Glaucoma Eye Drops Market is estimated to be valued at USD 2,062.0 Mn in 2026 and is expected to reach USD 3,420.9 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.5% from 2026 to 2033.
Analysts’ Views on Global Glaucoma Eye Drops Market:
Increasing number of product launches are expected to boost the growth of the global glaucoma eye drops market over the forecast period. For instance, in December 2022, ENTOD INTERNATIONAL- a research-based ophthalmic, ENT & dermatology pharmaceutical company, announced the launch of a new division for the Glaucoma treatment segment to expand its business.
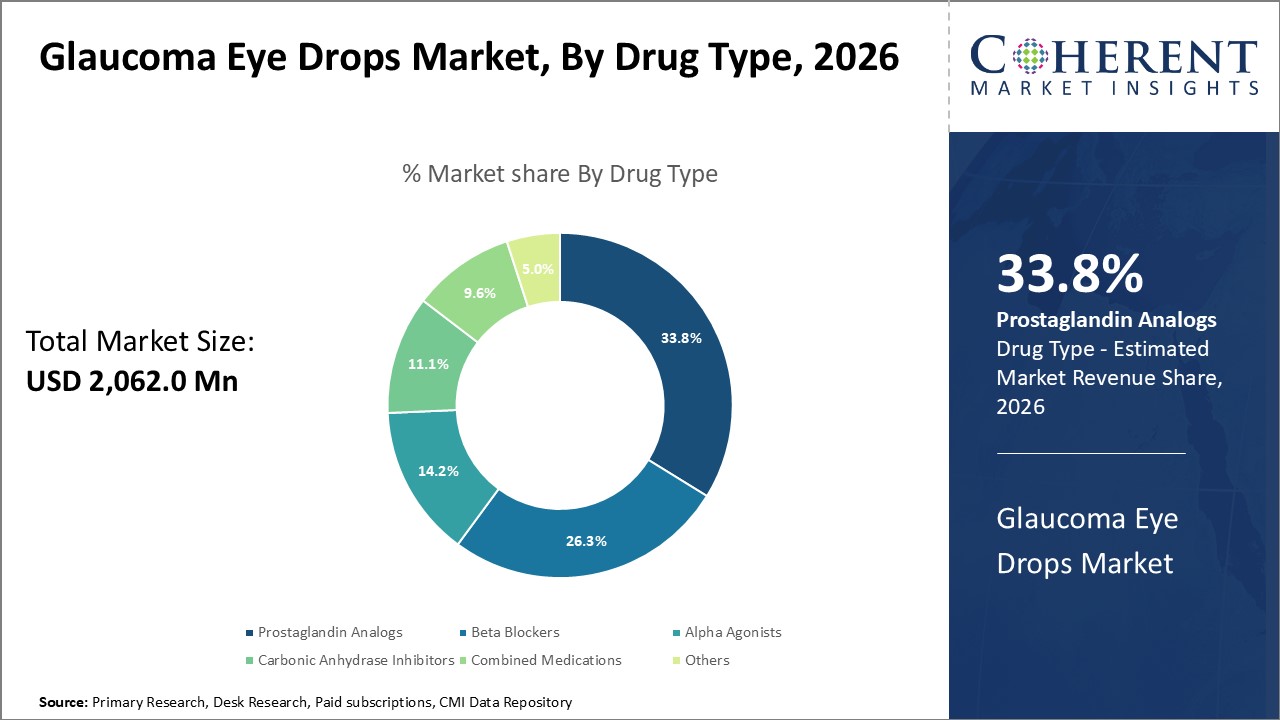
Figure 1. Global Glaucoma Eye Drops Market Share (%), by Drug Type, 2026
To learn more about this report, Request Free Sample
Global Glaucoma Eye Drops Market – Driver
Increasing product launches by key market players
The growing product launches by biopharmaceutical companies are a major factor leading to the high demand for glaucoma eye drops. For instance, on May 9, 2023, Upsher-Smith Laboratories, LLC- a U.S-based biopharmaceutical company, launched brimonidine tartrate and timolol maleate ophthalmic solution, 0.2%/0.5%. The eye drop lowers eye pressure in patients with open-angle glaucoma.
Increasing mergers, acquisitions, and partnership scenarios in the market
The increasing mergers, acquisitions, and partnership scenarios can drive the growth of the global glaucoma eye drops market. For instance, in October 2022, Oculis- a global biopharmaceutical company specializing to safeguard sight and improve eye care, and European Biotech Acquisition Corp- a business equity firm, announced a business combination agreement to create a new biopharmaceutical company driving innovations in ophthalmology.
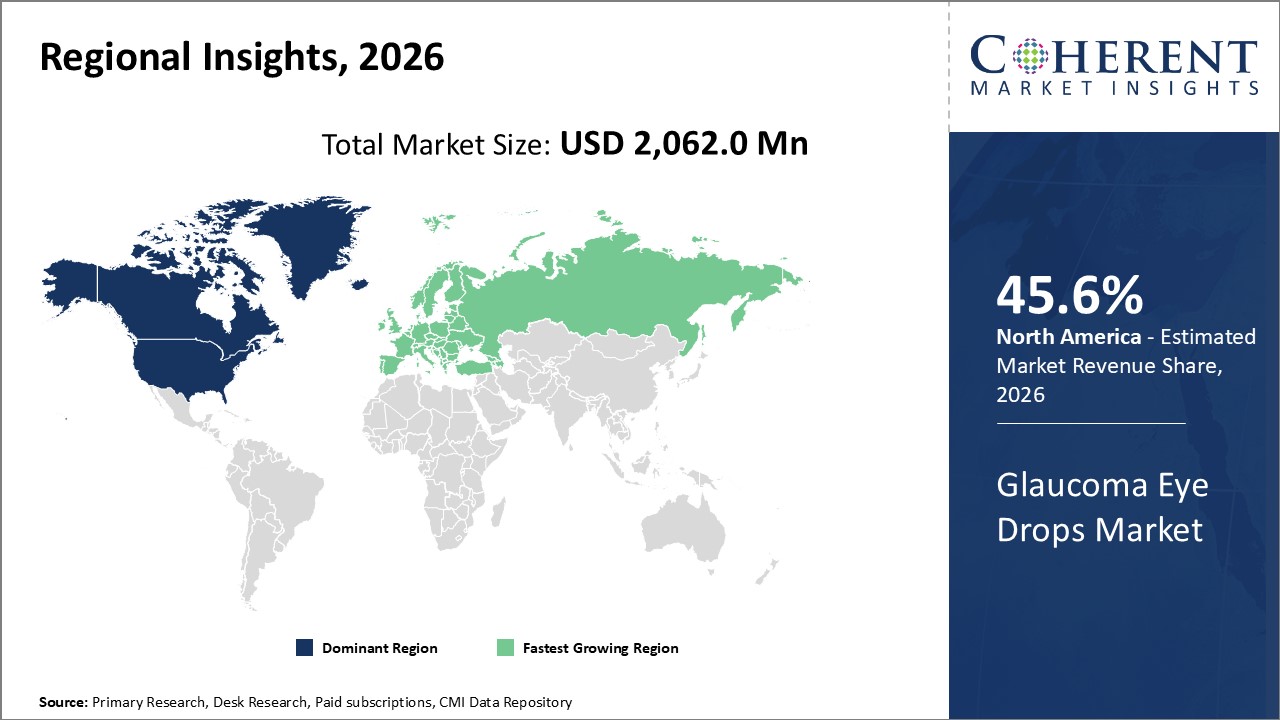
Figure 2. Global Glaucoma Eye Drops Market Share (%), by Region, 2026
To learn more about this report, Request Free Sample
Global Glaucoma Eye Drops Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global glaucoma eye drops market over the forecast period. This is due to the increasing number of partnerships and campaigns launched to raise awareness about glaucoma in this region. For instance, on January 3, 2026, Bausch + Lom- an eye health products company based in Vaughan, Ontario, Canada, and Glaucoma Research Foundation- an organization dedicated to developing a cure for glaucoma through innovative research, collaboration, and education, partnered together to launch the Screen, Protect, Cure campaign in recognition of glaucoma awareness month. A new educational campaign aims to educate individuals who may be at risk for glaucoma and raise funding for glaucoma research.
This is because ophthalmologists were urged to discontinue non-urgent care. Since ophthalmic visits and surgeries are usually for non-life-threatening conditions, According to an observational retrospective study that randomly selected 200 glaucoma patients with more than 3 months of unintended delay for their post-COVID visit, published in PubMed, on April 25, 2023, significantly higherIntra-ocular
Glaucoma Eye Drops Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2,062.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 3,420.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck KGaA, Allergan plc ( A subsidiary of AbbVie Inc.), Novartis AG, Teva Pharmaceutical Industries Ltd., Pfizer, Inc., Bausch & Lomb, Inotek, Jadran-galenski laboratorij d.d. JGL d.d, Aerie Pharmaceutical (A subsidiary of Alcon Management S. A) and Mylan N.V (A subsidiary of Viatris Inc.) |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Glaucoma Eye Drops Market Segmentation:
The global glaucoma eye drops market report is segmented into drug type, disease indication, distribution channel, composition, and region.
By Drug Type, the market is segmented into Prostaglandin analogs, Beta blockers, Alpha agonists, Carbonic Anhydrase Inhibitors (CAIs), Combined Medications, and Others (rho-kinase inhibitors, cholinergic agonists). Out of which, the Prostaglandin analogs segment is expected to hold a dominant position in the global glaucoma eye drops market during the forecast period and this is attributed to their ability in lowering intraocular pressure (IOP), a major risk factor for glaucoma.
By Disease Indication, the market is segmented into Angle-closure Glaucoma and Open-angle Glaucoma. Out of which, the Open-angle Glaucoma segment is expected to hold a dominant position in the global glaucoma eye drops market during the forecast period and this is attributed to the rising prevalence of the disease globally.
By Distribution Channel, the market is segmented into Hospital Pharmacy Retail Pharmacy, and Online Pharmacy. Out of which, the Hospital Pharmacy segment is expected to dominate the market over the forecast period and this is attributed to the increasing hospitalizations caused by glaucoma all over the world
By Composition, the market is segmented into Preservative Free, BAK-Based, and Non BAK-Based Preservatives. Out of which, the BAK-Based segment is expected to dominate the market over the forecast period and this is attributed to its effective bactericidal and fungicidal properties.
By Region, the market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, the North America segment is expected to dominate the market over the forecast period and this is attributed to the increased R&D activities regarding glaucoma eye drops in the region.
Among all the segmentations, the disease indication segment has the highest potential due to the increasing prevalence of eye diseases and disorders across the world over the forecast period. For instance, according to an article published in January 2020, by PubMed- a free search engine for biological databases and medical reports, an estimated 76 Mn people worldwide are affected by primary open-angle glaucoma in 2020.
Global Glaucoma Eye Drops Market Cross Sectional Analysis:
Among disease indication, open-angle glaucoma segment is expected to drive the market in Europe region. For instance, in October 2022, Nicox- a France-based ophthalmology company developing treatments to maintain vision and improve ocular health, announced that once a day dosing of their drug candidate NCX 470 1.1% caused a lowering of intraocular pressure (IOP) compared to the standard, latanoprost 1.005%, in the Phase 3 of clinical trial of the drug in patients with open-angle glaucoma or ocular hypertension.
Global Glaucoma Eye Drops Market: Key Developments
In November 2022, Alcon Management S. A.- an eye care company, completed the acquisition of Aerie Pharmaceuticals, Inc.- an ophthalmic pharmaceuticals company, to strengthen the company’s ophthalmic pharmaceutical business.
In July 2022, Novartis AG- a Switzerland-based multinational pharmaceutical corporation, announced that it has acquired Kedalion Therapeutics- developer of ophthalmic therapies, and its AcuStream technology, a device that may have the potential to facilitate precise dosing and accurate delivery of certain topical ophthalmic medications to the eye to treat chronic diseases like glaucoma.
In June 2022, Amring Pharmaceuticals Inc. - a generic and brand pharmaceutical company, announced that it had received FDA approval for generic Timolol Maleate Ophthalmic Solution, USP 0.5%. The eye drops are indicated for the treatment of elevated intraocular pressure in patients with ocular hypertension or open-angle glaucoma.
In April 2022, A Japan-based post marketing observational study confirmed the safety and efficacy of omidenepag isopropyl ophthalmic solution 0.002% (OMDI), a topical drug used to treat elevated intraocular pressure (IOP) in patients with glaucoma or ocular hypertension
Global Glaucoma Eye Drops Market: Key Trends
The increasing approvals by drug regulatory agencies across the globe with regard to glaucoma eye drops offer lucrative opportunities for the growth of the market over the forecast period. For instance, Visox Pharma- a privately funded biopharmaceutical company focused on the development and commercialization of ophthalmic drugs to address highly prevalent diseases in need of new treatment options, announced that it had received notification from the U.S. Food and Drug Administration (FDA) that the agency had completed its filing review and accepted for filing the New Drug Application (NDA) for PDP-716 (0.35% brimonidine tartrate) for the treatment of glaucoma.Visiox Pharma
In October 2022, Santen Pharmaceutical Co., Ltd. - a Japan-based pharmaceutical company, announced that the FDA approved OMLONTI, the company’s newest ophthalmic medication. This ophthalmic solution is composed of 0.002% omidenepag isopropyl. This drug will lower intraocular pressure (IOP) for patients with ocular hypertension or open-angle glaucoma.
Global Glaucoma Eye Drops Market: Restraints
Product recalls
Increasing product recalls due to faults in manufacturing and production can restrain the growth of the global glaucoma eye drops market. For instance, Apotex Inc. - a Canada-based pharmaceutical corporation, announced the voluntary recall of 6 lots of Brimonidine Tartrate Ophthalmic Solution, 0.15% due to cracks found in the unit caps of the solution bottles. The prescription drops are used for patients with open-angle glaucoma or ocular hypertension.
This restrain can be overcome by strict compliance with regulatory guidelines and the adoption of good manufacturing practices during the production process.
Side effects of eye drops used to treat glaucoma
The potential side effects of medications used to treat glaucoma can restrain the growth of the global glaucoma eye drops market. For instance, according to an article published in January 2022, by PubMed- a free biological database for medical reports, a class of medications called cholinergic agents, (used to treat glaucoma) such as pilocarpine, can cause reported side effects including headache, urinary retention, small pupils, and blurred vision.
This can be overcome by restricting the dose of the drug as recommended by the physician.
Global Glaucoma Eye Drops Market - Key Players
Major players operating in the global glaucoma eye drops market include Merck KGaA, Allergan plc ( A subsidiary of AbbVie Inc.), Novartis AG, Teva Pharmaceutical Industries Ltd., Pfizer, Inc., Bausch & Lomb, Inotek, Jadran-galenski laboratorij d.d. JGL d.d, Aerie Pharmaceutical (A subsidiary of Alcon Management S. A) and Mylan N.V (A subsidiary of Viatris Inc.)
*Definition: Glaucoma is a multifactorial optic degenerative neuropathy characterized by the loss of retinal ganglion cells. It is a combination of vascular, genetic, anatomical, and immune factors. Glaucoma poses a significant public health concern as it is the second leading cause of blindness after cataracts, and this blindness is usually irreversible.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients