The Implantable Drug Delivery Devices Market is estimated to be valued at USD 31.6 Bn in 2026 and is expected to reach USD 52.6 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.8% from 2026 to 2033.
The Implantable Drug Delivery Devices market is expanding as chronic diseases like cancer, diabetes, cardiovascular disorders, and neurological conditions drive demand for long-term, controlled drug administration. Manufacturers are enhancing therapeutic outcomes and patient adherence by developing programmable pumps, biodegradable implants, and targeted delivery systems. Clinicians and patients are increasingly adopting these devices, supported by advanced healthcare infrastructure, minimally invasive procedures, and ongoing research and development that fuels innovation in personalized and sustained drug delivery solutions.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Developments |
|
|
Technological Innovations and Nano-level Advancements |
|
|
Industry Collaborations and Market Competition |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Biodegradable Implants expected to hold largest market share of 52.2% in 2026. The implantable drug delivery devices market is increasingly adopting biodegradable implants as they provide sustained medication delivery without requiring surgical removal, lowering patient risk and healthcare costs. Manufacturers enhance safety and therapeutic effectiveness through advances in biocompatible polymers and controlled-release technologies. Patients and clinicians actively choose these implants for their minimally invasive, user-friendly design, and growing awareness of their temporary benefits drives wider adoption. Together, these factors establish biodegradable implants as a preferred solution for targeted, convenient, and efficient long-term therapy. For instance, in August 2024, Stryker launched InSpace, a biodegradable subacromial balloon implant in India for arthroscopic treatment of massive irreparable rotator cuff tears, a common cause of shoulder dysfunction.
Drug Infusion Pumps hold the largest market share of 34.6% in 2026. The Drug Infusion Pumps segment in the implantable drug delivery devices market grows as healthcare providers seek precise, continuous medication administration for patients with chronic pain, cancer, diabetes, and neurological disorders. Manufacturers enhance treatment effectiveness and convenience through innovations like programmable dosing, wireless monitoring, and compact designs. Patients increasingly choose these devices due to minimally invasive procedures, greater awareness, and supportive healthcare infrastructure, making drug infusion pumps a vital solution for delivering long-term, targeted, and personalized therapies. For instance, in August 2025, Terumo India introduced its Terufusion Advanced Infusion Systems in India, bringing connected infusion technology to improve safety and efficiency in drug administration in ICUs.
Cardiovascular acquired the prominent market share of 34.4% in 2026. The cardiovascular segment of the implantable drug delivery devices market grows as clinicians treat rising cases of coronary artery disease, heart failure, and arrhythmias. Healthcare providers use implantable devices, including drug-eluting stents and cardiac pumps, to deliver precise, localized therapy that improves outcomes and minimizes complications. Manufacturers drive adoption through innovations such as bioresorbable materials, programmable delivery systems, and minimally invasive procedures, while increasing patient awareness and advanced healthcare infrastructure further encourage the widespread use of cardiovascular-focused implantable drug delivery solutions. For instance, in March 2026, GAIA and Daiichi Sankyo Europe have formed an exclusive partnership to commercialize lipodia, a digital therapeutic for adults with hypercholesterolemia, combining GAIA’s expertise in evidence-based interventions with Daiichi Sankyo’s experience in cardiovascular care.
To learn more about this report, Request Free Sample
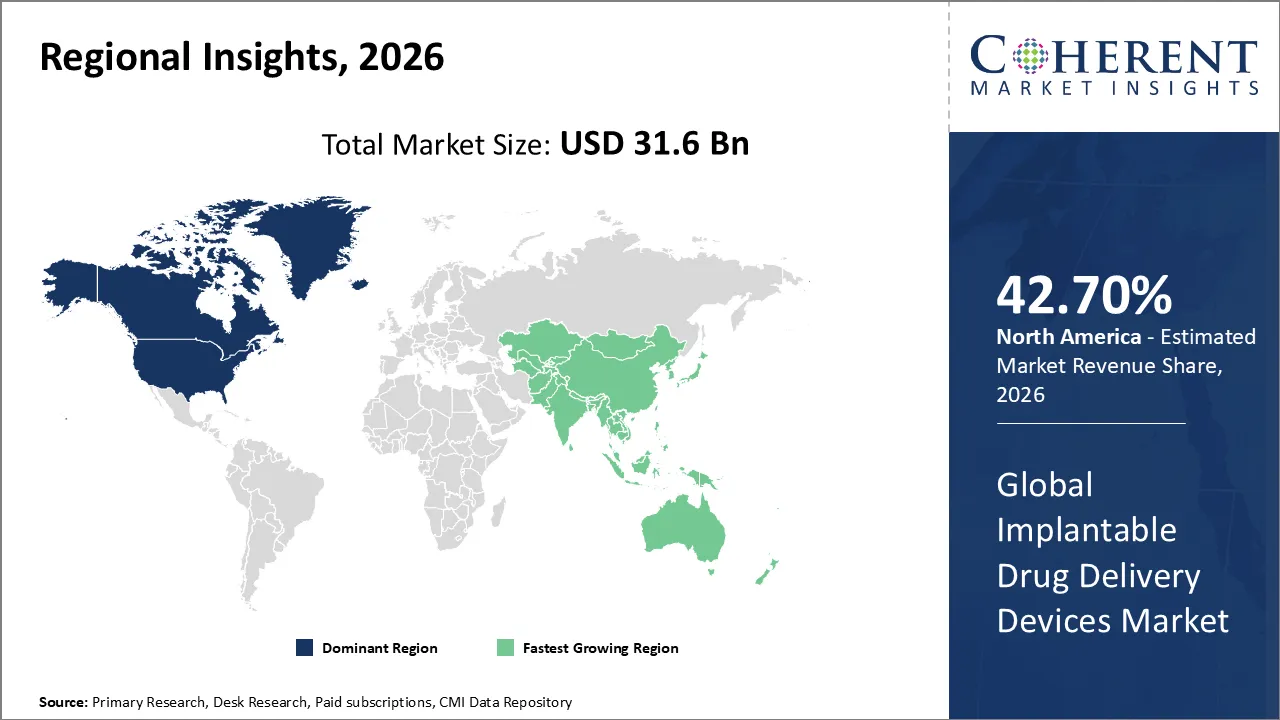
North America dominates the overall market with an estimated share of 42.7% in 2026. The North America implantable drug delivery devices market is advancing as manufacturers develop patient-focused, innovative therapies. Rising cases of chronic conditions, including cancer, diabetes, and cardiovascular diseases, push healthcare providers to adopt precise, long-term drug delivery solutions. Companies introduce programmable pumps, biodegradable implants, and smart monitoring systems, while clinicians increasingly perform minimally invasive procedures. Robust healthcare infrastructure, supportive reimbursement policies, and growing awareness among patients and providers actively drive the wider adoption of implantable drug delivery technologies throughout the region. For instance, in January 2026, Organon announced that the FDA has approved a supplemental New Drug Application for NEXPLANON (etonogestrel implant), a contraceptive indicated to prevent pregnancy in women of reproductive potential.
The Asia Pacific implantable drug delivery devices market grows as healthcare systems modernize and patients increasingly seek advanced therapies. Rising cases of chronic diseases, including cancer, diabetes, and cardiovascular disorders, push providers to implement long-term, precise drug delivery solutions. Manufacturers develop innovative devices such as programmable pumps, biodegradable implants, and targeted delivery systems, while hospitals actively adopt minimally invasive procedures. Expanding healthcare infrastructure, increasing patient awareness, and supportive government initiatives actively drive the broader adoption of implantable drug delivery technologies across the region. For instance, Biogen Inc. and Alcyone Therapeutics have entered a license and collaboration agreement to develop the ThecaFlex DRx System, an implantable device for delivering antisense therapies into the intrathecal space.
The United States implantable drug delivery devices market grows as healthcare providers implement more effective, patient-friendly treatment options. Increasing cases of chronic conditions such as cancer, diabetes, and cardiovascular diseases push demand for precise, long-term medication delivery. Manufacturers develop innovative technologies, including programmable pumps, biodegradable implants, and smart monitoring systems, while clinicians actively perform minimally invasive procedures. Robust healthcare infrastructure, supportive reimbursement policies, and rising awareness among patients and medical professionals actively drive the broader adoption of implantable drug delivery solutions nationwide. For instance, in September 2025, Medtronic plc received FDA approval for Altaviva, a minimally invasive implantable tibial neuromodulation device placed near the ankle to treat urge urinary incontinence.
The Japan implantable drug delivery devices market grows as healthcare providers enhance treatment efficiency and patient quality of life. Increasing cases of chronic diseases, including cancer, cardiovascular disorders, and neurological conditions, drive demand for precise, sustained drug delivery. Manufacturers develop advanced solutions such as programmable pumps, biodegradable implants, and targeted delivery systems, while hospitals actively implement minimally invasive procedures. Robust healthcare infrastructure, government support, and rising awareness among patients and clinicians actively encourage the broader adoption of implantable drug delivery technologies throughout Japan.
Healthcare providers increasingly prefer implantable devices that deliver drugs directly to affected sites, reducing systemic side effects. Innovations in programmable pumps and biodegradable implants allow tailored dosing schedules, improving patient outcomes and adherence. Targeted therapy also supports complex treatments for chronic pain, cancer, and neurological disorders, driving demand for devices that combine precision with long-term efficacy.
Manufacturers are incorporating smart monitoring systems, wireless connectivity, and remote programming into implants. These innovations enable clinicians to track drug delivery, adjust dosages in real time, and reduce hospital visits. Miniaturized designs and improved biocompatible materials also enhance safety, convenience, and patient comfort, making technology-driven devices a major market trend.
There is a growing demand for biodegradable implants and devices with smart monitoring capabilities. These solutions reduce the need for surgical removal, enhance safety, and enable real-time adjustments in therapy. Companies that invest in material science and digital integration can capture opportunities by offering patient-centric, technologically advanced solutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 31.6 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.8% | 2033 Value Projection: | USD 52.6 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Boston Scientific Corporation, Abbott Laboratories, Medtronic Plc., Merck & Co., Bausch & Lomb Inc., Allergan, Inc., Bayer Healthcare, Alimera Sciences, Eyepoint Pharmaceuticals, DSM Biomedical, Delpor Inc., Teleflex Incorporated, and 3M Company. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In July 2025, a team of researchers from Seoul National University (Prof. Seung-Kyun Kang), the Korea Institute of Science and Technology (Dr. Hyojin Lee), Kwangwoon University (Prof. Jeonghyun Kim), and Gyeongsang National University (Prof. Seongchan Kim) developed a wireless implantable drug delivery system that delivers anticancer drugs deep into solid tumors while minimizing damage to surrounding healthy tissue.
In September 2025, Biogen Inc. announced a definitive agreement to acquire Massachusetts-based Alcyone Therapeutics. Through their existing partnership, the companies are advancing ThecaFlex DRx™, an implantable subcutaneous port and catheter system designed for intrathecal delivery of antisense oligonucleotides (ASOs).
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients