Procalcitonin Antibody Market Size and Forecast – 2026 – 2033
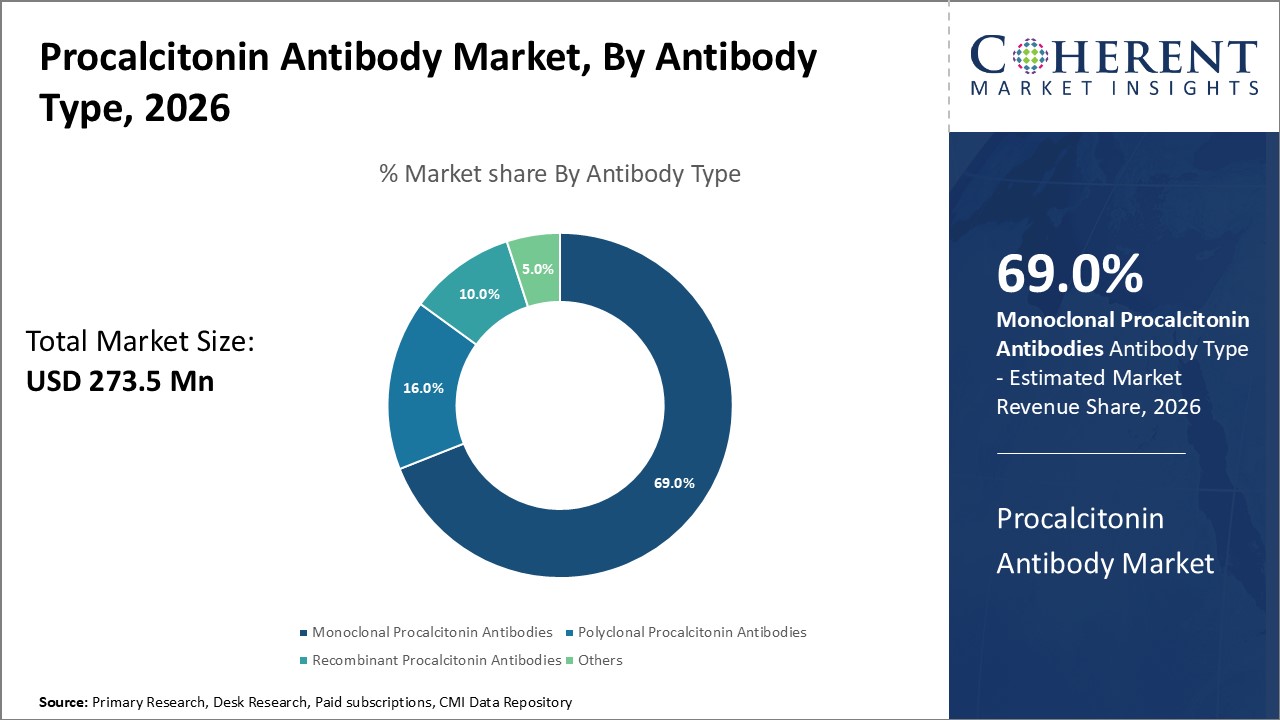
The Global Procalcitonin Antibody Market size is estimated to be valued at USD 273.5 million in 2026 and is expected to reach USD 460 million by 2033, exhibiting a compound annual growth rate (CAGR) of 9.4% from 2026 to 2033.
Global Procalcitonin Antibody Market Overview
Procalcitonin (PCT) antibodies are immunodiagnostic reagents used for detecting procalcitonin levels, a key biomarker for bacterial infections and sepsis. Products in this market include monoclonal and polyclonal antibodies utilized in ELISA, chemiluminescence, and rapid test kits. These antibodies are vital components in diagnostic assays that guide antibiotic stewardship and infection management in hospitals. Manufacturers focus on developing high-affinity antibodies with improved specificity, stability, and compatibility with automated analyzers. The demand for procalcitonin antibodies has risen with the increasing prevalence of sepsis and the growing use of rapid diagnostic platforms for infection monitoring and critical care decision-making.
Key Takeaways
Monoclonal procalcitonin antibodies dominate the market segment, accounting for 69% of the overall industry share, driven by superior specificity and sensitivity in diagnostic assays.
Clinical diagnostics remain the largest application segment, leveraging procalcitonin antibody-based assays for early infection detection, contributing significantly to market revenue growth.
North America leads the regional market, representing over 35% of the total market share, owing to advanced healthcare infrastructure and early adoption of innovative diagnostic tools.
Asia Pacific is the fastest-growing region with a CAGR surpassing 10%, propelled by expanding healthcare access, increasing prevalence of infectious diseases, and government initiatives boosting biotechnology investments.
Procalcitonin Antibody Market Segmentation Analysis
To learn more about this report, Request Free Sample
Procalcitonin Antibody Market Insights, By Antibody Type
Monoclonal Procalcitonin Antibodies dominate the market share. Monoclonal antibodies are preferred due to their high specificity and reproducibility, making them preferred for clinical diagnostic assays where precision is critical. Recent innovations in monoclonal antibody technology have enhanced affinity, enabling the detection of procalcitonin at ultra-low concentrations, which directly boosts diagnostic accuracy and market revenue. Recombinant procalcitonin antibodies represent the fastest-growing subsegment, owing to their enhanced batch-to-batch consistency and scalability, increasing adoption in pharmaceutical research and multiplex assay development.
Procalcitonin Antibody Market Insights, By Application
Clinical Diagnostics dominate the market share. They primarily focus on sepsis and infection biomarker testing, where procalcitonin antibody assays are integral to early detection protocols. Rising sepsis prevalence and increasing adoption of biomarker-based diagnostics in intensive care units drive this demand. Pharmaceutical Development is the fastest-growing subsegment, supported by expanding use of procalcitonin antibodies in drug discovery and validation, including clinical trials for anti-infective agents. Research Laboratories leverage these antibodies for exploratory studies into immunoinflammatory mechanisms, sustaining steady market participation.
Procalcitonin Antibody Market Insights, By End-User
Hospitals account for the majority market share due to their critical role in patient care for infections and sepsis management, supported by established procurement and lab workflows. Diagnostic Laboratories represent the fastest-growing subsegment, fueled by increasing outsourcing trends and demand for high-throughput testing facilities. Research Institutes continuously drive innovation through academic and clinical research into antibody applications. Biotechnology Companies, while smaller in share, significantly push technological advances and novel antibody development, facilitating market growth across segments.
Procalcitonin Antibody Market Trends
Emerging trends in the procalcitonin antibody market emphasize higher integration of multiplex diagnostic approaches, as demonstrated by product launches incorporating procalcitonin alongside inflammatory biomarkers in 2026, enabling holistic pathogen detection and prognosis.
This innovation aligns with the rising demand for personalized medicine and rapid diagnostic turnaround times in hospital settings.
Additionally, the development of recombinant antibodies is gaining prominence, supported by robust intellectual property portfolios and manufacturing scalability that enhance the reliability of clinical assays.
Digitization of diagnostic data integrated with procalcitonin antibody tests further enhances decision-making, a trend anticipated to reshape market dynamics.
Procalcitonin Antibody Market Insights, By Geography

To learn more about this report, Request Free Sample
North America Procalcitonin Antibody Market Analysis and Trends
North America holds the dominant market share of approximately 35% due to advanced clinical research infrastructure, higher healthcare spending, and an active base of market players headquartered in the U.S. The presence of stringent regulatory standards ensures quality and adoption of novel antibody products. Leading firms here invest heavily in product innovation, bolstering market revenue and growth.
Asia Pacific Procalcitonin Antibody Market Analysis and Trends
Asia Pacific is the fastest-growing region with a CAGR exceeding 10%, propelled by expanding healthcare facilities, supportive government policies promoting biotechnology, and a rising burden of infectious diseases. Countries such as China and India have catalyzed domestic manufacturing capacity, contributing significantly to market expansion and accessibility.
Procalcitonin Antibody Market Outlook for Key Countries
USA Procalcitonin Antibody Market Analysis and Trends
The U.S. remains a pivotal market accounting for over 28% of global procalcitonin antibody revenue. Its high adoption rate is fueled by widespread sepsis management protocols that prioritize biomarker-guided therapy. Major players such as Bio-Rad and Thermo Fisher Scientific maintain strong manufacturing and distribution networks, collaborating with leading hospitals to innovate assay techniques. Government incentives for diagnostic innovation and stringent FDA regulations ensure continued market growth, with investments exceeding USD 50 million in R&D during 2025–2026.
Germany Procalcitonin Antibody Market Analysis and Trends
Germany, a significant European market contributor, reflects substantial growth due to a well-established healthcare infrastructure and increasing incidence of hospital-acquired infections. The country’s regulatory environment supports rapid approvals for novel diagnostic antibodies. Local companies and multinational subsidiaries actively engage in product enhancements focused on assay sensitivity and ease of use. Germany is projected to continue its leadership role in Europe’s procalcitonin antibody market, supported by consistent government funding towards biotechnology innovation and infection control programs.
Analyst Opinion
Procalcitonin antibody production capacity has increased by approximately 15% year-over-year, notably in biomanufacturing hubs across the Asia Pacific, which supports rising market share and revenue expansion. For example, in 2025, China alone exported procalcitonin antibody reagents worth USD 55 million, reflecting strong supply-side growth.
Pricing strategies have evolved with bulk procurement contracts enabling cost reductions ranging from 6% to 9% for antibody reagents, fostering uptake in emerging markets where healthcare infrastructures are expanding rapidly. This dynamic pricing directly influences market revenue and underlines market dynamics favoring volume growth.
Nano and micro detection capabilities tied to next-gen antibodies are a growing focus. In 2025, over 41% of newly launched procalcitonin assay kits incorporated enhanced antibody constructs, enabling detection at picogram levels, an innovation that heightens analytical precision and expands applications in personalized medicine scenarios.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 273.5 million |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.4% | 2033 Value Projection: | USD 512.9 million |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Bio-Rad Laboratories, Thermo Fisher Scientific, Merck KGaA, Abcam plc, Sino Biological Inc., Agilent Technologies Inc., Abnova Corporation, Santa Cruz Biotechnology Inc., Fitzgerald Industries International, HyTest Ltd. | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Procalcitonin Antibody Market Growth Factors
The growing prevalence of sepsis and respiratory infections globally directly drives demand for effective procalcitonin antibodies in diagnostic applications. For instance, sepsis incidence in the U.S. rose by 12% from 2023 to 2026, intensifying the need for reliable biomarkers. Advancements in immunoassay technologies, including high-affinity monoclonal antibodies, have improved diagnostic accuracy, fuelled by significant R&D spending which exceeded USD 40 million globally in 2025, specifically for antibody enhancements.
Healthcare infrastructure development in emerging economies facilitates wider adoption of procalcitonin antibody-based diagnostics, supported by increased healthcare expenditures in regions such as the Asia Pacific, where expenditure grew by a CAGR of 7.5% during 2023–2026. Regulatory approvals for multiplex biomarker platforms incorporating procalcitonin antibodies create market expansion opportunities by broadening assay utility across diverse clinical conditions beyond sepsis.
Procalcitonin Antibody Market Development
In November 2025, Roche entered into a definitive agreement to acquire Poseida Therapeutics, a clinical-stage company developing allogeneic cell therapies and non-viral genetic-medicine platforms for cancer, autoimmune and rare diseases. Under the agreement announced on 26 November 2025, Poseida stockholders will receive US $9.00 per share in cash at closing, plus a non-tradeable contingent value right (CVR) to receive up to US $4.00 per share in cash upon achievement of specified milestones — representing a total equity value of up to approximately US $1.5 billion. The transaction is expected to close in the first quarter of 2026, subject to customary closing conditions.
In late October 2026, Thermo Fisher Scientific announced its intention to acquire Clario Holdings, Inc., a clinical-development and endpoint-data solutions provider, for approximately US $8.9 billion in cash (with potential earn-outs bringing the total up to ~US $9.4 billion). This move is aimed at enabling pharma and biotech customers to accelerate innovation through deeper clinical insights and enhanced trial-data infrastructure. The transaction is expected to close by mid-2026, subject to regulatory approvals.
Key Players
Leading Companies of the Market
Bio-Rad Laboratories
Thermo Fisher Scientific
Merck KGaA
Abcam plc
Sino Biological Inc.
Agilent Technologies Inc.
Abnova Corporation
Santa Cruz Biotechnology Inc.
Fitzgerald Industries International
HyTest Ltd.
Several market players have adopted collaborative research partnerships to enhance monoclonal antibody specificities, resulting in over 25% increased assay sensitivity reported by Thermo Fisher between 2023–2025. Bio-Rad has implemented strategic acquisitions, expanding its pipeline in procalcitonin reagents, positively impacting their market footprint and revenue streams. Sino Biological’s localized manufacturing in the Asia Pacific has enabled cost-effective supply and faster delivery times, thereby capturing growing regional market demand.
Procalcitonin Antibody Market Future Outlook
The market outlook for procalcitonin antibodies is promising as diagnostic applications expand beyond sepsis to include respiratory and systemic infections. Continued development of high-sensitivity antibodies and multiplexed panels will enable faster and more comprehensive infection profiling. The adoption of point-of-care and cartridge-based testing platforms will enhance accessibility in resource-limited settings. Furthermore, as antimicrobial stewardship gains policy attention, the role of PCT testing in guiding antibiotic therapy is expected to become even more central to hospital diagnostics.
Procalcitonin Antibody Market Historical Analysis
The Procalcitonin Antibody Market has grown in response to rising demand for accurate biomarkers in bacterial infection and sepsis management. Initially confined to research laboratories, procalcitonin testing gained clinical relevance as healthcare systems prioritized early sepsis detection to reduce mortality. Diagnostic companies developed high-affinity monoclonal antibodies and immunoassay kits compatible with automated analyzers, enhancing test sensitivity and reliability. Integration into clinical protocols made PCT a routine test in emergency and intensive care settings.
Sources
Primary Research Interviews:
Immunoassay Developers
Clinical Microbiologists
Biotech Scientists
Diagnostic Lab Directors
Databases:
GlobalData Diagnostic Reagents Reports
WHO Infectious Disease Data
PubMed Biomarker Studio
ClinicalTrials.gov
Magazines:
Clinical Laboratory News
MedTech Insight
Diagnostics World
BioProcess International
Journals:
Clinical Biochemistry
Journal of Immunoassay & Immunochemistry
Clinical Infectious Diseases
Biomarker Insights
Associations:
International Federation of Clinical Chemistry (IFCC)
American Society for Microbiology (ASM)
European Society of Clinical Microbiology
Infectious Diseases (ESCMID)
WHO Laboratory Diagnostics Division
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients