mission to deliver high-impact cardiovascular solutions.
*Definition: A cardiac pacemaker is a small-sized device that is implanted in the chest to control the slow heartbeat. The cardiac pacemaker implantation is carried out with a surgical procedure. There are three types of cardiac pacemakers such as single-chamber pacemaker, dual-chamber pacemaker, and biventricular pacemaker.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients