Electrotherapy System Market is estimated to be valued at USD 7,080.7 Mn in 2026 and is expected to reach USD 12,213.9 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033. For instance, key players operating in the market are focused on launching new products in the market, which is expected to drive the market growth over the forecast period. For instance, in April 2020, Omron Healthcare, Inc., a medical device company, announced the launch of the Focus TENS Therapy for Knee, a wearable device with transcutaneous electrical nerve stimulation technology, indicated for chronic, arthritic and acute pain associated with exercise strain or daily activity.
Analysts’ Views on Global Electrotherapy System Market :
The electrotherapy system market growth can be driven by the increasing approval of the electrotherapy system by the regulatory bodies. For instance, on February 23, 2023, ONWARD Medical N.V., a medical technology company, announced that it has received the Breakthrough Device Designation status from the U.S. Food and Drug Administration (FDA) for the use of its ARC-EX platform for bladder control, alleviation of spasticity, and blood pressure regulation in people with spinal cord injury.
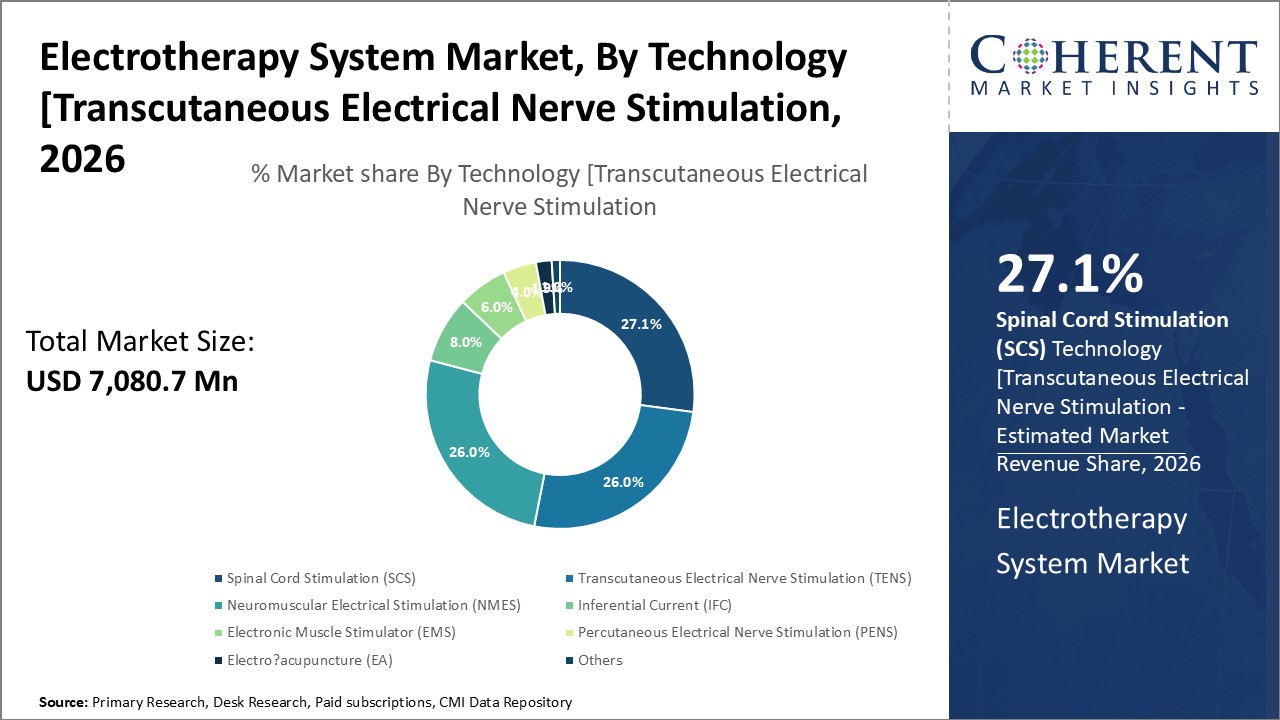
Figure 1. Global Electrotherapy System Market Share (%), by Technology, 2026
To learn more about this report, Request Free Sample
Global Electrotherapy System Market - Driver
Increasing approval of the electrotherapy system by regulatory bodies
Key players operating in the market are focused on getting approval for the new technology electrotherapy system by the regulatory bodies, which is expected to drive the global electrotherapy system market over the forecast period. For instance, on March 2, 2023, Neuro20 Technologies Corp., a wearable medical device manufacturing company, received the U.S. Food and Drug Administration (FDA) pre-market approval for its Neuro20 PRO System. The Neuro20 PRO System is the Whole-Body Electrical Muscle Stimulator Suit, Software and Operating System involuntarily contracts 42 muscles independently or by any combination of co-contraction, with control specificity for each muscle for 1 to 10 patients at a time.
Rapidly rising incidence of spinal injuries
Increasing number of incidence of spinal injuries due to rising geriatric population and hectic day to day life schedule is expected to drive the global electrotherapy system market over the forecast period. For instance, in December 2021, according to the data published by the National Spinal Cord Injury Statistical Center, it was found that there are approximately 18,000 new cases of spinal cord injury each year in the U.S. and about 282,000 persons are estimated to be living with spinal cord injury in the year 2022 in the U.S. Furthermore, according to the same source it was also estimated that in the year 2022, approximately 299,000 persons, with a range from 253,000 to 378,000 persons in the U.S. are living with spinal cord injury.
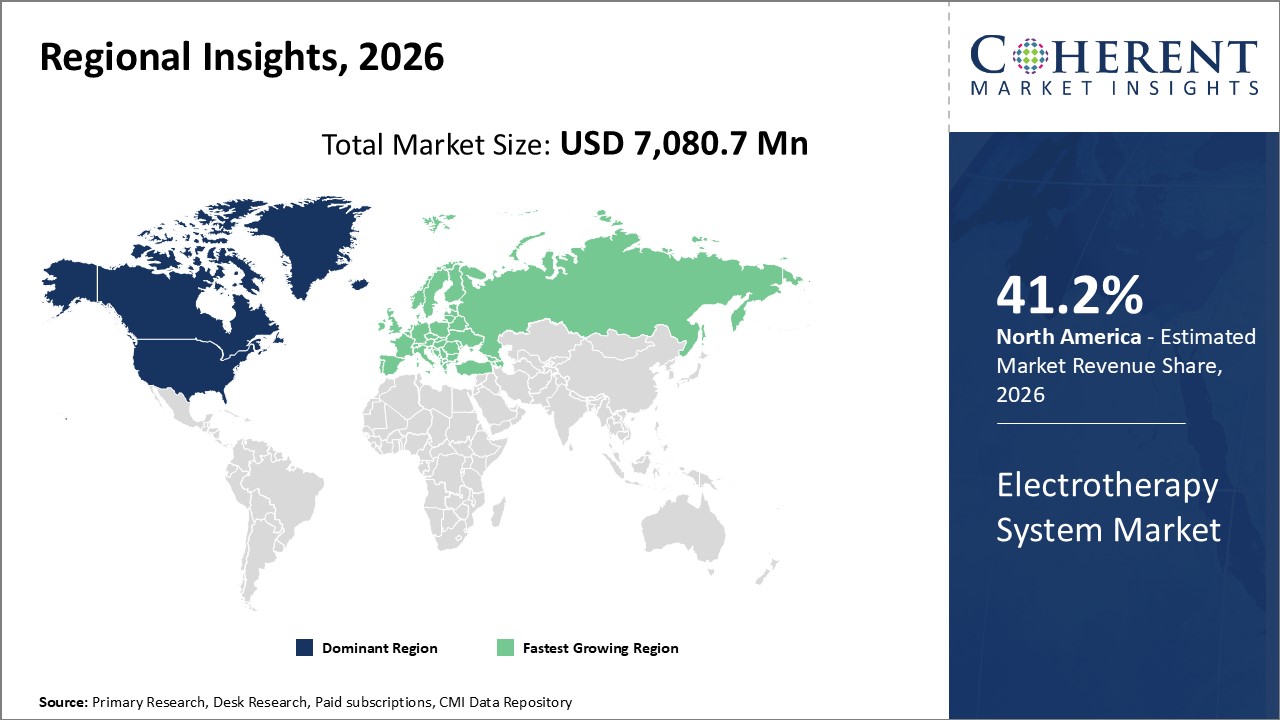
Figure 2.Global Electrotherapy System Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Electrotherapy System Market - Regional Analysis
Among all regions, North America is expected to dominate the market over the forecast period. This is attributed due to North America holding a 41.2% market share in the year 2026 and rising geriatric population in the North America is rising the demand for the electrotherapy in North America, which is expected to drive the global electrotherapy system market over the forecast period. For instance, in December 2022, according to the data published by the World Health Organization, it was estimated that from 2023–2053 period, 73 million people, on average, will be age 65 or older in the north america. Furthermore, according to the samre source, it is also estimated that 17% of the U.S. population above age 65 and older is suffering with the spinal injury and spinal pain issue.
Moreover, Europe electrotherapy system market is expected to witness significant growth over the forecast period, owing to the increasing prevelance of the spinal cord injury in the European region. For instance, in December 2022, according to the data published by the European Spinal Cord Injury Federation, it was found that in the Western European countries, an incidence of traumatic spinal cord injury (SCI) is 16 to 19.4 new cases per million inhabitants per year.
Furthermore, healthcare providers were facing challenges in terms of additional manpower, equipment, consumables, and other resources, which were required to ensure safety in hospitals and provide treatment to patients with other diseases. This has impacted the overall healthcare sector negatively. Additionally, there were delays in hospitalization as the number of patient visits decreased, which is expected to affect the global electrotherapy system market over the forecast period.
Global Electrotherapy System Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7,080.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 12,213.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic Plc, Zynex Medical Inc., DJO Global, Inc., Nevro Corporation, BTL Industries, Inc., Zealmax Innovations Pvt. Ltd., LivaNova PLC, EMS Physio Ltd., STYMCO Medical, Omron Corporation, TensCare Ltd, Cionic, Neuro20 Technologies Corp. and other prominent key players |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Electrotherapy System Market Segmentation:
The global electrotherapy system market report is segmented into technology, application, therapy, and end user.
Based on Technology, the market is segmented into Transcutaneous Electrical Nerve Stimulation (TENS), Neuromuscular Electrical Stimulation (NMES), Inferential Current (IFC), Percutaneous Electrical Nerve Stimulation (PENS), Electronic Muscle Stimulator (EMS), Spinal Cord Stimulation (SCS), Electro-acupuncture (EA), and Others. Out of which, Spinal Cord Stimulation is expected to dominate the market over the the forecast period and this is attributed due to increasing research and development acitivities to launch new product in the market. For instance, in September 2026, ONWARD Medical N.V., a medical technology company, announced that the Up-LIFT pivotal study evaluating ARC-EX Therapy achieved positive results in its primary effectiveness endpoint of improvement in upper extremity strength and function. ARC-EX Therapy is a proprietary non-invasive spinal cord stimulation technology designed to restore movement and other functions in people with movement disabilities.
Based on Application, the market is segmented into Chronic wound healing, neuromuscular dysfunction, pain management, tissue repair, musculoskeletal disorder, physical therapy, iontophoresis and others. Out of which, the pain management segment is expected to dominate the market over the forecast period, and this is attributed due to increasing prevelance of the trauma and sports injury. For instance, in March 2022, according to the data published by the Word Health Organization, it was estimated that about 30 million children and teens participate in some form of organized sports, and more than 3.5 million injuries each year. Furthermore, according to the same source, it was also estimated that the 570,000 people faced traumatic pain in the U.S. in the year 2021.
Based on End User, the market is segmented into hospitals, rehabilitation centers, clinics. Out of which, the hospitals segment is expected to dominate the market over the forecast period and this is attributed due to the prevalence of musculoskeletal disorder and orthopedic disorders and dysfunctions. For instance, in September 2022, according to the data published by the National Center for Biotechnology Information, during the period from 2017 to 2022, around 15,522 cartilage restoration procedures were reported by surgeons in the U.S. This increasing articular cartilage injuries is leading to increasing number of articular cartilage restoration procedures, which include microfracture, drilling, abrasion arthroplasty, and other procedures.
Among all segmentation, technology segment has the highest potential due to increasing approval of the electrotherapy system by the regulatory authorities. For instance, in January 2022, Medtronic Plc., a medical technology company, announced the approval from the U.S. Food and Drug Administration (U.S. FDA) for an Intellis recharheable nuerostimulator and Vanta recharge-free neurostimulator for the treatment of the chronic pain associated with diabetic peripheral neuropathy.
Global Electrotherapy System Market Cross Sectional Analysis:
In technology segment, Spinal Cord Stimulation (SCS) is expected to be a dominant segment in North America due to the increasing product approval in North America. For instance, in May 2022, Abbott Laboratories, a pharmaceutical and medical technology company, announced the approval of the U.S. FDA for its new Proclaim Plus spinal cord stimulation system, which provides tailored relief to multiple pain.
In application segment, pain management segment is dominant in hospitals due to increasing hospital admissions and visits for the sports injury, traumatic pains and accidental injury. For instance, in December 2022, according to the data published by the National Center for Biotechnology Information, it was estimated that in North America about 52,300 people visit hospital for pain management due to traumatic incidence in the year 2022.
Global Electrotherapy System Market : Key Developments
In March 2022, Cionic, a medical device company, announced that the Neural Sleeve received the U.S. Food and Drug Administration approval. The Neural Sleeve is the first algorithm-powered bionic garment designed to help those with mobility issues due to multiple sclerosis, stroke, cerebral palsy, and other conditions, regain functional movement.
In January 2022, TensCare Ltd, a medical device company, showcased its new-to-market products, which offer the latest in transcutaneous electrical nerve stimulation (TENS) indicated to be used for drug-free pain relief in the long-term treatment of chronic pain conditions such as back ache, diabetic neuropathy, sciatica, osteo-arthritis and the relief of the acute pain of childbirth. The TensCare Ltd showcased its transcutaneous electrical nerve stimulation (TENS) at the ABHI UK Pavilion at Arab Health 2022 conference.
In March 2021, Zynex Medical, a medical technology and biotechnology company, announced to attend the Raymond James Life Sciences and MedTech Conference 2022, to showcase all its pain management device and market its products in the different countries of Latin America.
Global Electrotherapy System Market : Restraint
Stringent regulatory compliance for electrotherapy products
The major factors that hinder the growth of the global electrotherapy system market is the stringent regulatory compliance for electrotherapy products and product recalls. For instance, in August 2022, according to data published by the National Center for Biotechnology Information (NCBI), stringent rules for developing new devices are set by regulatory authorities such as the U.S. Food and Drug Administration, the European Medicines Agency, among others, owing to which the development of new medical device has become a lengthy process, which involves coordination between drug sponsors, clinical researchers, and regulatory authorities and more time is required to verify drug safety. Furthermore, in August 2022, according to data published by the National Center for Biotechnology Information (NCBI), increasing cost of performing clinical trials has become hurdle for the approval of new drugs. The reason behind rising cost of performing clinical trial includes increasing comparator drugs (an investigational or marketed product (active control) or placebo (inactive control) used as a reference in a clinical trial), increasing clinical procedures, higher failure risk, and so on. Thus, the rising cost may lead to negative effect on drug manufacturers to manufacture fewer new drugs and perform research and development activities. The above restraint can be solved by following the Current Good Manufacturing Practice (CGMP) rules for manufacturing the electrotherapy devices.
Key Players
Major players operating in the global electrotherapy system market include Medtronic Plc, Zynex Medical Inc., DJO Global, Inc., Nevro Corporation, BTL Industries, Inc., Zealmax Innovations Pvt. Ltd., LivaNova PLC, EMS Physio Ltd., STYMCO Medical, Omron Corporation, TensCare Ltd, Cionic, Neuro20 Technologies Corp. and other prominent key players.
*Definition: A electrotherapy system is defined as the sum of therapeutic modalities of physical medicine capable to change the threshold of elicitation of nerve or muscle.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients