Global gastroparesis treatment market is estimated to be valued at USD 8.75 Bn in 2026 and is expected to reach USD 12.31 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.0% from 2026 to 2033. The market growth is driven by increasing prevalence of gastroparesis globally.
Discover market dynamics shaping the industry: Request Free Sample
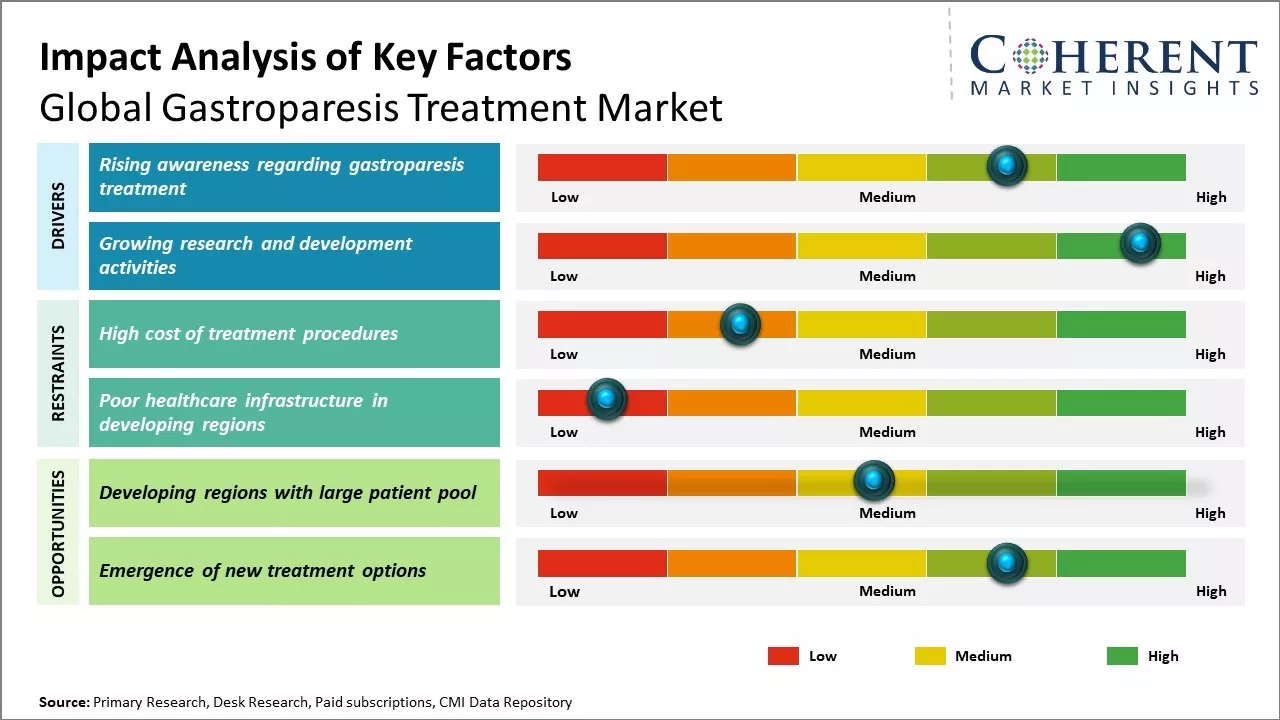
Market Driver - Rising awareness regarding gastroparesis treatment
Gastroparesis, commonly known as delayed gastric emptying, is a condition where the stomach takes too long to empty its contents. For many years, it went undiagnosed or was confused with other gastrointestinal disorders. However, over the past decade, recognition of gastroparesis as a distinct clinical entity has improved tremendously due to initiatives by patient advocacy groups who have helped spread awareness about the condition and its treatments. Support communities formed by patients on social media platforms have provided invaluable peer support for those suffering from gastroparesis. Through open discussions, reputed doctors and specialists have been able to address common myths and misconceptions about the disease. Their efforts have led to a better understanding that gastroparesis is not just a stomach problem but can severely impair quality of life, if left untreated. Individuals are now more knowledgeable about seeking timely medical help rather than suffering silently. Awareness drives conducted by advocacy organizations in collaboration with medical professionals have effectively reached both urban and rural populations. Educational campaigns through various online and offline mediums highlight the signs and symptoms of gastroparesis along with stressing the importance of early diagnosis. This allows patients to recognize warning signs in the early stages itself and get appropriate treatment before complications arise. Healthcare providers are also alert to include gastroparesis in their differential diagnosis for gastrointestinal disorders.
Get actionable strategies to beat competition: Request Free Sample
Growing research and development activities
Increased funding and focus on gastroparesis research can drive the market growth. Several academic institutes and pharmaceutical companies have ramped up clinical trials for developing novel drugs and medical devices. While metformin and prokinetic agents form the mainstay of current gastroparesis treatment, their effectiveness is limited in advanced cases. This has driven the medical community to investigate new therapeutic avenues. A number of startups are working on formulating targeted drug formulations for selectively delivering medications to the stomach. Their technology aims to enhance drug solubility, permeability, and release kinetics for optimizing symptom relief. Several clinical-stage biologics are also being assessed for their efficacy in modulating vagal nerve activity and immune responses linked to gastroparesis. Researchers are leveraging advancements in biologics to design more selective and less invasive treatment protocols. Meanwhile, medical device giants have invested substantially in R&D of gastric electrical stimulation devices. New generation devices are being miniaturized for improved comfort and battery life. Some incorporate remote monitoring capabilities as well. Emerging neuromodulation platforms study applying spinal cord or vagal nerve stimulation techniques for gastroparesis. Accelerated evaluation of novel therapeutic strategies can address the unmet needs of gastroparesis patients globally.
Key Takeaways from Analyst:
Global gastroparesis treatment market growth is driven by rising prevalence of gastroparesis worldwide. North America currently dominates the market, owing to developed healthcare infrastructure and high awareness regarding gastroparesis treatment options. However, Asia Pacific region is expected to emerge as fastest growing region due to increasing patient pool and healthcare spending in countries like China and India.
Increasing metabolic disorders like diabetes, which is a major cause of gastroparesis, can drive the market growth. Rising surgical procedures leading to neurological damage can also contribute to this disease burden. The market players have robust pipeline of drugs in the clinical trials which once approved can aid the market expansion. High cost of treatment and limited treatment options particularly for diabetic gastroparesis can hamper the market growth.
Technological advancements in the feeding pumps and pacemaker systems for treating gastroparesis through neuromodulation offers lucrative opportunities for the market growth. Adoption of non-invasive treatment techniques can also offer growth opportunities over the forecast period. Increasing R&D investments by companies and gradual shift towards value-based care can drive the market growth.
Market Challenge - High cost of treatment procedures
Global gastroparesis treatment market growth can be hampered due to high cost associated with treatment procedures. Gastroparesis often requires costly surgeries or other interventional procedures to manage symptoms. Surgical procedures like pyloroplasty and gastrojejunostomy aim to improve gastric emptying but are expensive. Even minor procedures and implanted drug delivery devices are expensive. The development and adoption of new treatment options is also slowed by the heavy investment required in research and clinical trials. Increasing healthcare costs have further exacerbated the issue and put gastroparesis treatment out of reach for patients from lower socioeconomic backgrounds. Addressing the expense of gastroparesis management remains a major obstacle to improving care and quality of life for affected individuals around the world.
Market Opportunity: Developing regions with large patient pool
Developing regions of the world present a major opportunity for gastroparesis treatment market growth due to their huge populations and increasing disease burden. Countries in Asia Pacific, Latin America, Middle East and Africa are witnessing rising incidence of gastroparesis, owing to growing obese and diabetic populations. At the same time, there is abundant room for penetration of new treatment products in these underserved markets where awareness and access to care is currently low. International players can gain a significant customer base by catering to the needs of developing healthcare systems and collaborating with local pharma companies. Low manufacturing costs allow firms to offer products at affordable price points tailored for price-sensitive markets. This helps address a large unmet medical need while also expanding revenues.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
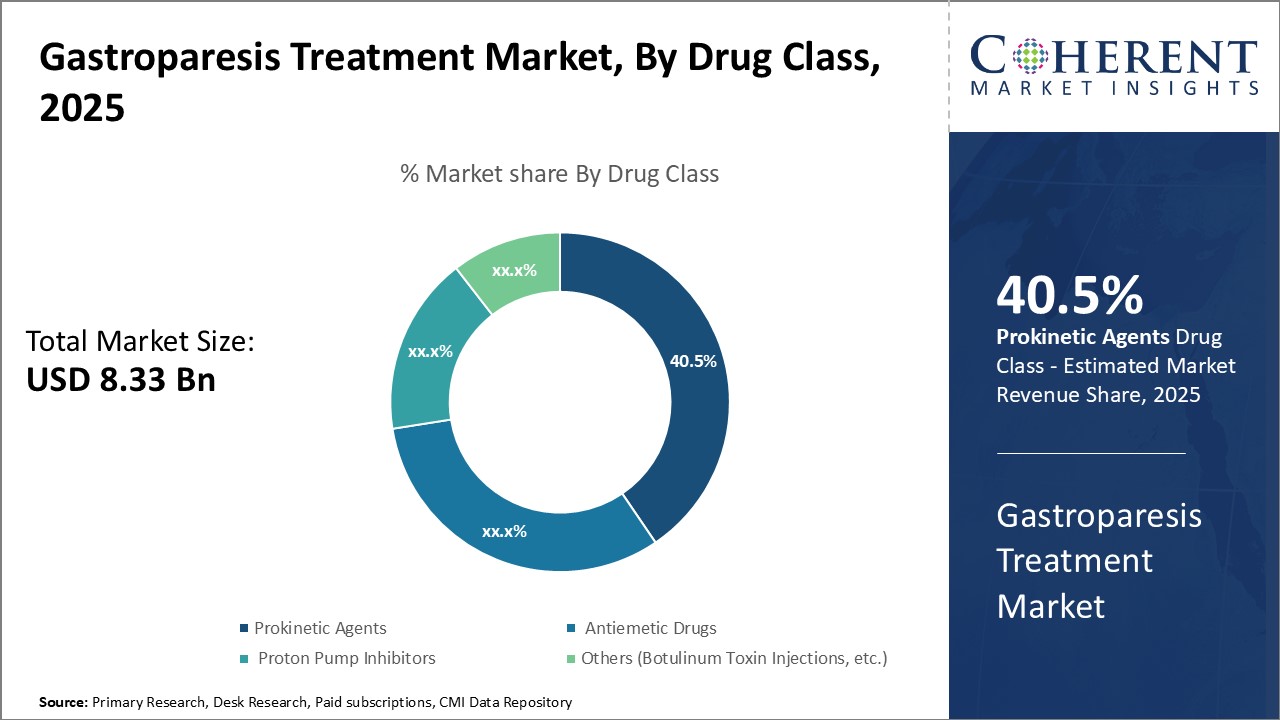
By Drug Class - Effective treatment of symptoms boosts demand for prokinetic agents
In terms of drug class, prokinetic agents segment is estimated to contribute the highest market share of 41.5% in 2026, owing to its effectiveness in treating various gastroparesis symptoms. Prokinetic agents work by increasing gastrointestinal motility to promote gastric emptying. Some of the most commonly used prokinetic agents include metoclopramide, domperidone, erythromycin, and prucalopride. Metoclopramide is one of the first line treatments for gastroparesis due to its prokinetic and antiemetic properties. It acts by stimulating smooth muscles in the stomach and small intestine to promote gastric emptying. However, its use is limited due to potential neurological side effects with long term use. Domperidone is an alternative for patients who cannot tolerate metoclopramide. It selectively interacts with receptors in the gastrointestinal tract to improve gastric motility with limited neurologic side effects. Erythromycin in non-antibiotic doses is also effective for gastroparesis by stimulating motilin receptors to induce gastrointestinal contractions. Newer agents like prucalopride work by selectively targeting 5-HT4 receptors in the gut. Their proven efficacy in alleviating debilitating symptoms like nausea, vomiting and bloating without significant safety issues boosts demand for prokinetic agents over other drug classes.
To learn more about this report, Request Free Sample
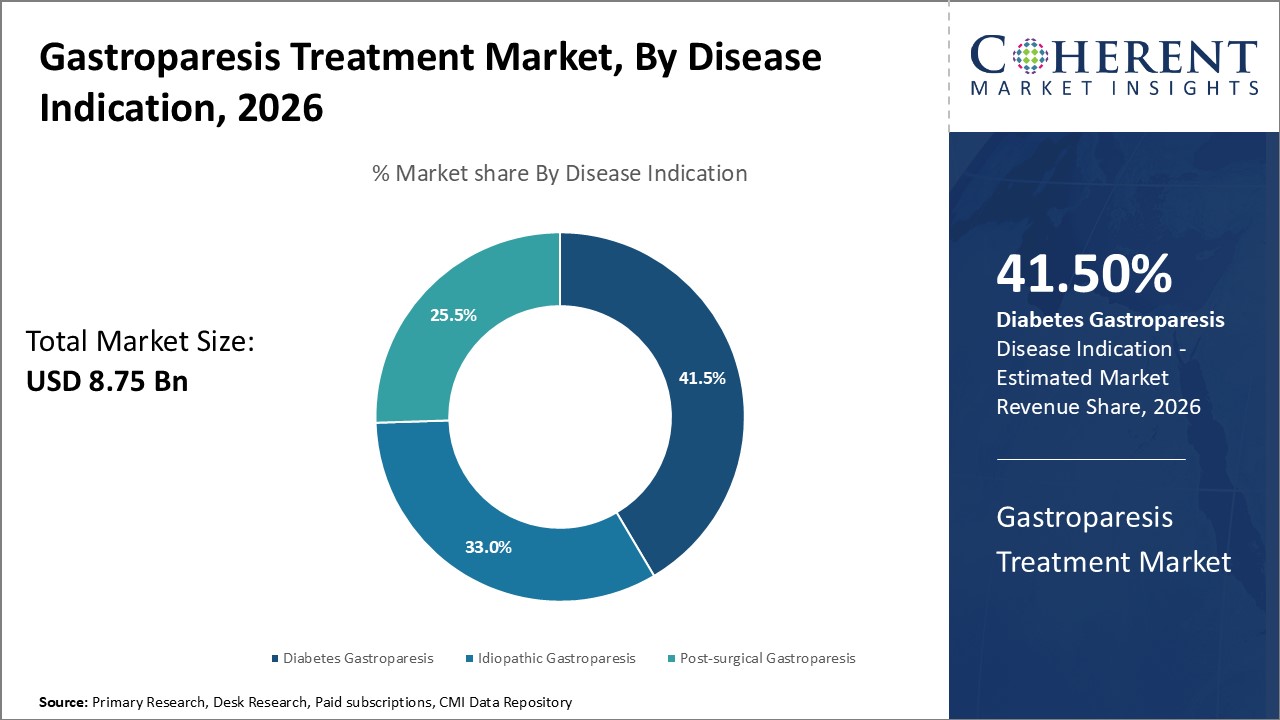
By Disease Indication- High prevalence of diabetes drives the diabetes gastroparesis segment growth
In terms of disease indication, diabetes gastroparesis segment is estimated to contribute the highest market share of 41.5% in 2026, owing to rising global prevalence of diabetes. Gastroparesis is a common complication of long-standing diabetes, which occurs when the vagus nerve controlling the stomach is damaged, resulting in delayed gastric emptying. Gradual increase in diabetes populations over the years leads to growing diabetes gastroparesis patient pool. According to estimates, about 30% of type 1 diabetes and 12-15% of type 2 diabetes patients eventually develop gastroparesis. Given diabetes currently afflicts over 400 million people worldwide, even a modest fraction of these patients experiencing gastroparesis results in a huge diabetes gastroparesis population. Changes in lifestyle and diet over last few decades have boosted the ongoing diabetes epidemic, further, driving the segment growth. Improved diagnostic tools have also aided earlier diagnosis and recognition of diabetes gastroparesis.
By Type- Better accessibility and convenience offered by OTC drugs drives segment growth
In terms of type, over-the-counter drugs segment is estimated to contribute the highest market share of 60.5% in 2026, owing to better accessibility and convenience offered by OTC drugs as compared to prescription drugs. Patients often prefer OTC options to quickly relieve mild to moderate gastroparesis symptoms without requiring a doctor's visit. Common OTC treatments include antacids containing H2-receptor blockers or proton pump inhibitors to reduce excess stomach acid and antiemetic drugs containing active ingredients such as diphenhydramine for nausea. These provide fast relief of symptom bouts without a prescription. Their unmonitored nature allows round-the-clock accessibility and on-demand intake for symptom flare-ups without strict dosage schedules. This flexibility and autonomy drives the popularity of OTC drugs over prescription medications requiring frequent physician appointments and close monitoring. Improvements and wider availability of OTC drug formulations are furthering their accessibility, convenience and popularity over prescription options.
Need a Different Region or Segment? Request Free Sample
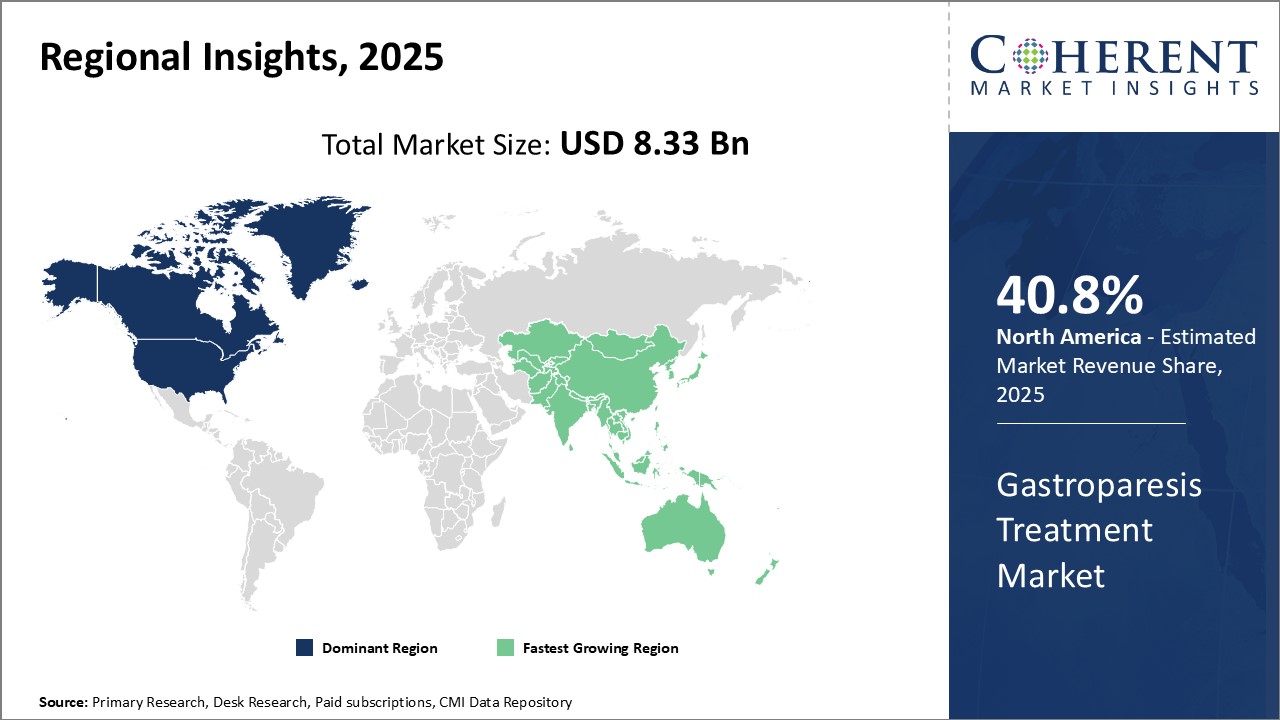
North America has established itself as the dominant region in the global gastroparesis treatment market with an estimated market share of 40.8% in 2026, due to high healthcare expenditure and widespread medical insurance coverage in countries like the U.S. Several leading pharmaceutical companies have their headquarters located in North America, enabling strong R&D capabilities and early adoption of latest treatment options. The presence of advanced healthcare infrastructure allows for easy access to specialized gastroparesis care.
Stringent regulatory guidelines ensure high standards of drug safety and efficacy. This provides patients with confidence in available therapeutics. Additional advantage comes from cultural factors where people are more willing to seek medical help and adhere to doctor's advice. North America also has a sizeable gastroparesis patient population base driven by growing prevalence of associated conditions like diabetes. Coupled with favorable reimbursement landscape, these demand side determinants make North America a lucrative market region.
Asia Pacific region is estimated to be the fastest growing gastroparesis treatment market. Countries like China, India and Japan have large patient pools suffering from gastroparesis due to their big populations and improving healthcare access. Asia Pacific offers lower costs for drug development and manufacturing compared to Western markets. This has encouraged local and multinational pharmaceutical manufacturers to strengthen their presence with new production facilities and clinical trials.
Improving economic conditions are enabling higher healthcare spends. Rising medical tourism is additionally boosting availability of gastroenterological services. Countries like India have a tradition of alternative treatment modalities that are gaining international acknowledgment. Their integration with modern therapies offers novel opportunities. Meanwhile, trade relationships are helping Asia Pacific countries to import latest drugs and partner with global leaders for product licensing.
Gastroparesis Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.75 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.0% | 2033 Value Projection: | USD 12.31 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
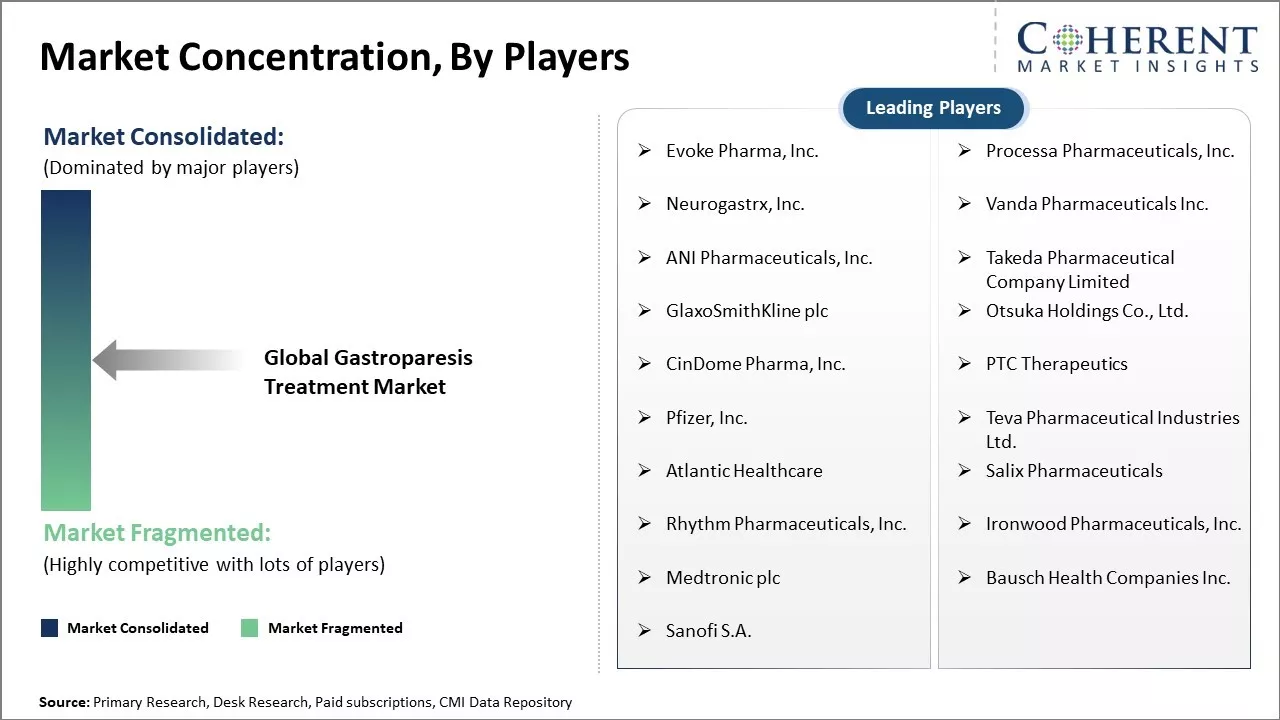
Evoke Pharma, Inc., Processa Pharmaceuticals, Inc., Neurogastrx, Inc., Vanda Pharmaceuticals Inc., ANI Pharmaceuticals, Inc., Takeda Pharmaceutical Company Limited, GlaxoSmithKline plc, Otsuka Holdings Co., Ltd., CinDome Pharma, Inc., PTC Therapeutics, Pfizer, Inc., Teva Pharmaceutical Industries Ltd., Atlantic Healthcare, Salix Pharmaceuticals, Rhythm Pharmaceuticals, Inc., Ironwood Pharmaceuticals, Inc., Medtronic plc, Bausch Health Companies Inc., Sanofi S.A. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Gastroparesis Treatment Market involves providing medical treatments to patients suffering from gastroparesis. Gastroparesis is a condition characterized by delayed emptying of food from the stomach into the small intestine. The global market covers treatment products and services across different regions internationally. Some of the common treatment options include medications to relieve symptoms, enteral feeding tubes for nutrition management, and in some severe cases, surgical procedures. Key players in this market offer a variety of pharmacological and non-pharmacological treatment solutions to patients dealing with gas.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients