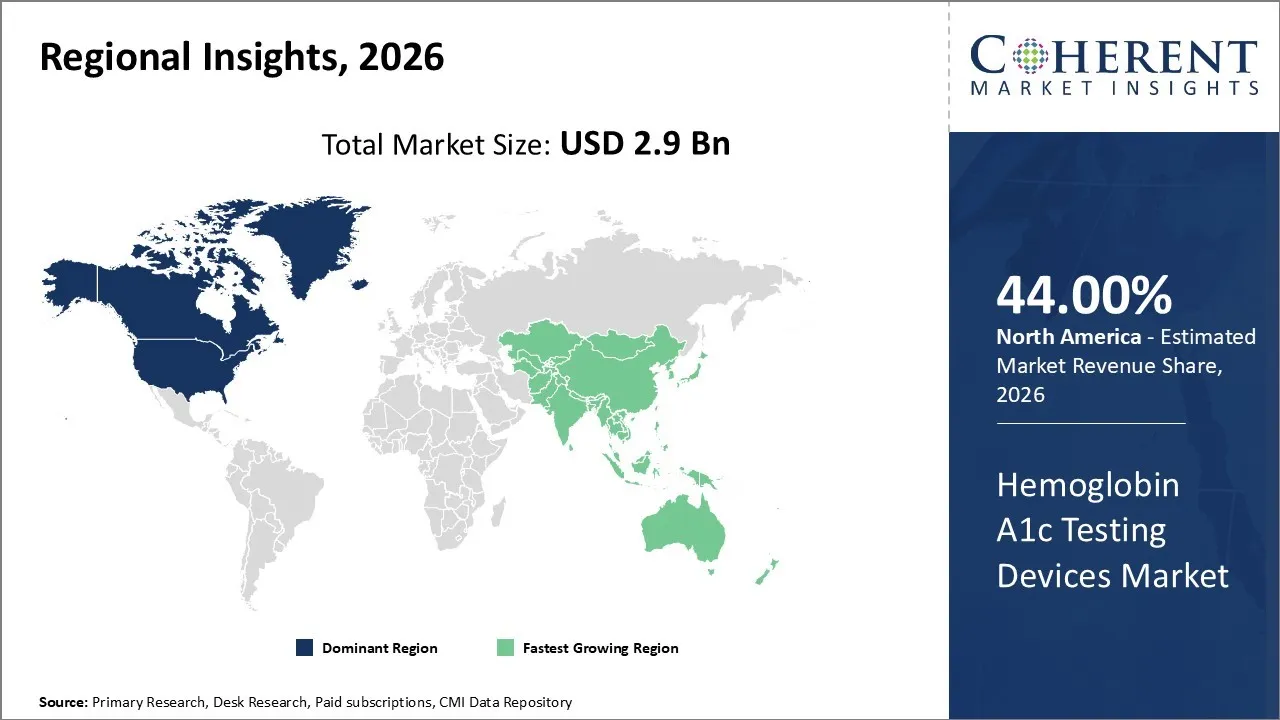
The hemoglobin A1c testing devices market was valued at USD 2.9 Bn in 2026 and is forecast to reach a value of USD 4.18 Bn by 2033 at a CAGR of 7.5% between 2026 and 2033.
Hemoglobin A1c test is a common blood test performed for diagnosis and monitoring of type 1 and type 2 diabetes. A1c test results reflect the average blood sugar levels of an individual for the past two to three months. Hemoglobin (Hb) is a protein that links with sugars such as glucose found inside red blood cells. High amount of glucose linked (glycated) with hemoglobin molecules increases the risk of diabetes. HbA1c test reports the amount of HbA1c as a proportion of total hemoglobin in the blood stream.
|
Current Event |
Description and its Impact |
|
Technological Advancements in A1c Testing Devices |
|
|
Policy and Healthcare Initiatives Supporting Diabetes Management |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of test type, the laboratory testing devices segment is expected to lead the market with 65% share in 2026. Laboratory testing for Hemoglobin A1c remains the gold standard for diabetes diagnosis and management, as it offers consistent as well as precise quantification of glycosylated hemoglobin, which reflects average blood glucose levels over the previous two to three months. The dominance of Laboratory Testing Devices is further reinforced by their extensive integration into healthcare institutions, including hospitals, reference labs, as well as diagnostic centers, which prioritize validated and standardized testing methods to ensure patient safety and diagnostic confidence.
For instance, laboratory-based HBA1c tests typically employ high-performance liquid chromatography (HPLC), immunoassay, or capillary electrophoresis methods, all of which are well-established techniques known for their accuracy as well as minimal variability. The National Glycohemoglobin Standardization Program (NGSP), run by non-profits and supported by groups like the World Health Organization (WHO), works to make lab tests the same everywhere. It makes sure test results match international reference standards.
In terms of technology, the Ion-Exchange HPLC segment is expected to hold 55% share of the market in 2026. Due to its superior accuracy, reproducibility, and established presence in clinical diagnostics. Ion-Exchange HPLC technology separates different types of hemoglobin by their electrical charges using a special column. This method is considered the best way to measure Hemoglobin A1c because it is very accurate. It can also check for different types of hemoglobin that might affect the test, making it reliable for many kinds of patients.
To learn more about this report, Request Free Sample
North America is expected to dominate the Hemoglobin A1c Testing Devices market with 44% share in 2026, primarily owing to advanced healthcare infrastructure, widespread awareness of diabetes management, as well as robust governmental initiatives targeting chronic disease control. The United States, which makes up the major portion of North America’s healthcare system, has implemented comprehensive screening programs such as the National Diabetes Prevention Program (NDPP), focusing early detection and continuous monitoring of diabetes through devices like Hemoglobin A1c testers.
For instance, in September 2025, Orange Biomed said it reached an important step in making a small A1C testing device. The company in Seattle got official approval from Korea to make medical test devices. This allows them to produce more of their small portable diabetes testing device.
Asia Pacific is expected to exhibit the fastest growth, because of a combination of increasing diabetes prevalence, expanding healthcare infrastructure, government initiatives, and rising awareness about early diagnosis and disease management in countries across this subcontinent. Diabetes is recognized by the International Diabetes Federation as being highly concentrated in Asia Pacific, with countries like India and China having some of the largest populations suffering from this chronic issue.
The Indian Council of Medical Research (ICMR) has reported an escalating number of prediabetic and diabetic individuals, which is directly driving the enhanced utilization of Hemoglobin A1c testing devices as a critical diagnostic and monitoring tool.
The U.S. contributes the highest share in the Hemoglobin A1c Testing Devices Market owing to several major factors rooted in its vast healthcare infrastructure, early adoption of advanced diagnostic technologies, as well as robust initiatives aimed at improving diabetes management. The U.S. healthcare system focuses a lot on accurate diabetes testing because many people have diabetes and there is strong awareness about keeping blood sugar under control.
For instance, according to the Centers for Disease Control and Prevention (CDC), over 37 million Americans are living with diabetes, and an estimated 88 million adults have prediabetes, underscoring the massive need for accurate and frequent HbA1c testing.
China contributes the highest share in the Hemoglobin A1c testing devices market, owing to a combination of factors rooted in demographic trends, healthcare infrastructure development, as well as initiatives by government authorities aimed at improving diabetes care. China faces a high burden of diabetes, with the International Diabetes Federation reporting over 140 million adults living with diabetes as of recent years, making it the country with the largest diabetic population worldwide.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.9 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 4.18 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, ARKRAY, Inc., Bio-Rad Laboratories, Inc., Chek Diagnostics (Polymer Technology Systems, Inc.), Danaher Corporation (Beckman Coulter), F. Hoffmann-La Roche Ltd., Menarini Group, Sebia, Siemens Healthcare, Tosoh Corporation, and Trinity Biotech plc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of diabetes worldwide has become a significant catalyst for the burgeoning demand for Hemoglobin A1c (HbA1c) testing devices. HbA1c testing is crucial for the effective management and diagnosis of diabetes, as it provides a reliable measure of average blood glucose levels over the past two to three months. Furthermore, the adoption of these devices in multiple settings, including hospitals, diagnostic laboratories, and home care environments, is increasing as awareness about diabetes management grows.
The increasing awareness about diabetes management has been a significant driving force behind the expansion of the Hemoglobin A1c (HbA1c) testing devices market. With diabetes emerging as a global health challenge, healthcare systems and patients alike are becoming more cognizant of the importance of regular monitoring to prevent complications. The HbA1c test, which provides a three-month average of blood glucose levels, has become an essential diagnostic and management tool, enabling both healthcare providers and patients to track long-term glycemic control efficiently.
The development of portable and user-friendly Hemoglobin A1c (HbA1c) testing devices represents a significant opportunity in the HbA1c testing devices market, driven primarily by the increasing demand for accessible diabetes management tools. Traditionally, HbA1c testing has been conducted in clinical laboratories, which can be time-consuming and less accessible, especially in rural or underserved regions. Portable devices enable patients and healthcare providers to perform testing quickly and accurately at point-of-care or even at home, facilitating timely decision-making and better disease management.
The Hemoglobin A1c (HbA1c) Testing Devices Market is experiencing major momentum driven by the high prevalence of diabetes globally. As diabetes management hinges critically on regular monitoring of blood glucose levels, HbA1c testing devices have become indispensable for both clinical as well as home-based settings.
Major drivers include high awareness about diabetes management, the rising adoption of point-of-care testing, as well as advancements in device technology that provide rapid and accurate results. Also, because more healthcare focuses on patients and preventing diseases, more people want these testing devices.
But the market has problems. Advanced testing machines are expensive, and poor areas often don’t have enough hospitals or clinics, which makes it hard for everyone to use these tests. Furthermore, regulatory challenges and the need for stringent quality control may slow down product launches and approvals.
Opportunities abound in the integration of digital health technologies, including IoT and mobile health apps, which enhance device connectivity and patient monitoring capabilities. Emerging markets in Asia-Pacific, particularly India and China, are currently the fastest-growing due to rising diabetic populations as well as improving healthcare access. Meanwhile, North America continues to dominate the market, supported by established healthcare infrastructure and strong research activities.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients