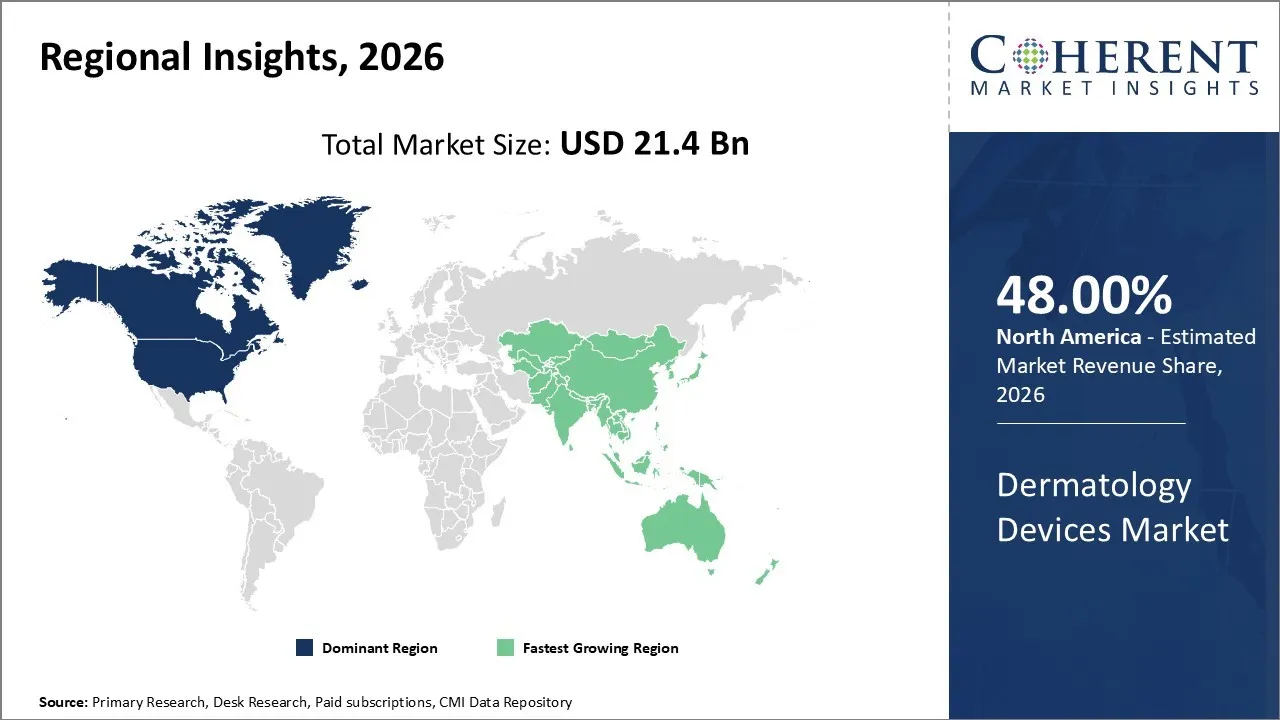
The dermatology devices market was valued at USD 21.4 Bn in 2026 and is forecast to reach a value of USD 64.7 Bn by 2033 at a CAGR of 17% between 2026 and 2033.
Devices used in dermatology are those that can be utilized during surgery, for treating skin conditions, or for diagnosing them. Dermatologists and surgeons can diagnose and treat skin conditions with the aid of dermatology devices. Various medical care facilities, including hospitals, dermatology clinics, doctor's offices, and university research institutions, use these devices. Increasing prevalence of skin diseases is expected to drive growth of the dermatology devices market.
|
Current Event |
Description and its Impact |
|
FDA Approvals for New Injectable and Aesthetic Devices |
|
|
Launch of Advanced Dermatology Treatment Platforms by Candela and Zimmer Biomet |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Artificial intelligence is transforming dermatology devices by enabling faster, more accurate detection and diagnosis of skin conditions such as melanoma, acne, and psoriasis. AI-powered imaging systems analyze patterns in skin lesions, assisting clinicians in early identification and risk assessment while reducing human error. These devices improve workflow efficiency by prioritizing high-risk cases and minimizing diagnostic delays. They also expand access to care through teledermatology and mobile-based tools, especially in underserved regions. Additionally, AI supports personalized treatment planning and continuous monitoring, leading to better patient outcomes and optimized resource use in healthcare systems facing specialist shortages.
In March 2025, the world’s first autonomous AI system for skin cancer detection, DERM, earned Class III CE marking in Europe. It achieved 99.8% accuracy in ruling out cancer—exceeding dermatologists’ 98.9%—and reduced assessment wait times from months to days.
In terms of product type, the Laser IPL segment is expected to lead the market with 45% share in 2026. The dominance of Laser IPL technologies can be attributed to their ability to cater to a comprehensive range of skin conditions including hair removal, pigmentation disorders, vascular lesions, acne treatment, skin rejuvenation treatments, etc. The Food and Drug Administration (FDA) has consistently approved many laser technologies for dermatological use, with over 200 laser devices currently cleared for different skin treatments, demonstrating the regulatory confidence in such technologies.
The American Academy of Dermatology reports that laser treatments have become the gold standard for various cosmetic as well as medical dermatology procedures, with dermatologists highly preferring laser-based solutions owing to their precision, controllability, and superior patient outcomes.
In terms of device type, the treatment devices segment is expected to hold 75% share of the market in 2026. The dominance of treatment devices stems from their direct impact on patient outcomes as well as their ability to cater to a broad range of dermatological conditions ranging from acne and rosacea to skin cancer and age-related skin issues.
For instance, Laser therapy systems have changed how many skin problems are treated. Machines like fractional CO₂ lasers are now used a lot to improve skin texture, reduce scars, and help smooth wrinkles. The American Academy of Dermatology reports that laser treatments have become one of the most requested treatments in dermatology practices, making substantial demand for advanced treatment devices.
In terms of application, the hair removal segment is projected to account for 35% share of the market in 2026. The prominence of hair-focused devices can be attributed to several key factors, including growing consumer demand for both in-clinic and at-home hair solutions, as well as increasing awareness of effective treatments for hair thinning and hair loss. The American Academy of Dermatology says that laser hair treatments and at‑home hair removal devices are widely used in the United States, with many people having these treatments or using the devices each year.
For instance, Hairmax, the pioneer of at-home laser hair growth technology, announced that it exhibited its full portfolio of FDA-cleared laser hair growth devices thinning hair care solutions at the 2026 American Academy of Dermatology (AAD) Annual Meeting, held March 27–29 in Denver, Colorado. This highlights the growing role of at-home laser devices in the U.S. hair treatment market and the high presence of consumer-friendly solutions alongside clinic-based procedures.
In terms of end user, the hospital segment is projected to capture a 45% share in 2026, due to their comprehensive infrastructure, extensive patient base, as well as ability to procure high-value dermatological equipment across multiple departments. Hospitals stay the biggest users of dermatology devices because they already buy and use expensive, advanced machines for skin treatments and tests.
To learn more about this report, Request Free Sample
North America is expected to dominate the dermatology devices market with 48% share in 2026, due to its robust healthcare infrastructure, high adoption of advanced medical technologies, along with comprehensive regulatory framework that facilitates innovation in dermatological treatments. The region's dominance stems from the established of world-class medical institutions such as Mayo Clinic, Cleveland Clinic, Johns Hopkins Hospital, etc., which serve as innovation center for dermatological device development as well as clinical research.
For instance, in March 2026, Galderma, a leading dermatology company, said that the U.S. FDA has approved its product Restylane Contour to treat hollow temples in adults over 21. This follows earlier approvals for cheek and midface treatments, helping doctors fix areas of lost facial volume.
Asia Pacific is expected to exhibit the fastest growth, due to the region's rapidly expanding middle class population looking for advanced aesthetic and therapeutic dermatological treatments. The World Health Organization has highlighted the escalating burden of skin diseases in Asia Pacific countries, fueled by urbanization, pollution exposure, as well as changing lifestyle patterns that necessitate sophisticated dermatological interventions. Government healthcare initiatives across countries like China, India, and Southeast Asian nations have prioritized dermatology care accessibility, leading to substantial investments in modern medical equipment and facility upgrades.
For instance, in February 2026, Zimmer Biomet has introduced new electric dermatome devices that have better imaging features. With the Zimmer Dermatome AN, Air or Electric Dermatomes, you choose the power source that works best for you. All deliver the same smooth cutting action that produce, clean uniform cuts.
The U.S. leads this segment primarily due to its well-established network of dermatology clinics, hospitals, as well as specialized treatment centers that extensively utilize advanced dermatological devices. The American Academy of Dermatology reports that there are over 12,000 practicing dermatologists across the United States, making substantial demand for cutting-edge dermatological equipment.
For instance, in September 2025, Dermalogica, mainly a skincare brand, has got FDA approval for its first medical aesthetics device. The PRO Pen Microneedling System will start being sold in the United States in 2026.
South Korea contributes the highest share in the dermatology devices market in Asia Pacific owing to its advanced healthcare infrastructure, sophisticated consumer awareness regarding skin health, as well as robust regulatory framework that promotes rapid adoption of innovative dermatology technologies. The Korean dermatology devices market leadership stems from the deeply embedded beauty culture of the country, where skincare is taken as a fundamental aspect of daily life rather than a luxury.
Because many people in Korea want advanced skin treatments like lasers and radiofrequency systems, the market there accepts these new dermatology devices quickly. South Korea has also made rules so medical devices can be approved much faster than before, helping new device technology reach the market sooner.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.4 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 17% | 2033 Value Projection: | USD 64.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Valeant Pharmaceuticals International Inc., Lumenis, Ltd., Hologic, Inc., Syneron Candela Ltd., Avita Medical Ltd., Alma Lasers, Ltd., Lutronics Corporation, Beijing Toplaser Technology Co. Ltd., Shanghai Fosun Pharmaceutical Group Co. Ltd., and Cutera, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of skin disorders globally is a pivotal factor propelling the demand for advanced dermatology devices, significantly shaping the trajectory of the dermatology devices market. Various skin conditions such as acne, eczema, psoriasis, rosacea, and skin cancers are becoming more common due to factors including urbanization, environmental pollution, changing lifestyles, and rising awareness about skin health.
The rising demand for minimally invasive cosmetic procedures has become a critical driver in the expansion of the dermatology devices market. Consumers increasingly favor these treatments due to their reduced recovery times, lower risks of complications, and natural-looking results compared to traditional surgical interventions. Procedures such as laser skin resurfacing, microdermabrasion, and injectable treatments like botulinum toxin and dermal fillers offer patients effective solutions for issues like wrinkles, pigmentation, and acne scars without the need for extensive surgery.
Emerging markets with rising disposable income represent a significant and transformative opportunity for the dermatology devices market, driven largely by evolving consumer awareness and increasing healthcare investments. In countries like India, Brazil, and some parts of Southeast Asia, more people are earning more money and joining the middle class. Because of this, people can spend more and are buying advanced skin care and treatment devices for health and beauty.
The Dermatology Devices Market is experiencing a major advancements driven by rising awareness about skin health as well as increasing prevalence of skin disorders globally. Major drivers include the growing demand for minimally invasive cosmetic procedures, technological innovations such as laser therapy, as well as enhanced diagnostic methods for skin cancer detection.
Additionally, an escalating aging population with heightened sensitivity to dermatological conditions is escalating the need for advanced treatment devices.
However, the market faces certain restraints, primarily regulatory challenges along with high costs associated with sophisticated devices, which can limit availability in developing regions. Moreover, apprehension regarding side effects from dermatological treatments may also slow adoption rates in some factors.
Opportunities abound in emerging economies, especially in Asia-Pacific, where rising disposable incomes and increasing urbanization are fueling demand for cosmetic dermatology and skin care treatments. This region is also registering the fastest growth due to expanding healthcare infrastructure and rising awareness campaigns. Furthermore, integration of artificial intelligence and teledermatology presents promising avenues for improving diagnostic accuracy and patient outreach.
North America currently dominates the dermatology devices market, fueled by well-established healthcare infrastructure, high healthcare expenditures, as well as continued investments in research and development.
Overall, the dermatology devices landscape is expcted for robust growth, leveraging technological innovations as well as expanding patient bases, though stakeholders must navigate regulatory complexities and cost barriers to maximize market potential.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients