Dermatomyositis is an idiopathic inflammatory myopathy characterized by skeletal muscle weakness and skin changes. Dermatomyositis is characterized by a rash accompanying, or more often, preceding muscle weakness. The most common symptom is muscle weakness, usually affecting those muscles that are closest to the trunk of the body (proximal). Eventually, patients have difficulty rising from a sitting position, climbing stairs, lifting objects, or reaching overhead. In some cases, distal muscles (those not close to the trunk of the body) may be affected later in the course of the disease.
The Global dermatomyositis drug market is estimated to be valued at US$ 773.7 million in 2022 and is expected to exhibit a CAGR of 4.3% during the forecast period (2022-2030).
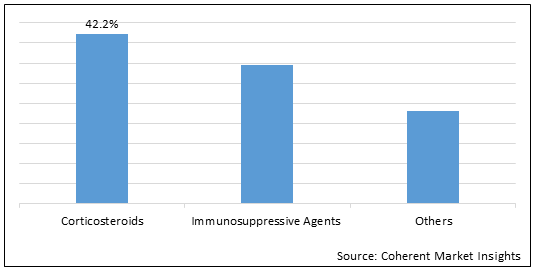
Figure 1.Global Dermatomyositis Drug Market Share (%) in Terms of Value, By Drug Type, 2022
To learn more about this report, Request Free Sample
Increasing prevalence of dermatomyositis is expected to drive the market growth during the forecast period
The Increasing prevalence of dermatomyositis is expected to drive the global dermatomyositis drug market growth over the forecast period. For instance, according to data published by Journal of Managed Care & Specialty Pharmacy in November 2020, estimated that the prevalence of Dermatomyositis is 2.0-10.0 cases per 100,000 persons, globally, in 2020. Moreover, the annual incidence rate of Dermatomyositis is 4.0-7.7 per 1 million persons, globally, in 2020.
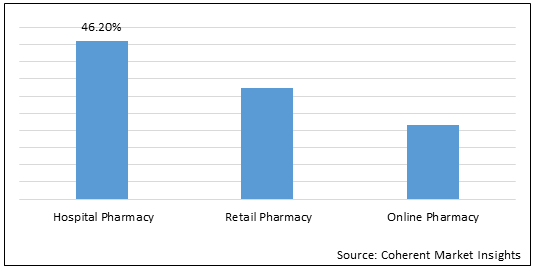
Figure 2.Global Dermatomyositis Drug Market Share (%), By Distribution Channel, 2022
To learn more about this report, Request Free Sample
Increasing product approval for the treatment of dermatomyositis are expected to drive the market growth during the forecast period
Increasing product approval for the treatment of dermatomyositis is expected to drive the global dermatomyositis drug market growth during the forecast period. For instance, in August 2021, The U.S. Food and Drug Administration (FDA) had granted approval to Octapharma USA, one of the largest human protein product manufacturers for Octagam 10%, the first and only intravenous immunoglobulin (IVIg) to be indicated for the treatment of adult dermatomyositis.
Dermatomyositis Drug Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2021 | Market Size in 2022: | US$ 773.7 Mn |
| Historical Data for: | 2017-2020 | Forecast Period: | 2022 to 2030 |
| Forecast Period 2022 to 2030 CAGR: | 4.3% | 2030 Value Projection: | US$ 1,081.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Teva Pharmaceuticals USA, Inc., Hikma Pharmaceuticals PLC, Asahi Kasei Corporation., Octapharma USA, Healthcare Pharmaceuticals Limited., Zydus Cadila, Alkem Laboratories Ltd, Apotex Inc, Eli Lilly and Co, and F.Hoffman-La Roche Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Dermatomyositis Drug Market– Impact of Coronavirus (COVID-19) Pandemic
The supply of raw materials required for production of pharmaceuticals has been severely disrupted due to the forced quarantine and lack of labor during the COVID-19 pandemic. As the link between regional warehouses is not smooth, transportation of raw materials between regions cannot be carried out successfully. This shortage of raw materials and components has negatively affected the supply chain of the global dermatomyositis drug market . Thus, COVID-19 pandemic is expected to have negative impact on supply chain of intraocular melanoma treatment market.
According to an article published by the Exploratory Research in Clinical and Social Pharmacy journal in June 2021, a study was carried out to evaluate the impact of COVID-19 on pharmaceutical systems and supply chain in resource-limited countries in Sub-Saharan countries such as Namibia. This study revealed negative impact on availability and access of essential drugs, sanitation and hygiene products, and antimicrobials. Most pharmaceutical companies and pharmacies in Namibia experienced delayed manufacturing and distribution of drugs, which is attributed to reduced inter-country transportation of pharmaceutical goods and limited in-country capacity to manufacture drugs. For instance, according to an review article published by the European Pharmaceutical Review journal in November 2020, COVID-19 pandemic has caused major challenges with respect to drug shortages and increased manufacturing costs across the globe. Moreover, the same source stated that COVID-19 pandemic has also led to problems such as stockpiling drugs, transportation delay, and others. Some measures that can be taken to ease the supply of pharmaceuticals during the pandemic include next generation technologies such as digital network platforms designed to work across multiple pharmaceutical enterprises and ensure the timely delivery of drugs to patients across the globe.
Global Dermatomyositis Drug Market: Restraint
The major factors that hinder growth of the global dermatomyositis drug market include high treatment cost of dermatomyositis treatment. IVIG (intravenous immune globulin) is very effective treatment for dermatomyositis, but it’s also very expensive. Health insurance companies, in an effort to cut costs, often don’t want to pay for such costly treatments. So, Health insurance companies create roadblocks for patients, such as requiring them to first try a number of other treatments without success before they allow IVIG. This practice is called ‘step therapy’ If the drug is prescribed off-label, many companies simply refuse to pay for it.
Key Players
Key players operating in the global dermatomyositis drug market include Pfizer Inc., Teva Pharmaceuticals USA, Inc., Hikma Pharmaceuticals PLC, Asahi Kasei Corporation., Octapharma USA, Healthcare Pharmaceuticals Limited., Zydus Cadila, Alkem Laboratories Ltd, Apotex Inc, Eli Lilly and Co, and F.Hoffman-La Roche Ltd
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients