Chronic Idiopathic Constipation (CIC) Treatment Market Size and Forecast – 2026 – 2033
The Global Chronic Idiopathic Constipation Treatment Market size is estimated to be valued at USD 3.8 billion in 2026 and is expected to reach USD 6.5 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 7.5% from 2026 to 2033.
Global Chronic Idiopathic Constipation (CIC) Treatment Market Overview
The Chronic Idiopathic Constipation (CIC) treatment market is driven by the rising global prevalence of gastrointestinal disorders linked to sedentary lifestyles, poor dietary habits, and increasing stress levels. Growing awareness of digestive health and expanding access to healthcare services are encouraging more patients to seek diagnosis and treatment. The aging population, which is more prone to chronic constipation, further supports market growth. Advancements in pharmacological therapies, including prosecretory agents and newer targeted drugs, are improving treatment outcomes. Additionally, ongoing clinical research, increasing over-the-counter product availability, and rising demand for safer, long-term management options are contributing to market expansion globally.
Key Takeaways
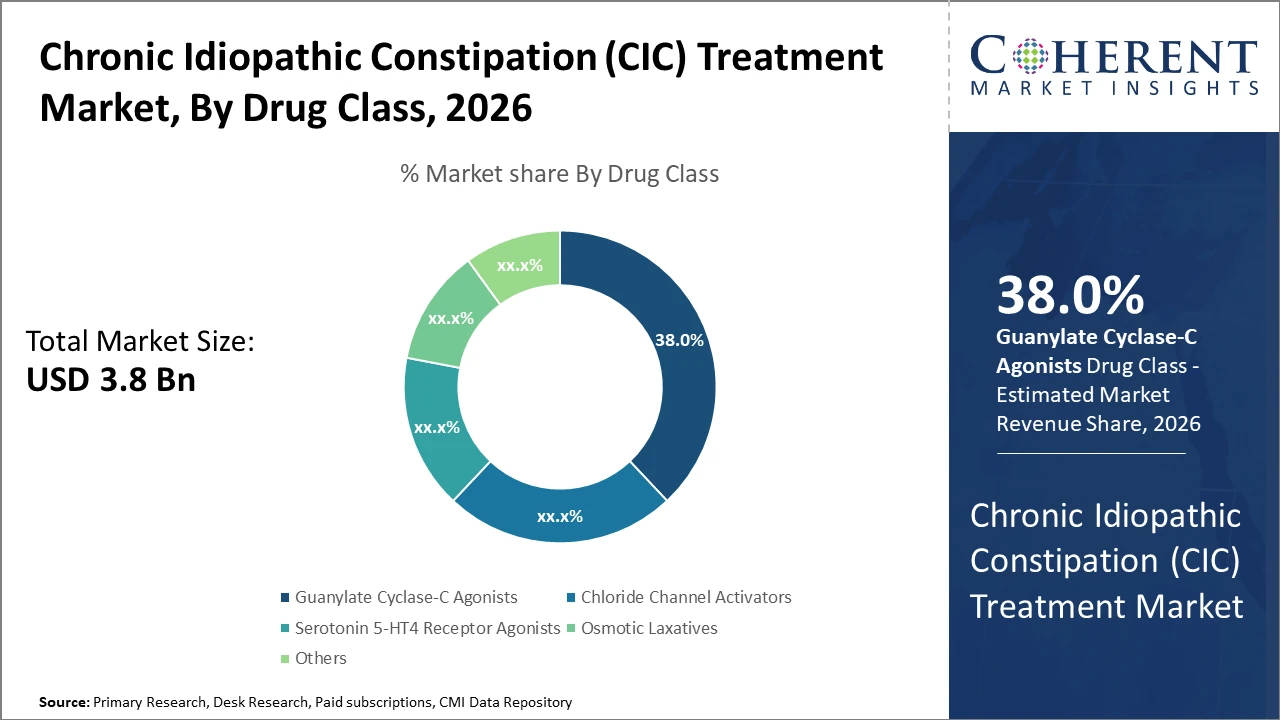
Guanylate Cyclase-C agonists dominate the CIC treatment market with around 38% share, driven by high efficacy, safety, and strong clinical adoption of drugs like linaclotide and plecanatide.
Oral administration remains the leading route with approximately 82% share, supported by ease of use, better patient compliance, and advancements in oral drug formulations.
Specialty clinics are experiencing the fastest growth, reflecting a shift toward specialist-driven diagnosis and personalized gastrointestinal disorder management.
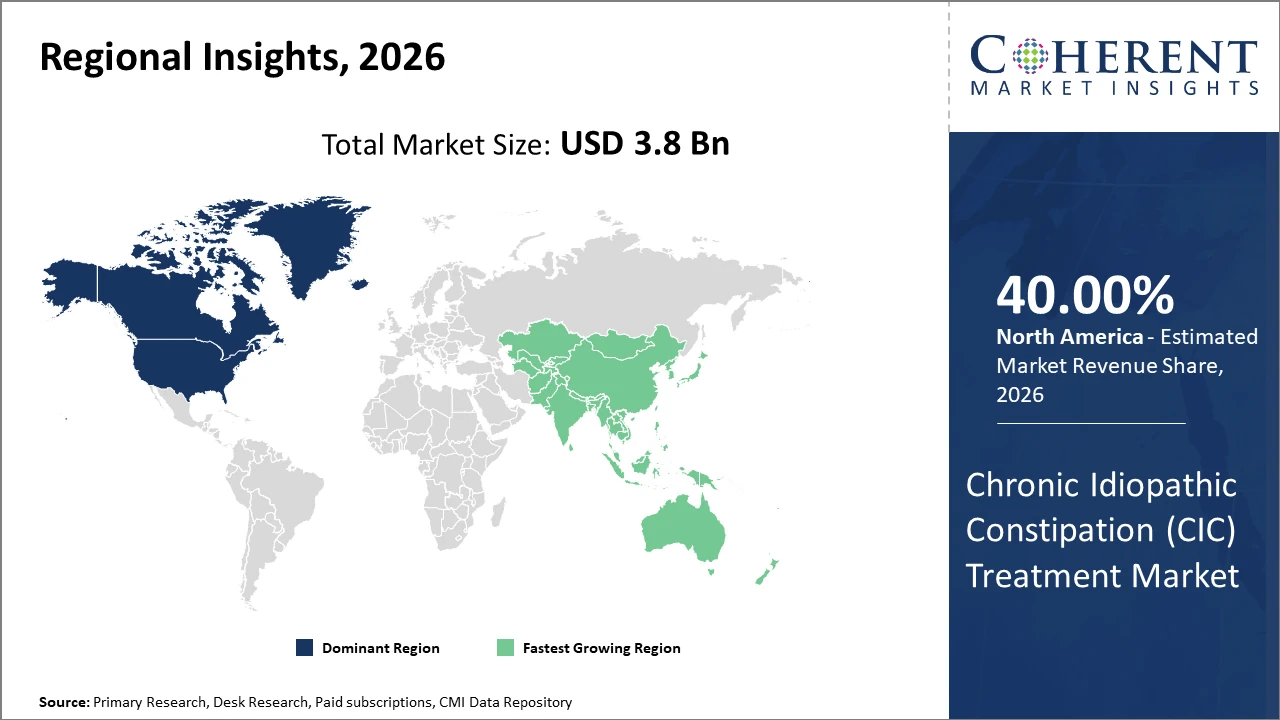
North America holds the largest market share due to advanced healthcare infrastructure, strong reimbursement systems, and early adoption of novel therapies.
Asia Pacific is the fastest-growing region, fueled by rising disease prevalence, improving healthcare access, and increasing awareness of chronic gastrointestinal conditions.
Chronic Idiopathic Constipation (CIC) Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Chronic Idiopathic Constipation (CIC) Treatment Market Insights, By Drug Class
Guanylate Cyclase-C agonists lead the Chronic Idiopathic Constipation (CIC) treatment market with about 38% share, supported by strong clinical efficacy and superior symptom relief outcomes. Clinical findings in recent studies, including 2025 data, indicate up to 25% greater improvement in bowel movement frequency versus traditional therapies, reinforcing their dominance. Chloride channel activators represent the fastest-growing segment, driven by increasing adoption of agents like plecanatide and rising awareness of safety benefits, contributing to around 20% year-on-year growth. Serotonin 5-HT4 receptor agonists maintain stable demand, particularly in complex cases, while osmotic laxatives remain widely used in cost-sensitive markets. Emerging therapies continue evolving in early-stage development.
Chronic Idiopathic Constipation (CIC) Treatment Market Insights, By Route of Administration
The oral segment dominates the Chronic Idiopathic Constipation (CIC) treatment market with approximately 82% share, primarily due to high patient preference, ease of administration, and improved long-term adherence. Between 2024 and 2026, clinical and real-world studies showed better compliance with oral therapies compared to other routes. Advancements in sustained-release and targeted oral formulations have further enhanced efficacy and patient outcomes, strengthening segment leadership. Rectal administration remains important for severe or acute cases but has limited adoption due to discomfort and procedural complexity. The “others” segment, including emerging delivery systems like transdermal approaches, remains niche but is being explored for refractory CIC cases.
Chronic Idiopathic Constipation (CIC) Treatment Market Insights, By End User
Specialty clinics account for the highest growth share in the Chronic Idiopathic Constipation (CIC) treatment market, driven by concentrated gastroenterology expertise and personalized care models that improve treatment outcomes. In 2025, data indicated a 15% rise in CIC patient visits and therapy administration through specialty clinics, reflecting a shift toward more specialized and structured care pathways. Hospitals continue to hold a substantial share due to broad inpatient and outpatient services, although they face operational and capacity constraints. Home care settings are expanding with digital health tools enabling remote monitoring and better adherence.
Chronic Idiopathic Constipation (CIC) Treatment Market Trends
The CIC treatment market is experiencing accelerated adoption of next-generation therapies, particularly guanylate cyclase-C agonists and improved oral formulations that enhance efficacy and patient adherence.
Innovation in drug delivery and formulation is enabling regional players to expand their market presence and strengthen competitive positioning.
Ironwood Pharmaceuticals’ commercialization efforts in North America contributed to an estimated 15% increase in market penetration in 2025, reflecting strong product uptake.
Digital health integration, including remote monitoring and adherence tools, is improving real-world treatment outcomes and supporting long-term therapy management.
Regulatory incentives in Europe are accelerating approvals of novel CIC therapies, driving faster product launches and intensifying competition in 2026.
Chronic Idiopathic Constipation (CIC) Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Chronic Idiopathic Constipation (CIC) Treatment Market Analysis and Trends
North America dominates the Chronic Idiopathic Constipation (CIC) treatment market, acquiring share of 40%, supported by advanced healthcare infrastructure, strong reimbursement frameworks, and high patient awareness levels. The United States alone accounts for over 45% of the regional market share in 2026, driven by widespread access to advanced prescription therapies and strong presence of leading pharmaceutical companies such as AstraZeneca and Ironwood Pharmaceuticals. Rising prevalence of gastrointestinal disorders and an expanding aging population continue to fuel demand for CIC treatments. Additionally, government-led health initiatives promoting early diagnosis and effective disease management further strengthen market growth, ensuring sustained adoption of innovative and targeted therapeutic options across the region.
Asia Pacific Chronic Idiopathic Constipation (CIC) Treatment Market Analysis and Trends
Asia Pacific is the fastest-growing region in the Chronic Idiopathic Constipation (CIC) treatment market, projected to record a CAGR above 9% during the forecast period. This growth is driven by improving healthcare infrastructure, rising prevalence of chronic gastrointestinal disorders, and expanding access to prescription therapies. Key countries such as China and India play a central role, supported by evolving regulatory systems that streamline drug approvals and increasing disposable incomes. Additionally, the region’s growing pharmaceutical manufacturing capabilities are strengthening supply chains. Leading companies are investing in regional R&D centers to develop cost-effective and population-specific treatments tailored to diverse patient needs and clinical profiles.
Chronic Idiopathic Constipation (CIC) Treatment Market Outlook for Key Countries
USA Chronic Idiopathic Constipation (CIC) Treatment Market Analysis and Trends
The USA leads the global Chronic Idiopathic Constipation (CIC) treatment market, supported by advanced research infrastructure, high healthcare expenditure, and strong adoption of innovative therapies. In 2025, the approval and widespread use of guanylate cyclase-C agonists such as linaclotide significantly boosted market performance, contributing to an estimated 12% revenue growth. The presence of major players like Ironwood Pharmaceuticals, along with strategic partnerships and collaborations, continues to drive therapeutic innovation and expand patient access. Additionally, strong reimbursement support from public and private payers enhances treatment affordability, further strengthening market expansion and ensuring sustained leadership of the U.S. in the global CIC therapeutics landscape.
Germany Chronic Idiopathic Constipation (CIC) Treatment Market Analysis and Trends
Germany Chronic Idiopathic Constipation (CIC) treatment market is driven by an aging population, rising gastrointestinal disorders, and strong healthcare reimbursement systems. Prescription therapies such as linaclotide, prucalopride, and lubiprostone are widely adopted due to improved efficacy over laxatives. Increasing physician awareness and preference for targeted pro-secretory and prokinetic agents are supporting market growth. OTC laxatives and fiber supplements remain common for mild cases. Germany’s hospital and pharmacy networks ensure strong drug accessibility. Key trends include rising demand for long-term symptom management, growing digital health consultations, and increased focus on gut microbiome research influencing future therapeutic development. Competition among pharmaceutical firms
Analyst Opinion
Demand-side growth is driven by rising use of guanylate cyclase-C agonists and chloride channel activators, with linaclotide and plecanatide boosting North American treatment prevalence by ~15% in 2025.
Supply expansion in Asia Pacific, where manufacturing capacity grew by ~22% in 2025, improved affordability and widened access in emerging markets.
Treatment use is expanding beyond gastroenterology into preventive and geriatric care settings, with non-specialist prescriptions rising about 10% year-over-year in 2026.
Geriatric patients account for nearly 35% of CIC cases in 2025, making age-related prevalence a key driver of future market demand and treatment customization.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.8 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 6.5 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | AstraZeneca, Allergan, Novartis AG, Shire Plc, Abbott Laboratories, Ipsen, Lupin Limited, Ironwood Pharmaceuticals, Viatris | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Chronic Idiopathic Constipation (CIC) Treatment Market Growth Factors
The rising prevalence of gastrointestinal disorders globally is significantly driving demand for Chronic Idiopathic Constipation (CIC) treatments, with diagnosed cases increasing by approximately 9% between 2024 and 2026. Growth is further supported by an expanding geriatric population experiencing age-related bowel motility issues, which increases long-term treatment needs. Technological advancements in drug delivery, especially improved oral formulations, have enhanced patient compliance and therapeutic effectiveness, contributing to a 14% rise in oral treatment adoption by 2025. Additionally, favorable reimbursement frameworks in North America and Europe are improving patient access, thereby boosting market revenue and strengthening growth across hospitals, specialty clinics, and other care settings.
Chronic Idiopathic Constipation (CIC) Treatment Market Development
In August 2025, Dr Reddy’s Laboratories launched Linaclotide, a GC-C agonist for adult chronic constipation in India under the brand name Colozo, available in 72 mcg and 145 mcg strengths, becoming the first company to introduce the drug in the country following regulatory approval.
Key Players
Leading Companies of the Market
AstraZeneca
Allergan
Novartis AG
Shire Plc
Abbott Laboratories
Ipsen
Lupin Limited
Ironwood Pharmaceuticals
Viatris
Competitive strategies in the Chronic Idiopathic Constipation (CIC) treatment market are increasingly focused on pipeline expansion, collaborations, and precision-driven drug development. For example, Ironwood Pharmaceuticals’ co-development agreement with AbbVie in 2025 accelerated the commercialization of advanced CIC therapies, contributing to revenue growth of over 18% in 2026 and strengthening its market presence in North America. Similarly, AstraZeneca’s emphasis on biomarker-based, precision medicine approaches through clinical trials has improved treatment targeting and efficacy, resulting in increased market share across regulated markets. Overall, these strategies highlight a shift toward innovation-led growth, strategic partnerships, and personalized therapy development in the global CIC therapeutics landscape.
Chronic Idiopathic Constipation (CIC) Treatment Market Future Outlook
The Chronic Idiopathic Constipation (CIC) treatment market is expected to grow steadily over the coming years, driven by aging populations, rising gastrointestinal disorders, and increasing awareness of gut health. Future growth will be supported by advancements in targeted therapies such as prokinetic and secretagogue agents, improving patient outcomes and long-term symptom control. Digital health adoption, personalized treatment approaches, and expanding online pharmacy access are also expected to enhance treatment accessibility worldwide. Additionally, ongoing clinical research and pipeline innovation focusing on gut-brain axis modulation and microbiome therapies will further strengthen the market outlook through 2033 supporting long-term sustainable global market growth
Chronic Idiopathic Constipation (CIC) Treatment Market Historical Analysis
Historically, the Chronic Idiopathic Constipation (CIC) treatment market relied heavily on over-the-counter (OTC) laxatives, stool softeners, and fiber supplements as first-line therapy due to limited targeted treatment options. Over time, growing understanding of gastrointestinal physiology led to the development of prescription therapies such as lubiprostone, linaclotide, and prucalopride, which significantly improved treatment efficacy and patient outcomes. Increased diagnosis rates and physician awareness further expanded the patient base. The market gradually shifted from symptomatic relief to mechanism-based treatments targeting intestinal secretion and motility. In recent years, advancements in drug development and clinical research have transformed CIC management into a more specialized therapeutic area.
Sources
Primary Research Interviews:
Gastroenterologists, general physicians, colorectal specialists, hospital pharmacists, retail pharmacy managers, and healthcare providers involved in prescribing and managing Chronic Idiopathic Constipation (CIC) treatments.
Clinical researchers, pharmacologists, gastrointestinal drug development experts, regulatory consultants, clinical trial investigators, and R&D directors specializing in functional gastrointestinal disorders, gut motility agents, secretagogues, and prokinetic therapies.
Magazines:
Gastroenterology & Endoscopy News – Coverage of GI disorder management, constipation treatment trends, and endoscopic insights into bowel dysfunction.
Digestive Disease Week (DDW) Publications – Updates on clinical advancements in constipation therapies, gut motility research, and microbiome studies.
Pharmacy Times – Insights into prescription trends, OTC laxative use, and pharmaceutical innovations in gastrointestinal care.
Journals:
The American Journal of Gastroenterology – Research on CIC pathophysiology, clinical treatment outcomes, and therapeutic advancements.
Neurogastroenterology & Motility – Studies on gut-brain axis, intestinal motility disorders, and novel pharmacologic treatments.
Alimentary Pharmacology & Therapeutics – Clinical trials and drug efficacy studies for CIC therapies including secretagogues and prokinetics.
Newspapers:
Financial Times – Coverage of pharmaceutical investments, GI drug pipelines, and market expansion strategies.
Reuters – Reporting on drug approvals, clinical trial results, and healthcare industry developments.
Bloomberg – Insights into pharmaceutical company performance, product launches, and global gastrointestinal drug market trends.
Associations:
American Gastroenterological Association (AGA) – Clinical guidelines, research updates, and best practices in gastrointestinal disorder management.
European Society of Neurogastroenterology & Motility (ESNM) – Research and standards for motility disorders and CIC treatment approaches.
World Gastroenterology Organisation (WGO) – Global guidelines on constipation management and digestive health initiatives.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients