Rheumatoid Arthritis Treatment Market Size and Forecast – 2026 – 2033
The global Rheumatoid Arthritis Treatment Market is projected to grow from about USD 32.7 billion in 2026 to around USD 48.2 billion by 2033, expanding at a CAGR of 5.7 % over 2026–2033, driven by rising RA prevalence and uptake of targeted therapies.
Global Rheumatoid Arthritis Treatment Market Overview
Rheumatoid arthritis (RA) treatment focuses on reducing inflammation, relieving pain, preventing joint damage, and improving quality of life. Management typically combines medications, physical therapy, and lifestyle changes. Key drug classes include nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, conventional disease-modifying antirheumatic drugs (DMARDs) like methotrexate, and advanced biologic and targeted synthetic therapies. Early diagnosis and aggressive treatment are critical to slow disease progression. Biologics targeting TNF, IL-6, and JAK pathways have significantly improved outcomes. Ongoing research aims to enhance efficacy, safety, and personalized treatment approaches.
Key Takeaways
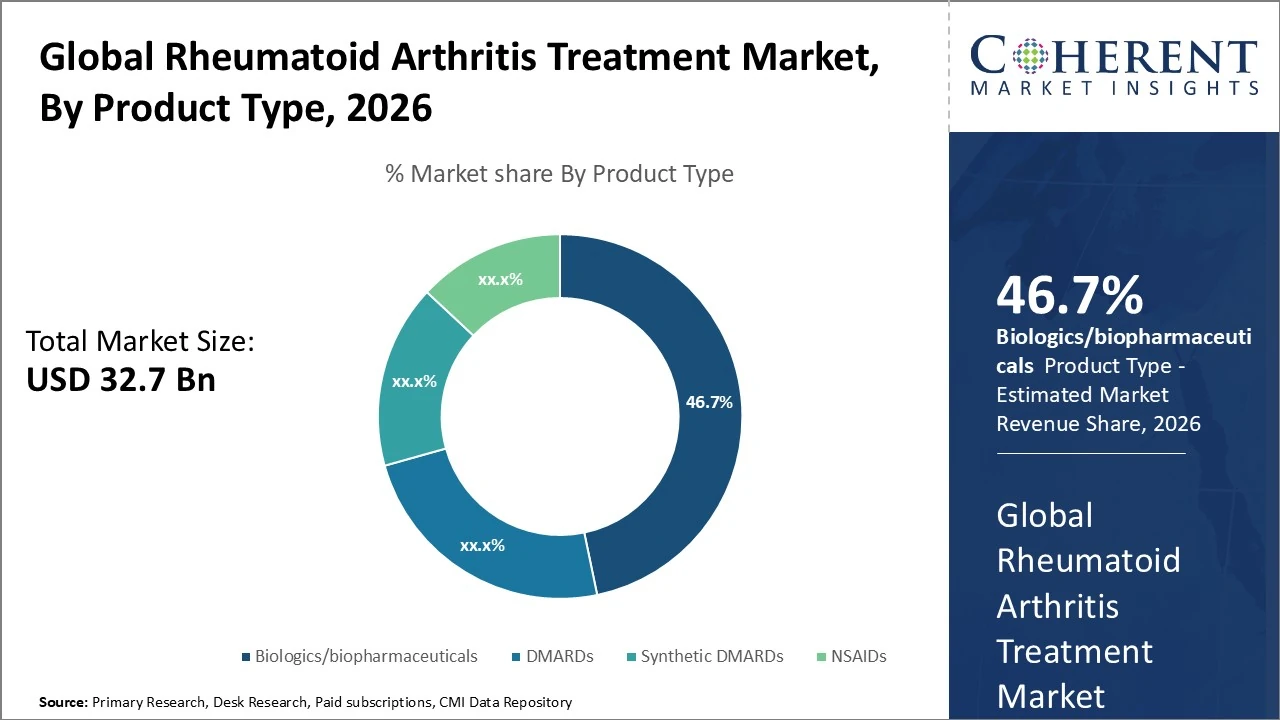
In the Rheumatoid Arthritis Treatment Market by product type, Biologics/biopharmaceuticals dominate, holding about 46.7% of global market share.
By end-user, Hospitals dominate with about 60% share.
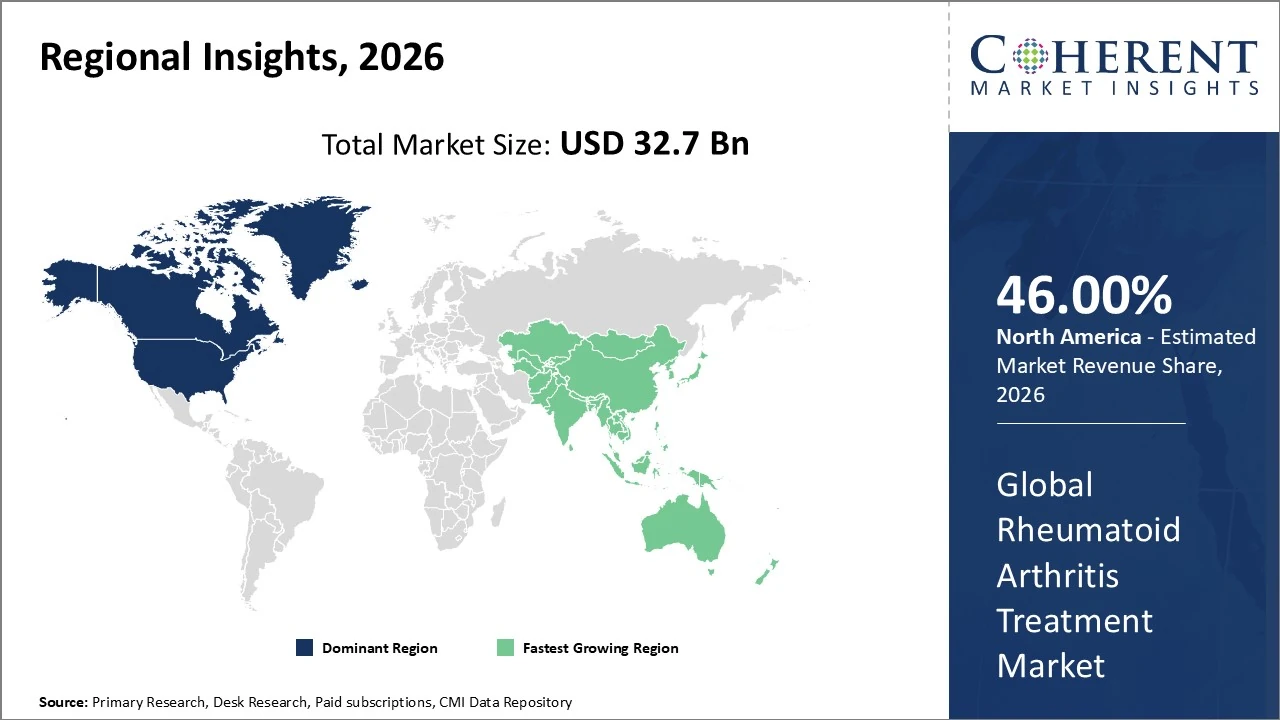
In North America, the Rheumatoid Arthritis Treatment Market leads globally, holding roughly 46% of total market share.
In the Asia Pacific Rheumatoid Arthritis Treatment Market, the region held about 17 % of global market share in 2026.
In the USA Rheumatoid Arthritis Treatment Market, the U.S. accounted for about ~90% of the North American RA treatment market.
Rheumatoid Arthritis Treatment Market Segmentation Analysis
To learn more about this report, Request Free Sample
Rheumatoid Arthritis Treatment Market Insights, By Product Type
In the Rheumatoid Arthritis Treatment Market by product type, Biologics/biopharmaceuticals dominate, holding about 46.7% of global market share due to targeted efficacy and rising adoption. Conventional DMARDs represent roughly 22%, while targeted Synthetic DMARDs account for around 15 %. NSAIDs typically hold about 12 %. Biologics and targeted synthetics are the fastest-growing segments through 2033, driven by innovation and expanding indications.
Rheumatoid Arthritis Treatment Market Insights, By End-User
In the Rheumatoid Arthritis Treatment Market by end-user, Hospitals dominate with about 60% share, as they provide comprehensive diagnosis, infusion services, and multidisciplinary care for complex RA cases. Specialty clinics/clinics hold roughly 28%, focusing on outpatient management and follow-ups. Homecare and other settings account for the remaining ~12 %, driven by increasing at-home administration of oral drugs and growing patient demand for convenience. Hospitals remain the primary revenue source, though homecare is the fastest growing segment.
Rheumatoid Arthritis Treatment Market Trends
Adoption of biologics, biosimilars, and targeted small-molecule drugs (e.g., JAK inhibitors) continues to grow, reshaping treatment paradigms and expanding market value.
AI-assisted monitoring, telehealth, and precision medicine tools are increasingly used to tailor treatments and improve patient outcomes.
Greater clinical development of novel agents and preference for combination regimens enhances therapeutic options and market expansion.
Rheumatoid Arthritis Treatment Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Rheumatoid Arthritis Treatment Market Analysis and Trends
In North America, the Rheumatoid Arthritis Treatment Market leads globally, holding roughly 46% of total market share due to high disease prevalence, strong healthcare spending, robust clinical infrastructure, and early adoption of biologics and advanced therapies. The U.S. alone accounts for about 90 % of the region’s value and is expanding at a ~6 % CAGR through the decade, driven by innovative drugs, favorable reimbursement, and high patient awareness. Canada’s contribution supports broad access to biologics and biosimilars. Market growth is bolstered by continuous launches and rising RA diagnosis and treatment uptake.
Asia Pacific Rheumatoid Arthritis Treatment Market Analysis and Trends
In the Asia Pacific Rheumatoid Arthritis Treatment Market, the region held about 17 % of global market share in 2026, with rapid growth driven by expanding healthcare access, rising RA prevalence, and increasing biosimilar adoption. APAC is the fastest-growing market, with projected CAGR of 8 % through the forecast period, led by major economies such as China, Japan, and India improving treatment infrastructure and affordability. Enhanced disease awareness, government health initiatives, and greater availability of advanced therapies like biologics and targeted DMARDs are key trend drivers.
Rheumatoid Arthritis Treatment Market Outlook for Key Countries
USA Rheumatoid Arthritis Treatment Market Analysis and Trends
In the USA Rheumatoid Arthritis Treatment Market, the U.S. accounted for about ~90% of the North American RA treatment market in 2026, valued at approximately US $14.1 billion and expected to grow at a ~5.5% CAGR through 2033. This growth is driven by rising RA prevalence, strong adoption of biologic DMARDs and JAK inhibitors, and advanced healthcare infrastructure supporting early diagnosis and specialty care. Biologics and targeted therapies dominate treatment uptake, enhancing patient outcomes and fueling market expansion. Continued innovation and favorable reimbursement also support sustained market growth.
Germany Rheumatoid Arthritis Treatment Market Analysis and Trends
In Germany’s Rheumatoid Arthritis Treatment Market, the market is growing steadily with strong adoption of biologic therapies and biosimilars, supported by a well-developed healthcare system and universal coverage. Biologics dominate due to superior efficacy, while biosimilars are increasingly used to reduce costs and expand access. Personalized treatment approaches and precision medicine are emerging, improving outcomes. The market is forecast to grow at around ~6 % CAGR through the next decade, driven by advanced rheumatology care networks and systematic disease management. Germany also leads Western Europe in RA therapeutics consumption share.
Analyst Opinion
Analysts highlight the rapid adoption of biologics, biosimilars, and JAK inhibitors as central to market expansion.
APAC and Latin America are expected to grow faster due to rising RA awareness, healthcare access, and affordability.
Precision therapies, AI-assisted monitoring, and patient-centric approaches are enhancing outcomes and treatment adherence.
Market growth is influenced by healthcare policies, insurance coverage, and cost-effectiveness of advanced drugs.
Ongoing clinical trials and novel drug combinations are anticipated to sustain long-term market growth.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 32.7 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.7% | 2033 Value Projection: | USD 48.2 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | AbbVie, Johnson & Johnson, Pfizer, Roche, Novartis, Bristol‑Myers Squibb | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rheumatoid Arthritis Treatment Market Growth Factors
The Rheumatoid Arthritis Treatment Market is driven by several growth factors. Rising global RA prevalence, particularly among aging populations, fuels demand for effective therapies. Advancements in biologics, biosimilars, and targeted synthetic DMARDs improve patient outcomes, encouraging adoption. Early diagnosis and increasing awareness of RA symptoms support treatment uptake. Favorable healthcare infrastructure and reimbursement policies in developed regions boost accessibility, while emerging markets benefit from expanding healthcare access. Additionally, ongoing clinical trials, combination therapies, and personalized medicine approaches enhance treatment efficacy. Growing focus on patient-centric care, telemedicine, and digital monitoring further supports sustained market growth through 2033.
Rheumatoid Arthritis Treatment Market Development
In July 2025, Agilus Diagnostics, in collaboration with Sebia, introduced the anti-MCV antibody test in India to aid early diagnosis of rheumatoid arthritis, especially in patients negative for anti-CCP and rheumatoid factor.
Key Players
Leading Companies of the Market
AbbVie
Johnson & Johnson
Pfizer
Roche
Novartis
Bristol Myers Squibb
Key players in the Rheumatoid Arthritis Treatment Market include AbbVie, Johnson & Johnson, Pfizer, Roche, Novartis, Bristol‑Myers Squibb. These companies lead in biologics, biosimilars, and targeted therapies, driving innovation, expanding global presence, and shaping treatment adoption trends across major markets.
Rheumatoid Arthritis Treatment Market Future Outlook
The future outlook of the Rheumatoid Arthritis Treatment Market is highly positive, driven by rising disease prevalence, increasing awareness, and technological advancements. Biologics, biosimilars, and targeted synthetic DMARDs will continue dominating, with personalized medicine and precision therapies improving patient outcomes. Emerging markets in Asia Pacific, Latin America, and the Middle East are expected to grow faster due to expanding healthcare infrastructure and affordability. Digital health solutions, telemedicine, and AI-assisted monitoring will enhance treatment adherence and disease management. Continuous R&D, novel drug launches, and strategic collaborations by leading companies will sustain innovation and market expansion through 2033.
Rheumatoid Arthritis Treatment Market Historical Analysis
The Rheumatoid Arthritis Treatment Market historical analysis shows steady growth over the past decade, primarily driven by increasing RA prevalence and limited treatment options initially dominated by NSAIDs and conventional DMARDs. The launch of biologics in the early 2000s revolutionized treatment, offering targeted therapies with higher efficacy and safety, which accelerated market expansion. Gradually, biosimilars and JAK inhibitors entered the market, enhancing affordability and accessibility. Developed regions like North America and Europe historically accounted for the largest share due to advanced healthcare systems, while emerging markets began contributing significantly as awareness, infrastructure, and reimbursement improved, shaping current market dynamics.
Sources
Primary Research Interviews:
Pharmaceutical & Biotech Companies
Healthcare Professionals
Distributors & Wholesalers
Regulatory & Policy Experts
Databases:
PubMed / Medline
ClinicalTrials.gov
World Health Organization
Journals:
Arthritis & Rheumatology
Annals of the Rheumatic Diseases
Rheumatology
Clinical Rheumatology
Newspapers:
The Wall Street Journal
Financial Times
The Guardian
The New York Times
Associations:
American College of Rheumatology (ACR)
European Alliance of Associations for Rheumatology (EULAR)
Arthritis Foundation (USA)
International Rheumatology Association (IRA)
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients