Genomic biomarkers are indicators based on gene expression, gene mutation, gene regulation, and other gene characteristics that can be measured for clinical diagnosis. Genomic biomarkers can be used to detect neurological, oncological, and cardiovascular diseases. Genomic biomarkers help in early detection of a disease, in monitoring progress of the disease, and also assessing the biological response towards a drug treatment or therapy. Genomic biomarkers can be used either as a standalone diagnostic indicator or used in a combination with other biomarkers such as protein biomarkers. Some of the genomic biomarkers used for clinical diagnosis include mRNA, miRNA, cell-free DNA, long coding RNA, and others.
Global Genomic Biomarker Market - Impact of the Coronavirus (COVID-19) Pandemic
Coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19, as pandemic on March 11, 2020.
According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 177,866,160 cases and 3,857,974 deaths due to coronavirus disease (COVID-19) were reported till June 20, 2020 across the globe.
Impact of COVID-19 on Demand and Supply of Genomic Biomarker
The COVID-19 pandemic and lockdown in various countries across the globe have impacted the financial status of businesses across all sectors. The private healthcare sector is one such sector, which has been majorly impacted by the pandemic.
The COVID-19 pandemic has impacted the entire supply chain of the biotechnology industry mainly due to strict lockdown in several regions. The coronavirus pandemic has impacted development, production, and supply of healthcare products and affected businesses of various companies operating in the healthcare sector across the globe. The pandemic has led to the closure of industrial establishments, except manufacturing of essential commodities, and disruption in supply chain of healthcare products.
However, the rising incidences of cardiovascular diseases, arthritis, and neurological diseases associated with COVID-19 infection is expected to have a positive impact on the market growth of genomic biomarker as genomic biomarkers are used for detection of such diseases. According to an article published by the National Centers for Biotechnology Information in January 2021, a study conducted in Wuhan, China, revealed that out of 221 COVID-19 positive patients, approximately 5% had ischemic stroke and 0.5% had hemorrhagic stroke. In another study from the same region, out of 214 COVID-19 positive patients, about 5.7% people with severe COVID infection were reported with acute cerebrovascular disease.
Thus, impact of the coronavirus (COVID-19) pandemic is expected to aid the growth of the global genomic biomarker market during the forecast period, owing to the need for reliable genomic biomarkers as a diagnostic tool for detection of comorbid diseases associated with COVID-19 infection.
The global genomic biomarker market is estimated to be valued at US$ 4,396.6 Mn in 2021, and is expected to exhibit a CAGR of 9.5% over the forecast period (2021-2028).
Genomic Biomarker Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 4,396.6 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 9.5% | 2028 Value Projection: | US$ 8,298.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, F. Hoffmann-La Roche AG, Liquid Genomics, Inc., AROS Applied Biotechnology A/S, Myriad Genetics, Inc., QIAGEN, Eurofins Scientific, Genomic Health, Bio-Rad Laboratories, Inc., Epigenomics AG, and Aepodia |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 1: Global Genomic Biomarker Market Share (%) Analysis, By Indication, 2021
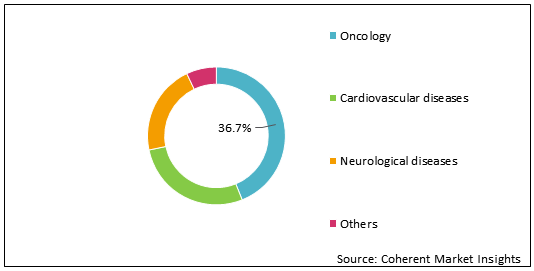
To learn more about this report, Request Free Sample
The increasing burden of chronic diseases is the ma jor factor that is expected to drive the market growth of genomic biomarker over the forecast period.
The rising burden of chronic diseases such as stroke, heart attack, cancer, arthritis, and others are expected to drive growth of genomic biomarker market over the forecast period. According to the data provided by the International Agency for Research on Cancer (IRAC) in 2018, it is estimated that there will be more than 27.5 million new cancer cases by 2040 majorly due to factors such as unhealthy lifestyle, smoking, and reduced physical activities. Also, according to the statistics provided by the World Health Organization in 2018, over 795,000 people have stroke each year in the U.S. This is expected to create opportunities for genomic biomarker market as a clinical diagnostic tool and aid in the development of personalized therapy that will further propel the growth of global genomic biomarker market.
Moreover, the increasing number of research and development activities for identification of novel genomic biomarkers and achieving better specificity associated with the use of genomic biomarker is expected to aid in the growth of the market over the forecast period. For instance, according to an article published by the Intestinal Research Journal in November 2019, a meta-analysis of 1,258 patients suffering from colorectal cancer and 803 healthy people as control, revealed that the use of circulating (cell-free) cfDNA as a genomic biomarker for the diagnosis of colorectal cancer showed 91.8% specificity. This specificity was found to be higher than the genomic biomarker diagnosis using absolute DNA concentration. sSuch new revelations in the field of genomic biomarkers is expected to drive its market growth.
Technological advancements in the field of healthcare and genomics is expected to drive the growth of the global genomic biomarker market over the forecast period. For instance, in November 2019, Exact Sciences Corp., an American molecular diagnostics company, received the U.S Food and Administration’s ‘breakthrough device’ designation for its blood-based diagnostic test for liver cancer. This test evaluates two protein biomarkers and four methylated DNA biomarkers for the detection of hepatocellular carcinoma with a high specificity of 90% and sensitivity of 80%. Moreover, in May 2020, Qiagen, a German molecular diagnostics technology provider, announced the launch of technologically advanced solutions for genomic analysis in different types of cancers. The novel QIAseq pan-cancer multimodal panel utilizes unique QIAseq multimodal technology to identify and enrich more than 600 DNA and RNA biomarkers for the assessment of tumor mutational burden and microsatellite instability status from a single sample input.
Global Genomic Biomarker Market – Restraints
However, certain drawbacks associated with the utility and analysis of genomic biomarkers is expected to hamper the growth of its market over the forecast period. For instance, according to an article published by the Intestinal Journal in November 2019, some genomic biomarkers such as miRNA present in stool sample cannot be used as a standalone diagnostic tool for the detection of colon cancer, due to concerns regarding stability of biomarker and failure to establish adequate predictive value for the diagnosis of colon cancer.
Global Genomic Biomarker Market – Regional Analysis
On the basis of region, the global genomic biomarker market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa.
North America is expected to hold dominant position in the global genomic biomarker market over the forecast period, owing to involvement of market players in inorganic market strategies such as collaborations, partnerships, acquisitions, and others. For instance, in August 2020, GeneCentric Therapeutics Inc., an American genomic technology provider, announced its collaboration with Janssen Research & Development, LLC (an American pharmaceutical company) to develop a novel non-invasive RNA-based genomic biomarker for the diagnosis of bladder cancer.
Key players are focused on launching new products and receiving approvals (from regulatory bodies) for their products indicated for diagnosis of various diseases such as cancer, stroke, and arthritis, which is expected to drive growth of the market in North America and Europe. For instance, in May 2021, Qiagen, a German molecular diagnostic technology provider, launched its first U.S FDA approved diagnostic kit for the detection of KRAS mutations in non-small cell lung cancer (NSCLC) patients. KRAS mutations act as a genomic biomarker for cancer detection and also help in checking the eligibility of patients for undergoing treatment therapy with a newly developed Lumakras therapy developed by Amgen Inc.
Figure 2: Global Genomic Biomarker Market Value (US$ 1,710.3 Mn), by Region, 2021
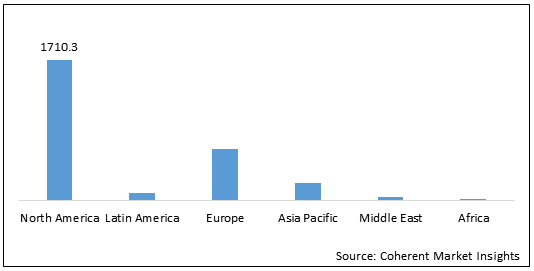
To learn more about this report, Request Free Sample
Global Genomic Biomarker Market – Competitive Landscape
Major players operating in the global genomic biomarker market include Thermo Fisher Scientific, F. Hoffmann-La Roche AG, Liquid Genomics, Inc., AROS Applied Biotechnology A/S, Myriad Genetics, Inc., QIAGEN, Eurofins Scientific, Genomic Health, Bio-Rad Laboratories, Inc., Epigenomics AG, and Aepodia
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients