Global genome engineering market is estimated to be valued at USD 8.85 Bn in 2026 and is expected to reach USD 23.39 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 14.9% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Global genome engineering market trends have shown significant advancements in techniques such as CRISPR, TALENs, and ZFNs. These new technologies have streamlined the genome editing process, thus, allowing for increased efficiency and precision. Reduction in time and costs involved has led to wider adoption of genome engineering across various industry verticals such as biotechnology and pharmaceutical companies. The applications of genome editing continue to grow as well with active research being done for treating genetic diseases, developing genetically modified crops, and even biological weapon deterrence programs. As the technology evolves further and matures, it is anticipated that genome engineering will transform multiple sectors of the economy globally.
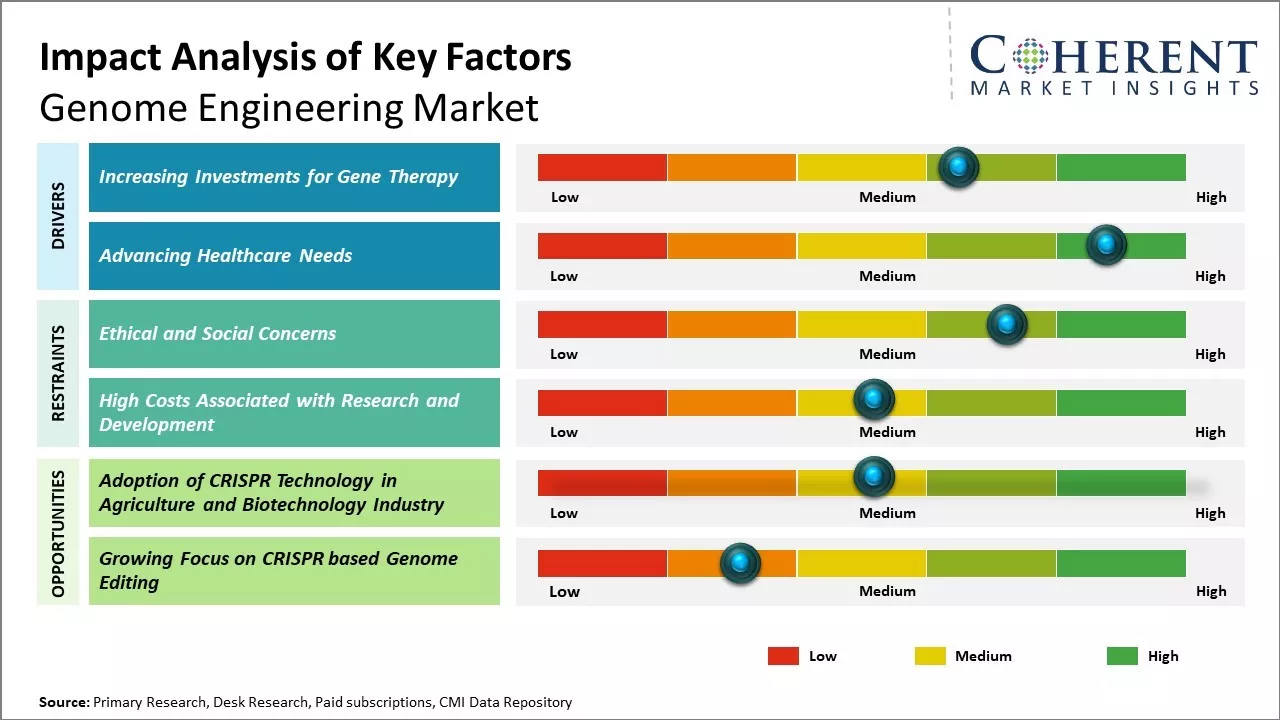
Increasing Investments for Gene Therapy
Increasing investments for CRISPR-based gene therapy development is expected to drive the market growth over the forecast period. For instance, in October 2023, National Institutes of Health (NIH) announced that it had granted US$ 40 million of funding to Yale School of Medicine, Medical school in Connecticut, U.S. to support the development of a gene-editing platform, capable of reaching the human brain. The innovative new genome-editing technology, which was developed from the first phase of the NIH Common Fund Somatic Cell Genome Editing (SCGE) program, could potentially lead to treatments or cures for many neurogenetic diseases.
Get actionable strategies to beat competition: Request Free Sample
Advancing Healthcare NeedsRising global healthcare expenditure leads to growing demand for more effective and affordable medical solutions. As population ages and chronic diseases become more prevalent worldwide, there is immense pressure on healthcare systems to innovate. Genome engineering technology represents a major opportunity to address some of the biggest challenges facing public health. Nearly 8,000 rare genetic disorders have been identified that currently lack satisfactory treatment options. Diseases like cancer, heart disease, diabetes and neurological disorders like Alzheimer’s continue to take a major human and economic toll.

To learn more about this report, Request Free Sample
Market Challenge – Ethical and Social ConcernsEthical and societal concerns surrounding gene editing technologies can hamper the market growth. The lack of consensus around appropriate regulation and governance can hamper the market growth. Companies and researchers are wary of developing applications that could invite public backlash or regulatory issues down the line. The regulatory environment remains ambiguous and complex involving multiple agencies. This policy uncertainty creates hurdles for companies to confidently invest in long-term R&D initiatives.
Market Opportunity – Adoption of CRISPR Technology in Agriculture and Biotechnology Industry
The adoption of CRISPR technology in agriculture and biotechnology industry provides a huge opportunity for growth of genome engineering market. CRISPR is revolutionizing how one modify genomes and has the potential to help feed the billions. CRISPR can help address this issue by enabling more precise and efficient breeding of climate-resilient and nutritious crop varieties. It allows for more targeted introductions of beneficial traits and faster development of new hybrid varieties as compared to conventional breeding methods. This ability to precisely edit plant genomes can help farmers to boost yields while using less water, land and agrochemicals to produce food.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
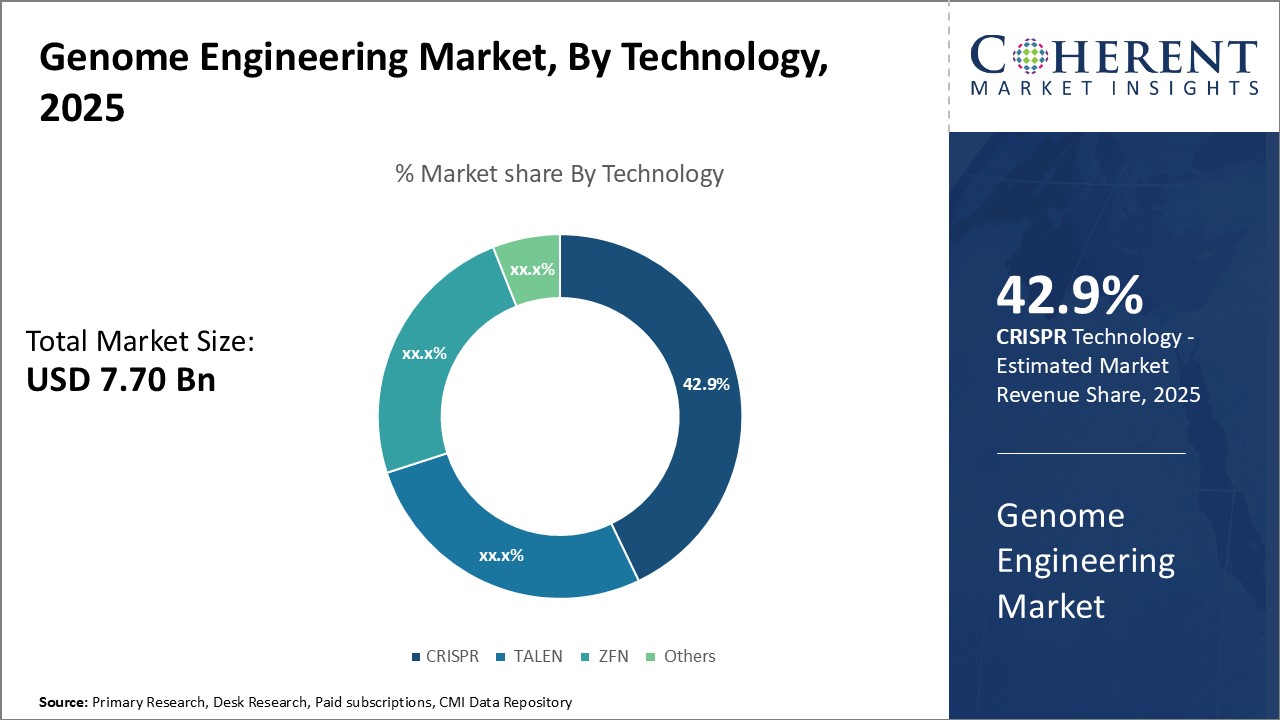
Insights, By Technology: Precision and affordability boosts adoption of CRISPR technologyTechnology segment is sub-segmented into CRISPR, TALEN, ZFN, others. CRISPR segment is estimated to hold 43.9% of the market share in 2026, owing to its precision and cost-effectiveness relative to other technologies. CRISPR uses a dual-RNA structure to target specific segments of genomic DNA with unprecedented accuracy. By utilizing a customized CRISPR-associated protein Cas9 along with programmable RNA molecules, scientists can efficiently edit genomes at the exact loci of interest. The simpler design and delivery of CRISPR components has significantly reduced the cost per genome editing experiment. Researchers can perform high-throughput screens and multiplexed gene edits for a fraction of previous prices. Both the precision and affordability of CRISPR have accelerated its adoption across scientific fields including developmental biology, immunology, and disease modeling. CRISPR's clear advantages over other tools continue drives its leadership position within the genome engineering market.
To learn more about this report, Request Free Sample
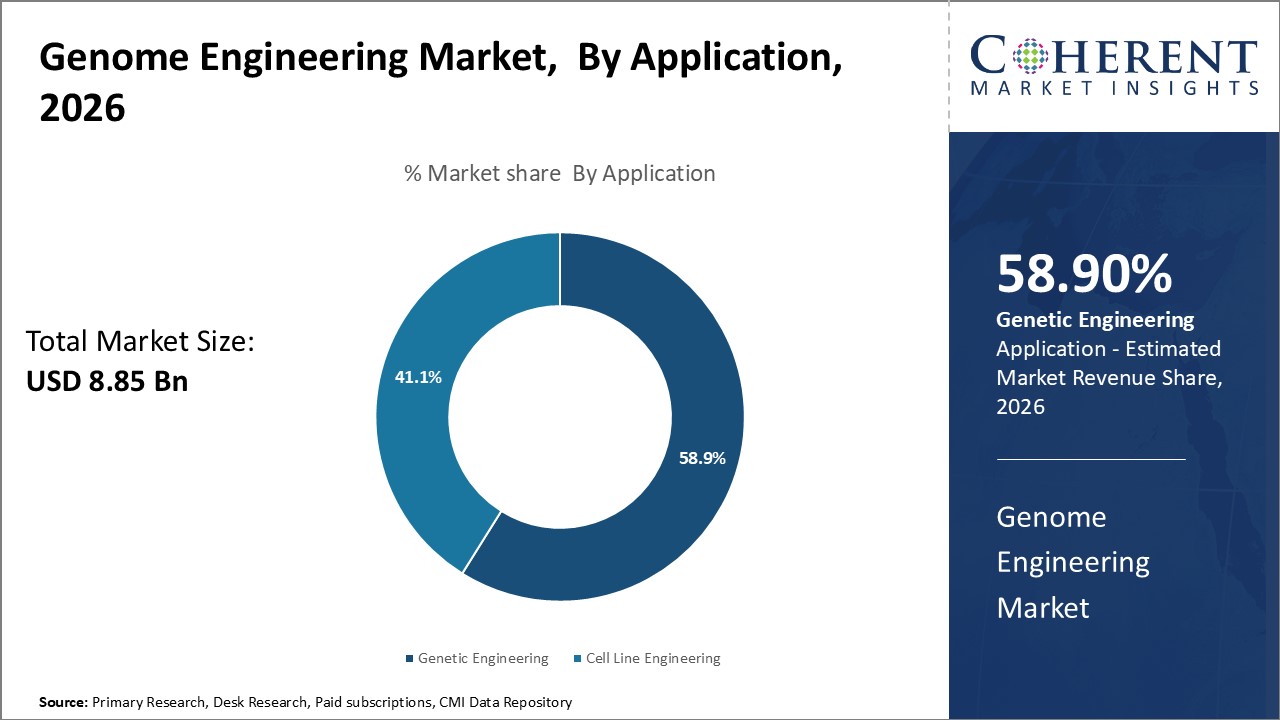
Insights, By Application: Genetic engineering drives application in biotechnology and healthcare
Application segment is sub-segmented into cell line engineering and genetic engineering. Genetic engineering segment is estimated to hold 58.9% of the market share in 2024, due to its diverse and impactful uses across biotechnology and healthcare. Genetic engineering utilizes tools like CRISPR to modify organisms for improved traits, create new cell lines, and advance therapeutics development. In agriculture, gene editing enhances crop yields, disease resistance, and nutritional content. Manufacturers produce novel biological products like enzymes, antibiotics, biofuels through genetic engineering of microbial cell lines. Genetic engineering plays a pivotal role in developing advanced cell and gene therapies. Modifying immune cells or adding corrective genes holds promise for curing numerous diseases. Overall, genetic engineering applications span multiple high-growth industries and have potential to revolutionize fields like pharmaceuticals, agriculture, and manufacturing. Its diverse impact fuels extensive research and commercialization, ensuring genetic engineering's dominance in application segment.
Insights, End User: Increase in collaborations between biopharmaceutical companies for the advancements in gene editing
End User segment is sub-segmented into biotechnology & pharmaceutical companies, research institutes, contract research organizations. Biotechnology & pharmaceutical companies segment is estimated to hold 42.2% of the market share in 2026, due to increasing collaborations between biopharmaceutical companies for the advancements in gene editing. For instance, in September 2023, Ginkgo Bioworks, a U.S.-based biotechnology company, announced a collaboration with Pfizer Inc., a U.S.-based multinational pharmaceutical and biotechnology firm. Through this collaboration, Pfizer will use Ginkgo’s proprietary RNA technology to advance the discovery and development of novel RNA molecules across priority research areas. Ginkgo will receive an upfront payment and will be eligible to receive research fees and development and commercial milestone payments, up to an aggregate total of US$ 331 million across three programs. Ginkgo is entitled to potential further downstream value in the form of royalties on sales.
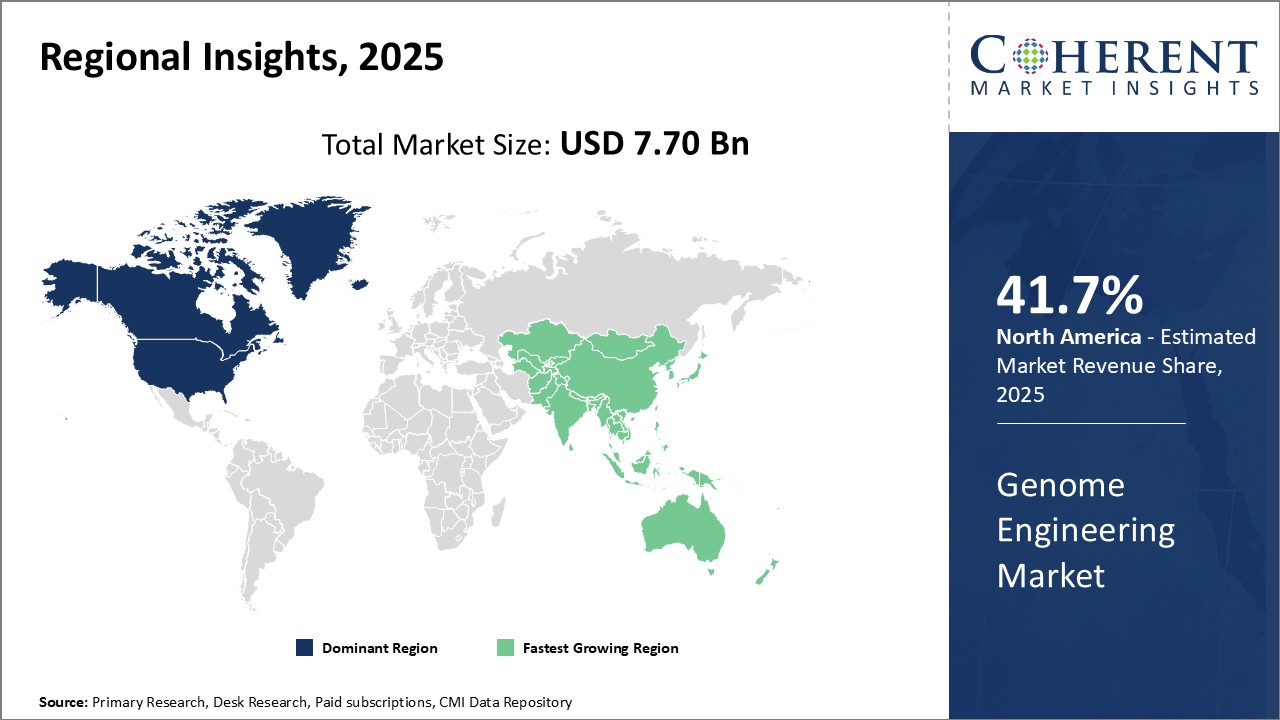
Need a Different Region or Segment? Request Free Sample
North America remains the dominant region in the global genome engineering market and is estimated to hold 41.7% of the market share in 2026, owing to strong presence of leading biotechnology and pharmaceutical companies in the U.S. and Canada. The region is home to prominent genome engineering technology developers and service providers who have been undertaking significant R&D activities in the field. Increasing government funding for genomics research and presence of advanced healthcare infrastructure can drive the market growth. Many top universities in the U.S. and research institutes have played a pivotal role in advancing genome engineering capabilities through innovative projects.
Asia Pacific has emerged as the fastest growing regional market. The market growth is driven by rising healthcare investments, increasing focus on genomic research, and growing bioeconomy in major countries. China and India have witnessed rapid development of their genomics capabilities in the recent past. Considerable funding from public and private sectors for building genomics infrastructure and talent pool has created conducive environment for market expansion. Furthermore, several global players have established manufacturing and research centres in Asia to leverage low-cost skilled resources and grow their regional footprint. Countries like South Korea, Japan, Singapore, and Australia are also actively investing in genome engineering research and technologies.
Genome Engineering Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.85 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 14.9% | 2033 Value Projection: | USD 23.39 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
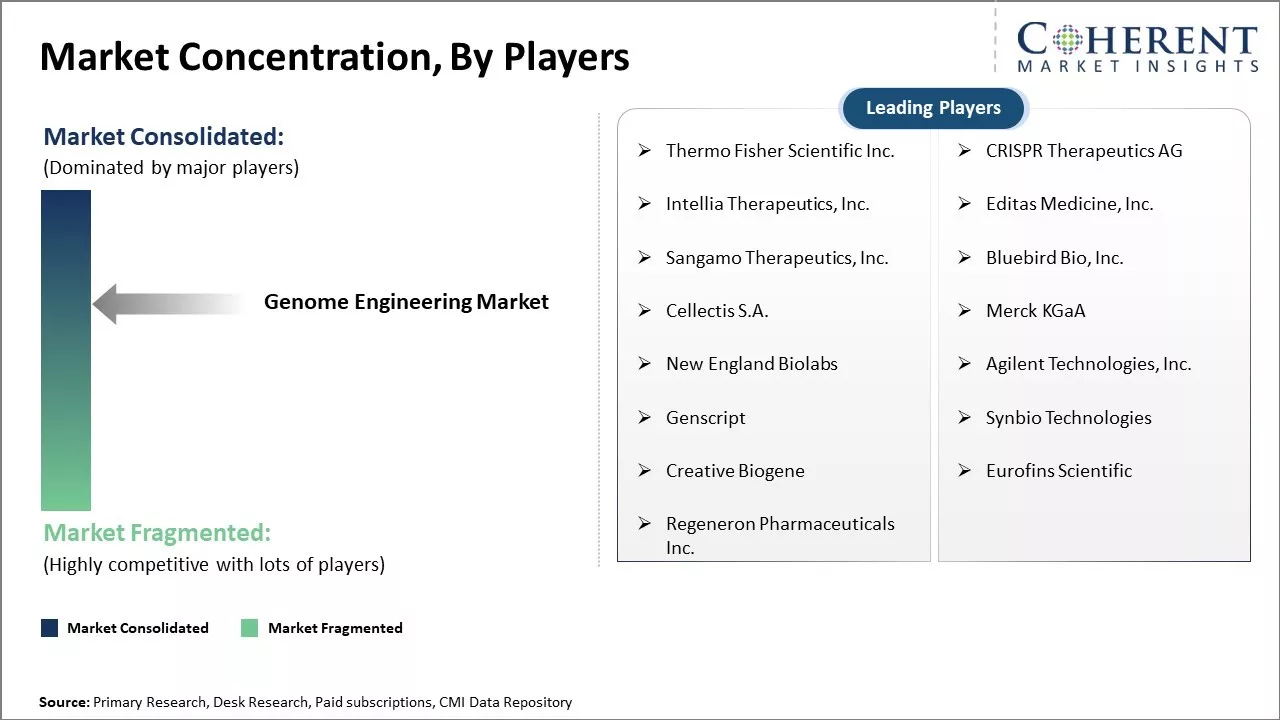
| Companies covered: |
Thermo Fisher Scientific Inc., CRISPR Therapeutics AG, Intellia Therapeutics, Inc., Editas Medicine, Inc., Sangamo Therapeutics, Inc., Bluebird Bio, Inc., Cellectis S.A., Merck KGaA, New England Biolabs, Agilent Technologies, Inc., Genscript, Synbio Technologies, Creative Biogene, Eurofins Scientific, Regeneron Pharmaceuticals Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients