Hemoglobinopathy Treatment Drugs Market is estimated to be valued at USD 5.74 Bn in 2026 and is expected to reach USD 10.24 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 8.6% from 2026 to 2033.
Analysts’ views on global hemoglobinopathy treatment drugs market:
Increasing prevalence of genetic diseases, new product launches, and strategies like mergers, acquisitions, and collaboration are expected drive the global hemoglobinopathy treatment drugs market growth over the forecast period. For instance, according to the data published by the World Health Organization, on September 7, 2021, between 300,000 and 400,000 babies are born with a serious hemoglobin disorder each year and that up to 90% of these births occur in low- or middle-income countries.
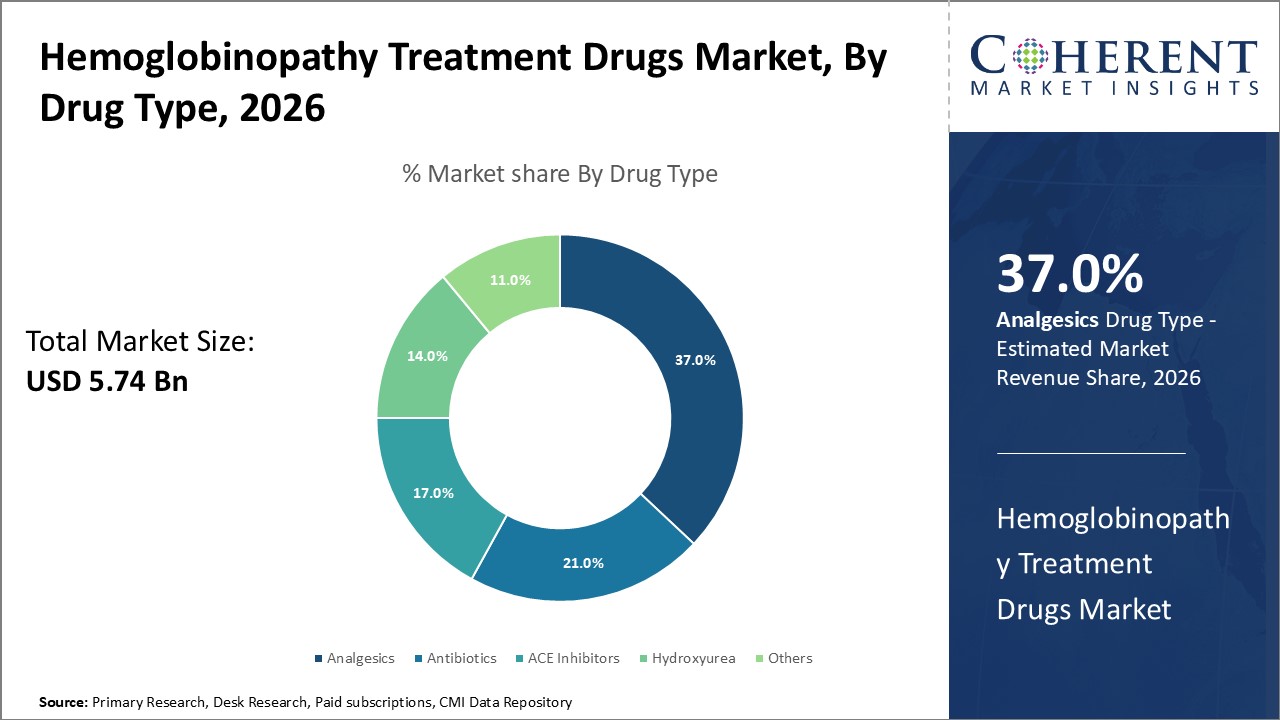
Figure 1. Global Hemoglobinopathy Treatment Drugs Market Share (%), By Drug Type, 2026
To learn more about this report, Request Free Sample
Global Hemoglobinopathy Treatment Drugs Market– Drivers
Increasing research & development activities in hemoglobinopathy treatment
Increasing research & development activities in hemoglobinopathy treatment is expected to drive the global hemoglobinopathy treatment drugs market growth over the forecast period. For instance, on June 16, 2022, researchers at The National Heart, Lung, and Blood Institute (NHLBI), a U.S.-based research organization, announced that activating a protein in red blood cells may improve anemia and alleviate acute episode of severe pain for people living with sickle cell disease. Treatments that stimulate pyruvate kinase (PKR) activity may prevent or alleviate severe pain crisis as PKR deficiency is due to mutation in PKLR gene. A new drug is being develop that can treat patients with inherited pyruvate kinase deficiency. Early phase 1 clinical research trial, showed that activating PKR in patients with SCD through an oral therapy was safe and well tolerated. The researchers will continue to assess the long-term safety and efficacy of this approach with more research. They will specifically look at how this oral therapy may reduce the number of sickling events people experience and also see if it can improve anemia.
Increasing approvals of newer therapies for hemoglobinopathy treatment
Increasing approvals of newer therapies for hemoglobinopathy treatment is expected to drive the global hemoglobinopathy treatment drugs market growth. For instance, on February 17, 2022, Agios Pharmaceuticals, Inc., a U.S.-based pharmaceutical company in the field of cellular metabolism pioneering therapies for genetically defined diseases, announced that the U.S. Food and Drug Administration (FDA) has approved PYRUKYND (mitapivat) in the U.S. for the treatment of hemolytic anemia in adults with pyruvate kinase (PK) deficiency, a rare, debilitating, lifelong hemolytic anemia. PYRUKYND is a first-in-class, oral PK activator and the first approved disease-modifying therapy for this disease. The successful ACTIVATE and ACTIVATE-T studies demonstrate the impact of mitapivat in significantly improving hemolysis and anemia in PK deficiency.
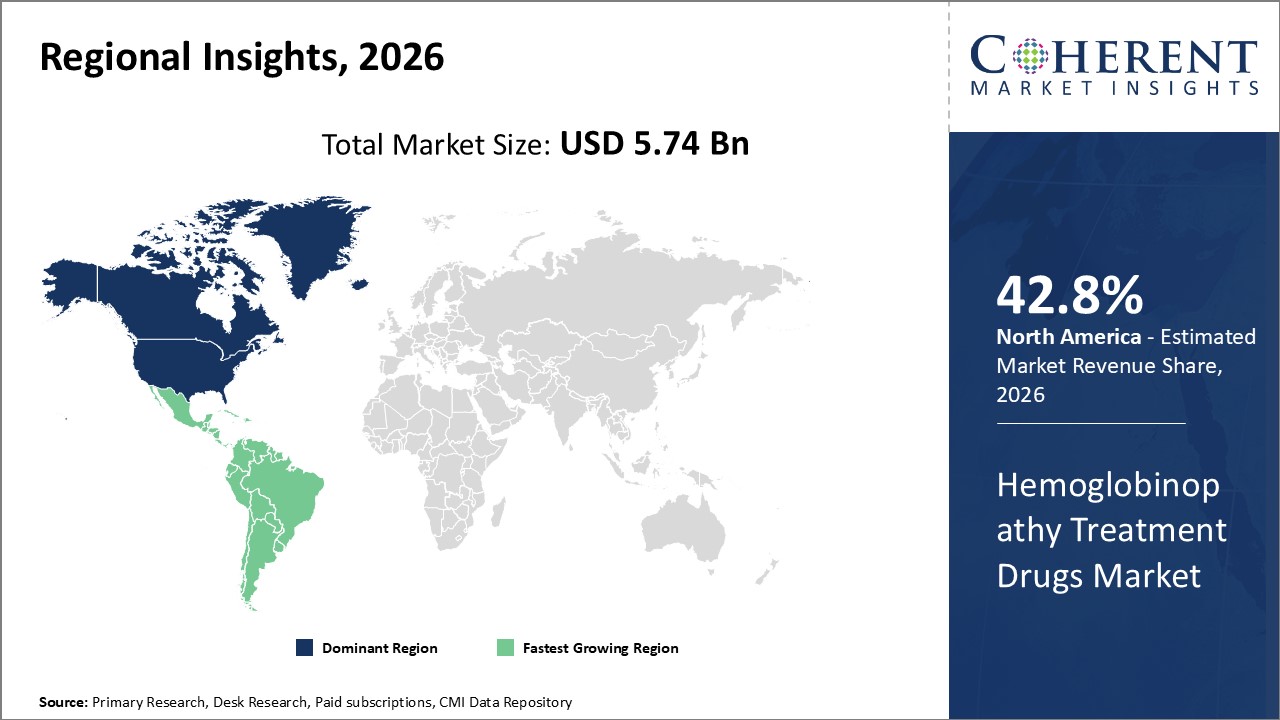
Figure 2. Global Hemoglobinopathy Treatment Drugs Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Hemoglobinopathy Treatment Drugs Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global hemoglobinopathy treatment drugs market over the forecast period, owing to increasing partnerships. For instance, on November 11, 2021, Emmaus Life Sciences, Inc., a U.S.-based commercial-stage biopharmaceutical company and leader in the treatment of sickle cell disease, announced a partnership with UpScript IP Holdings, LLC., a U.S.-based direct to consumer pharmaceuticals provider to offer telehealth solutions to sickle cell disease patients, expanding access to Endari, Emmaus’ prescription L-glutamine oral powder for the treatment of sickle cell disease.
Blood transfusion is required as treatment in hemoglobinopathy treatment.
Global Hemoglobinopathy Treatment Drugs Market Segmentation:
Global hemoglobinopathy treatment drugs market report is segmented into drug type, disease type, distribution channel, and region.
Based on drug type, the global hemoglobinopathy treatment drugs market is segmented into analgesics, antibiotics, ace inhibitors, hydroxyurea, and others. Out of which, the analgesic segment is expected to dominate the market due to increasing product launch.
Based on disease type, the global hemoglobinopathy treatment drugs market is segmented into thalassemia, sickle cell disease, and other Hb variants diseases. Among these, sickle cell disease segment is expected to dominate the market over the forecast period due to increasing treatments for this disease.
Based on distribution channel, the global hemoglobinopathy treatment drugs market is segmented into hospitals pharmacies, retail pharmacies, and online pharmacies. Among these, hospitals pharmacies segment is expected to dominate the market over the forecast period due to increasing number of hospitals pharmacies for obtaining drug.
Based on region, the global hemoglobinopathy treatment drugs market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East, and Africa. Out of which, the North America segment is expected to dominate the market due to increasing products launch in this region.
Among all segmentation, the drug type segment has the highest potential due to increasing research & development activities by the key market players. For instance, on October 19, 2021, The U.S. Food and Drug Administration (FDA) approved a combination pill containing celecoxib and tramadol (Seglentis) for the treatment of adults with acute pain severe enough to require an opioid analgesic and for which alternative treatments fail to provide adequate pain relief. Celecoxib is a nonsteroidal anti-inflammatory drug and tramadol is an opioid agonist. Seglentis contains 56 mg of celecoxib and 44 mg of tramadol. It is manufactured by Esteve Pharmaceuticals, S.A., a Spain-based international pharmaceutical company.
Global Hemoglobinopathy Treatment Drugs Market Cross Sectional Analysis:
Introduction of newer products and technologies in treatment of hemoglobinopathy by key market players in Europe region is expected to drive growth of disease type segment in the region. For instance, on April 28, 2023, Akebia Therapeutics, Inc., a U.S. based biopharmaceutical company, has received approval from the European Commission to market its oral hypoxia-inducible factor prolyl hydroxylase inhibitor Vafseo in Europe. Vafseo (vadadustat) was approved as a once-a-day treatment for anemia in adults on chronic maintenance dialysis. The approval is applicable to all 27 European Union member states and Iceland, Norway and Liechtenstein. Vadadustat is approved for use to treat anemia in 32 countries. The oral hypoxia-inducible factor prolyl hydroxylase inhibitor was approved in 150 mg, 300 mg and 450 mg tablets by the EC. Approval is based on phase 3 trials done on 7,500 adults on dialysis treated for anemia.
Hemoglobinopathy Treatment Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5.74 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.6% | 2033 Value Projection: | USD 10.24 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Bristol-Myers Squibb Company, GlycoMimetics Inc., Pfizer Inc., Anthera Pharmaceuticals Inc., Eli Lilly and Company, Mast Therapeutics, Daiichi Sankyo Company Ltd., Novartis International AG, Bluebird Bio Inc., HemaQuest Pharmaceuticals Inc., Emmaus Medical Inc., Prolong Pharmaceuticals, Merck & Co., Inc., Medunik USA Inc., Sangamo Therapeutics, Inc., Global Blood Therapeutics, Alnylam Pharmaceuticals, and Acceleron Pharma, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Hemoglobinopathy Treatment Drugs Market: Key Developments
On October 18, 2021, Esteve, a Spain-based international pharmaceutical company, announced U.S. Food and Drug Administration (FDA) approved Seglentis (celecoxib and tramadol hydrochloride), a proprietary product developed by Esteve’s R&D team. It is an innovative first-in-class product comprised of a co-crystal form of celecoxib (an anti-inflammatory) and tramadol (an analgesic) for the treatment of acute pain in adults. This is ESTEVE's first proprietary research product to enter the U.S. market. Seglentis is the trade name for tablets that contain a co-crystal composed of celecoxib and tramadol hydrochloride. It is a new analgesic designed for acute pain management in a multimodal treatment approach targeting four complementary pain relief mechanisms. It offers a new treatment option for acute pain management aligned with the multimodal analgesia now considered standard of care.
On March 22, 2021, Pacira BioSciences, Inc., a U.S. based pharmaceutical company, announced the U.S. Food and Drug Administration (FDA) has approved the submission of its supplemental new drug application (sNDA) seeking expansion of the EXPAREL label to include use in patients 6 years of age and older for single-dose infiltration to produce postsurgical analgesia. With this approval, EXPAREL is the first and only FDA approved long-acting local analgesic for the pediatric population as young as age six. The sNDA was based on the positive data from the Phase 3 PLAY study of EXPAREL infiltration in pediatric patients undergoing surgeries. Overall findings were consistent with the pharmacokinetic and safety profiles for adult patients with no safety concerns identified at a dose of 4 mg/kg. The PLAY study enrolled 98 patients to evaluate safety and the pharmacokinetics of EXPAREL for two patient groups: patients aged 12 to less than 17 years and patients aged 6 to less than 12 years. Per U.S. FDA guidance, the primary objectives of the PLAY study were to evaluate the pharmacokinetics and safety of EXPAREL.
On July 1, 2023, India launched national sickle cell anaemia elimination mission aiming to screen more than seven crore young tribals so that the debilitating disease can be detected early and managed with therapies. The mission, announced in the Union budget earlier in 2023, focus on awareness creation and universal screening of people in the 0-40 years age group in tribal areas in 278 districts in 17 states. The government plans to eliminate the disease by 2047. This mission includes distribution of sickle cell genetic status card to beneficiaries that would save lives of children and families affected by disease. Government have also established integrated centers for hemophilia and hemoglobinopathies in 22 tribal districts for treatment and diagnosis of patients.
On June 21, 2022, Precision BioSciences, Inc., a U.S. based biotechnology company, announced it has entered into an exclusive worldwide in vivo gene editing research and development collaboration and license agreement with Novartis AG, Switzerland based global healthcare company . As part of the Agreement, Precision BioSciences, Inc will develop a custom ARCUS nuclease that will be designed to insert, in vivo, a therapeutic transgene at a safe harbor location in the genome as a potential one-time transformative treatment option for diseases including certain hemoglobinopathies such as sickle cell disease and beta thalassemia. While, Novartis AG taking responsibility for all subsequent research, development, manufacturing, and commercialization activities. Precision will receive an upfront payment of US$ 75 million and is eligible to receive up to an aggregate amount of approximately US$ 1.4 billion in additional payments for future milestones.
Global Hemoglobinopathy Treatment Drugs Market: Key Trends
Increasing clinical trials for hemoglobinopathy treatment
Research & development activities for hemoglobinopathy treatment can drive growth of market. On November 6, 2021, Bristol Myers Squibb, a U.S.-based multinational pharmaceutical company, and Acceleron Pharma Inc., a U.S.-based clinical stage biopharmaceutical company, announced the first data from the Phase 2 BEYOND study evaluating Reblozyl (luspatercept-aamt), a first-in-class erythroid maturation agent, plus best supportive care in adult patients with non-transfusion dependent (NTD) beta thalassemia. Results demonstrated that 77.7% of patients treated with Reblozyl achieved a hemoglobin increase (≥1.0 gram/deciliter) compared to 0% of patients in the placebo arm. Changes in patient-reported outcomes also correlated with increases in hemoglobin.
Strategies like agreement by key market players
Strategies like agreement between key market players can drive growth of market. On September 8, 2020, Global Blood Therapeutics, a U.S.-based biopharmaceutical company, announced that it has entered into an exclusive agreement with Biopharma-Middle East and Africa, UAE-based pharmaceutical company to distribute Oxbryta (voxelotor) tablets in Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, and UAE, collectively known as the Gulf Cooperation Council (GCC) region. There are estimated to be more than 100,000 people age 12 years and older in this region living with sickle cell disease (SCD). Oxbryta is a once-daily, oral therapy that directly inhibits hemoglobin polymerization, the root cause of the sickling and destruction of red blood cells in SCD.
Global Hemoglobinopathy Treatment Drugs Market: Restraints
Withdrawal of clinical trials
Withdrawal of clinical trials is expected to hamper the global hemoglobinopathy treatment drugs market growth. For instance, according to clinical trials site clinicaltrials.gov a study was conducted to study peripheral blood stem cell collection from patients with sickle cell disease (scd) using plerixafor. This study was terminated on April 7, 2023 as the safety and efficacy of plerixafor in adult and pediatric SCD patients was an unknown when it was started. Hence recruiting patients without a therapeutic option was not ethically justifiable.
To counterbalance this restraint, proper safety and efficacy evaluation should be carried out to avoid withdrawal of study.
Availability of alternative treatment
Availability of alternative treatment like bone marrow is expected to hamper the global hemoglobinopathy treatment drugs market growth. Bone marrow transplant is only reliable treatment for hemoglobinopathy as most treatment options that are available focus on reducing symptoms of the disorders. For instance, according to the data published by Health Resources and Services Administration, a U.S. government agency, in, June 13, 2023, in 2021, 5,073 unrelated and 4,276 related bone marrow and cord blood transplants were performed in the U.S. The donor registry contains more than 9 million potential donors in the U.S. Nearly 268,000 cord blood units are on the donor registry. This total includes more than 111,000 units from the National Cord Blood Inventory (NCBI), with more than 3,000 NCBI units added in 2022.
To counterbalance this restrain, more awareness on advantages of hemoglobinopathy treatment drugs should be spread.
Global Hemoglobinopathy Treatment Drugs Market- Key Players
Major players operating in the global hemoglobinopathy treatment drugs market include Bristol-Myers Squibb Company, GlycoMimetics Inc., Pfizer Inc., Anthera Pharmaceuticals Inc., Eli Lilly and Company, Mast Therapeutics, Daiichi Sankyo Company Ltd., Novartis International AG, Bluebird Bio Inc., HemaQuest Pharmaceuticals Inc., Emmaus Medical Inc., Baxter International Inc., Prolong Pharmaceuticals, Medunik USA Inc., Sangamo Therapeutics, Inc., Global Blood Therapeutics, Alnylam Pharmaceuticals, and Acceleron Pharma, Inc.
*Definition: Hemoglobinopathy is a group of hereditary blood disorders involving an abnormality in the structure of hemoglobin. These disorders include both sickle cell disease (SCD) and thalassemia. Common symptoms of the condition, include severe pain, shortness of breath, enlarged spleen, and growth problems in children. Other types of hemoglobinopathies linked to the structural hemoglobin variants are HbS, HbE, and HbC. The highly variable clinical manifestations of the hemoglobinopathies range from mild hypochromic anemia to moderate hematological disease to severe, lifelong, transfusion-dependent anemia with multiorgan involvement. Drugs to treat the symptoms of hemoglobinopathy include analgesics, antibiotics, ACE inhibitors, and hydroxyurea. According to the data of National Center for Biotechnology Information, more than 90% of patients currently survive the adulthood. Optimally treated patients have a projected life span of 50 to 60 years.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients