Global inflammatory bowel disease market is estimated to be valued at USD 24.46 Bn in 2026 and is expected to reach USD 33.96 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
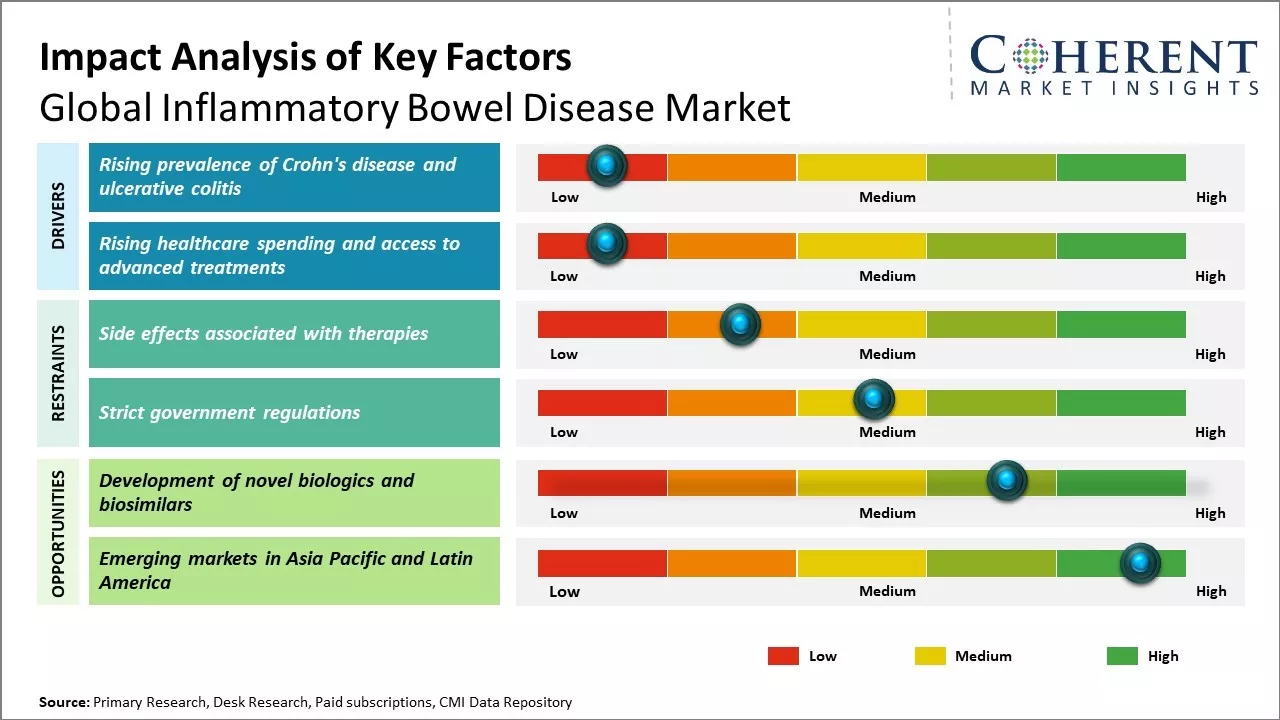
Increasing prevalence of Crohn’s disease and ulcerative colitis across the can drive the market growth. As per the National Center for Biotechnology Information (NCBI), Crohn's disease cases are more common in northern parts of the world such as North America and Northern and Western Europe. While ulcerative colitis cases are more common in the southern parts of the world such as South America and the Middle East. Rising awareness regarding the disease symptoms and management options available can also drive the market growth. However, high costs associated with branded drugs can hamper the market growth during the forecast period.
Rising prevalence of Crohn's disease and ulcerative colitis
The rising prevalence of Crohn's disease and ulcerative colitis globally is one of the major factors fueling the growth of the inflammatory bowel disease (IBD) market. These conditions cause chronic inflammation and ulcers in the digestive tract. According to the latest data from the Crohn's & Colitis Foundation, over 3 million Americans currently suffer from IBD. In Europe as well, nearly 2.5-3 million new cases are reported each year as per the European Federation of Crohn's & Ulcerative Colitis Associations. The increasing disease incidence can be attributed to changing lifestyle patterns and diets around the world. While genetics play a role, experts indicate environmental triggers like smoking, lack of breastfeeding, and low fiber intake as primary causes behind the disease growth. As per the World Health Organization, Canada and North American countries have witnessed the highest surge, with the incidence rates reaching over 200 per 100,000 population in the last decade alone. The growing morbidity demonstrates the urgent need for medications and overall disease management.This rising prevalence has created considerable commercial opportunities in the IBD market globally. Pharmaceutical companies are investing heavily in R&D to develop novel biologics, biosimilars, and small molecules effective for both induction and maintenance of remission. Some of the latest therapies approved include JAK inhibitors, IL-23 and IL-12/23 antibodies catering to the unmet needs. Further, demand for non-drug therapeutics like enteric-coated preparations, nutrition supplements is also growing. The increase in product pipeline and diversity of treatment options available today are positively impacting the revenue potential of this market. Growing patient pool, changing reimbursement programs supporting Biologics in various countries, and emergence of pharma companies in developing regions like Asia Pacific and Latin America will propel the IBD market to greater heights in the next 5 years. However, ensuring drug affordability and access will play a crucial role in maximizing this potential, especially in lower resource settings.
Get actionable strategies to beat competition: Request Free Sample
Rising healthcare spending and access to advanced treatments
Rising social and economic burden of inflammatory bowel diseases has led to greater allocation of healthcare budgets towards effectively managing these conditions. Improved access to healthcare facilities combined with higher disposable income levels have enabled patients to seek and afford superior medical care. This evolving treatment landscape can offer lucrative opportunities for companies offering biologics and other novel drug classes. While still primarily used in refractory cases or as rescue therapy due to their high costs, biologics targeting factors like TNF-alpha and integrins have revolutionized management of Crohn's disease and ulcerative colitis. Constant innovations can further increase convenience of administration through devices like pen injectors, thus improving patient adherence as well. The complex interplay of encouraging reimbursement schemes, enhanced patients' welfare initiatives and the promising drug development pipeline are expected to significantly elevate overall spending on advanced gastrointestinal therapies globally.
Key Takeaways from Analyst:
Global inflammatory bowel disease market growth is driven by rising prevalence of Crohn's disease and ulcerative colitis worldwide. Increasing awareness and better diagnosis of these conditions can also drive the market growth. Moreover, advancements in biologics and targeted therapies for managing IBD symptoms and improving patient outcomes will support revenue generation in the industry.
High costs of biologics can hamper the market growth. Patent expiries of major drugs may reduce prices but also impact sales volumes in the short-term. Regional expansions by emerging manufacturers can increase competition and pricing pressure. Strict regulations for drug approval can delay product launches.
North America currently dominates the market due to higher healthcare expenditure, presence of major players, and growing disease prevalence. Meanwhile, Asia Pacific is likely to emerge as fastest growing market. Rising healthcare standards, growing medical tourism, and increasing patient pools can drive the market growth in the region.
Market Challenges: Side effects associated with therapies
The side effects associated with various inflammatory bowel disease therapies can hamper the global inflammatory bowel disease market growth. Many of the existing treatment options for conditions like Crohn's disease and ulcerative colitis have severe adverse effects that can negatively impact patients' quality of life. Corticosteroids are commonly used as the first line of treatment for inducing remission during flare-ups of IBD. However, long term use of corticosteroids is associated with several problems like weight gain, mood swings, diabetes, high blood pressure, osteoporosis, cataracts, and others. Their use is often limited to achieving remission from acute symptoms. Biologics like anti-TNF therapies have also revolutionized IBD management but these injectable drugs also have a risk of side effects ranging from injection site reactions, headache and nausea to rare but serious infection risks that require long term monitoring. Immunosuppressants used in combination with biologics to maintain remission too have challenges. Drugs like azathioprine cause adverse gastrointestinal reactions and risk of infection in many patients. Their use further increases the vulnerability to opportunistic infections. All these side effect concerns often lead to non-adherence to treatment protocols. Many patients discontinue medications on their own or demand alternative options. This adversely impacts the potential for repeat prescriptions and growth for pharmaceutical companies.
Market Opportunities: Development of novel biologics and biosimilars
The development of novel biologics and biosimilars can offer opportunities for the global inflammatory bowel disease market growth. Biologics have transformed the treatment of various chronic inflammatory conditions over the last few decades. These complex large molecule drugs offer targeted mechanisms of action with greater effectiveness and fewer side effects compared to traditional small molecule drugs. In the case of inflammatory bowel diseases like Crohn's disease and ulcerative colitis, several biologics targeting tumor necrosis factor alpha (TNFα) like infliximab, adalimumab and golimumab along with novel targets like integrins (vedolizumab) and interleukins (ustekinumab) have shown positive results in achieving remission and mucosal healing in patients with moderate to severe forms of the disease. However, high cost of these biologic therapies has been a major barrier to widespread adoption across countries. This is where biosimilars come into play with their reduced costs while maintaining similar safety and efficacy. The patent expiries of some of the blockbuster biologics in the near future can increase competition from biosimilars. For example, Humira, the bestselling drug worldwide, lost its exclusivity in European markets in 2018. This has led to over 80% price reductions with the introduction of biosimilar versions. Similarly, remicade's exclusivity will expire in key markets by 2023. This influx of affordable biosimilar options can boost the adoption rate of biologic therapies, with an estimated over 60% of prescriptions being accounted for by biosimilars by 2025 according to the WHO.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
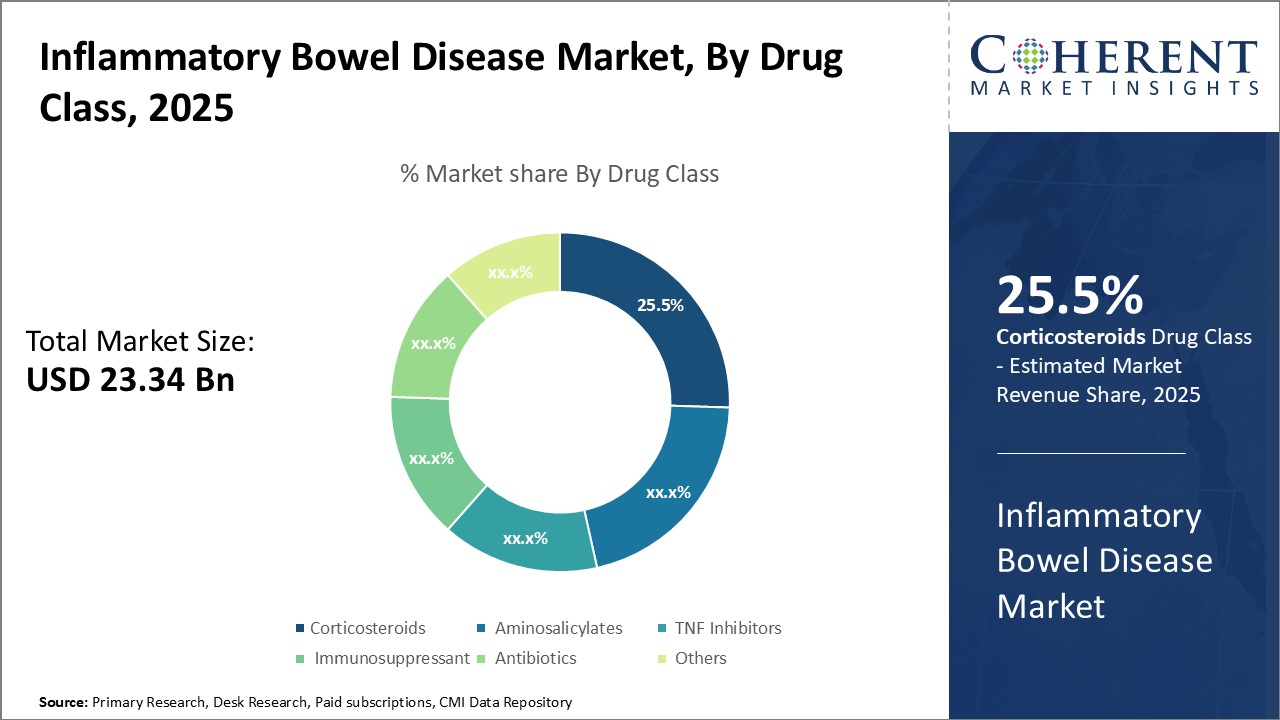
By Drug Class- Growing demand for effective treatment that eases joint pain can drive the corticosteroids segment growth
In terms of drug class, corticosteroids segment is estimated to contribute the highest market share of 26.5% in 2026, owing to its effectiveness in treating ulcerative colitis and Crohn's disease symptoms. As an anti-inflammatory medication, corticosteroids work by reducing swelling, irritation and redness in the colon and small intestine. This makes it very effective in providing immediate relief from abdominal cramps, diarrhea and rectal bleeding associated with inflammatory bowel disease. For patients experiencing moderate to severe flare ups, corticosteroids become the preferred choice of treatment due to its rapid response. Moreover, some corticosteroid enemas and foams allow localized delivery, bypassing absorption into the bloodstream and minimizing harmful systemic effects. Due to increasing diagnosis rates of ulcerative colitis and Crohn's disease worldwide, there has been huge demand for highly effective and fast acting medication like corticosteroids. Its wide availability in both oral and topical formulations also increase accessibility and adherence. Corticosteroids' reputation as the most effective drug in controlling inflammation and symptoms onset during IBD episodes can drive the segment growth.
To learn more about this report, Request Free Sample
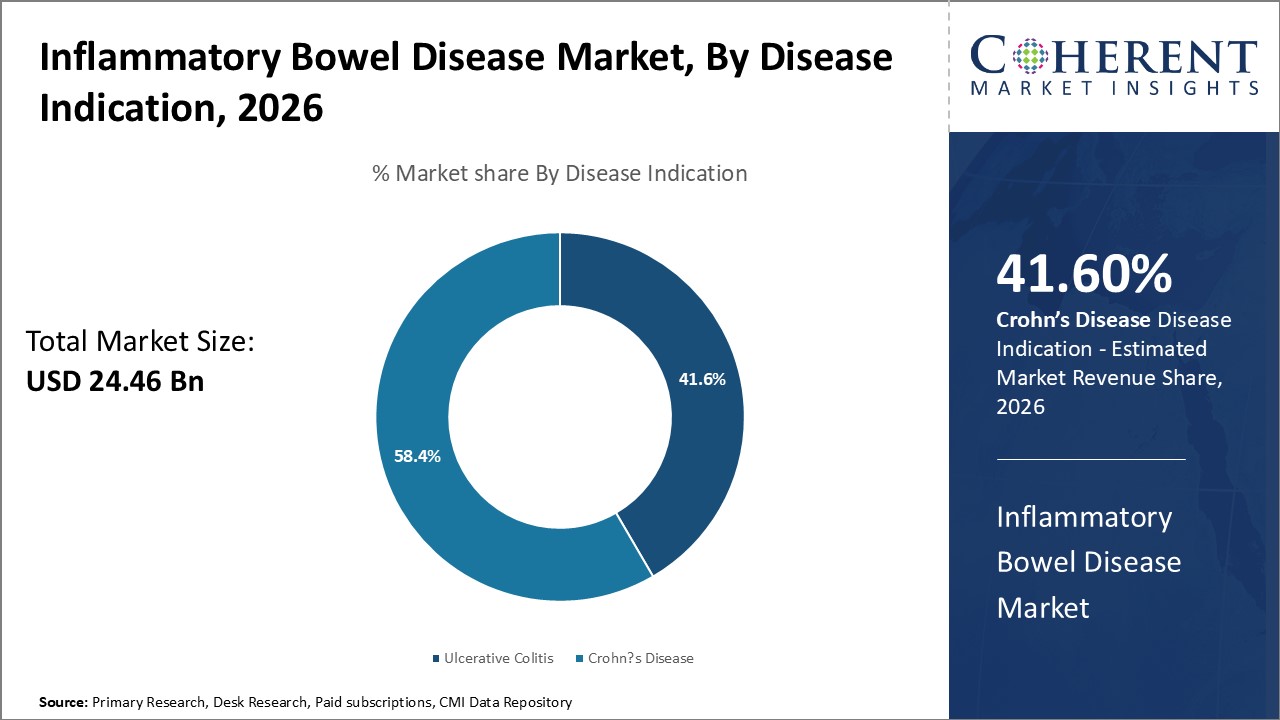
By Disease Indication- Preference for targeted delivery drives ulcerative colitis segment growth
In terms of disease indication, ulcerative colitis segment is estimated to contribute the highest market share of 41.6% in 2026, due to rising preference for targeted medication delivery. Ulcerative colitis only affects the large intestine or colon, leaving the small intestine unaffected. This allows development of treatments focused on the specific delivery site. Routes like suppositories, enemas and foam formats enable the administration of medication directly into the colon. Targeted localization minimizes systemic distribution and exposure to other organs, reducing unwanted side effects. It also ensures that optimal concentration of the drug is achieved at the primary site of inflammation. The benefits of targeted delivery appeal to patients with ulcerative colitis, making it the recommended initial approach during mild to moderate flares. The ability of current therapeutic arsenal to selectively act on only the diseased areas of the large intestine drives the segment growth.
By Route of Administration- Widespread adoption of oral therapies
In terms of route of administration, oral segment is estimated to contribute the highest market share of 55.5% in 2026. The widespread preference for orally administered therapies is primarily driven by patient compliance factors. Being a chronic gastrointestinal condition, inflammatory bowel disease requires long term management and medication adherence. Oral drugs allow patients independent self-administration without requiring assistance or medical supervision. This improves treatment convenience and flexibility for incorporating drugs into daily routines. Moreover, oral formulations involve non-invasive delivery devoid of risks associated with injections. The ease of oral ingestion combined with self-administration capabilities enhance patient acceptance and compliance to prescribed medication regiments long term. Thus, a bulk of therapeutic armamentarium is focused on developing new oral drugs and reformulations of existing ones. This has boosted accessibility while driving market infiltration of oral treatment modalities.
Need a Different Region or Segment? Request Free Sample
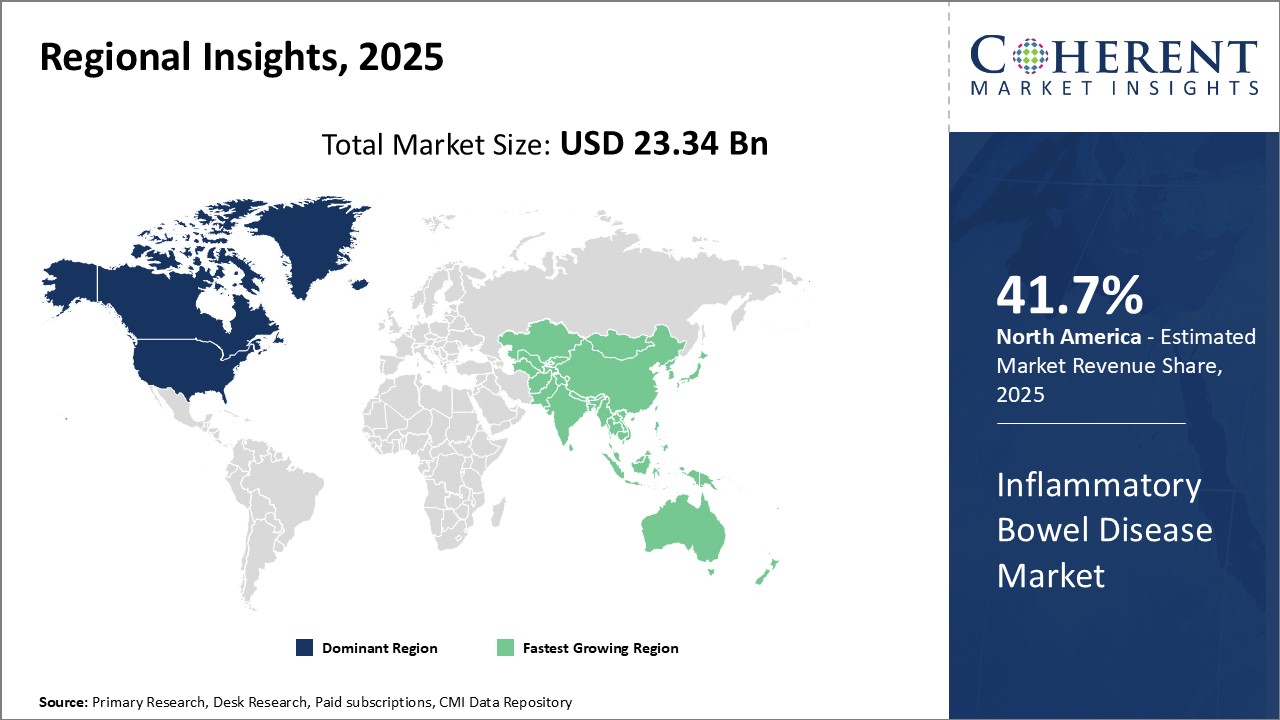
North America dominates the global inflammatory bowel disease market with an estimated market share of 41.7% in 2026. The strong presence of leading pharmaceutical companies in the U.S. drives the market growth in North America. Countries like the U.S. and Canada have witnessed significant investments by market players in R&D activities for developing novel treatment options. Moreover, easy availability of generic drugs at competitive prices has ensured wider patient access to treatment in the region.
However, Asia Pacific region is poised to witness fastest growth rate, owing to rising disease prevalence in emerging nations. Increasing disposable incomes and growing awareness among consumers have boosted healthcare expenditure in countries like China, India and Japan. These nations are focusing on building healthcare infrastructure and expanding drug manufacturing capabilities. International players are also looking at Asia Pacific as an important market for future growth due to large patient pool and improving medical facilities.
While North America remains the largest inflamed bowel disease market globally, the pricing landscape differs across countries. For example, branded drugs tend to be cheaper in Canada compared to the U.S. due to government intervention. India has emerged as a key exporter, owing to its competitive generics industry. This has allowed global drug makers to outsource production to lower costs. However, regulations around intellectual property rights can impact drug approvals and trade between nations.
Inflammatory bowel disease market is witnessing shifts in demand and supply across major geographical territories. While the U.S. wields significant influence due to an established pharmaceutical sector and supportive healthcare policies, Asia Pacific's rise is projected to reshape competitive dynamics in the long run.
Inflammatory Bowel Disease Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 24.46 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 33.96 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
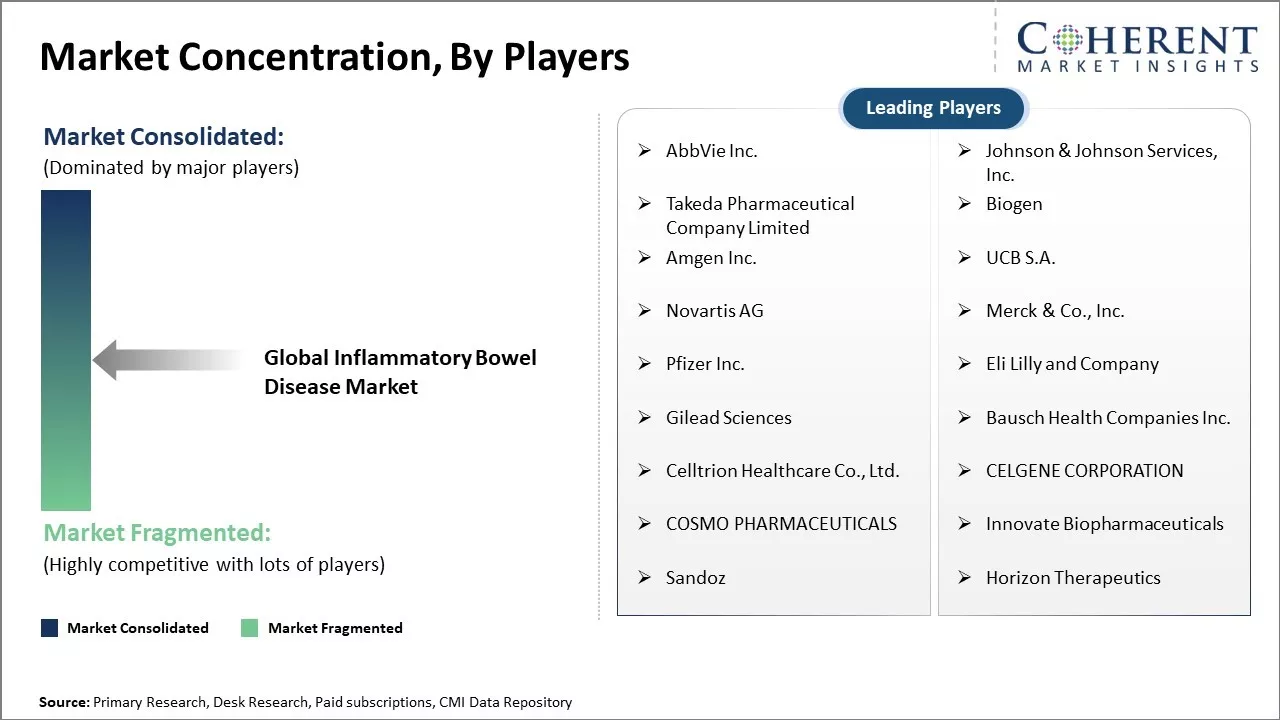
| Companies covered: |
AbbVie Inc., Johnson & Johnson Services, Inc., Takeda Pharmaceutical Company Limited, Biogen, Amgen Inc., UCB S.A., Novartis AG, Merck & Co., Inc., Pfizer Inc., Eli Lilly and Company, Gilead Sciences, Bausch Health Companies Inc., Celltrion Healthcare Co., Ltd., CELGENE CORPORATION, COSMO PHARMACEUTICALS, Innovate Biopharmaceuticals, Sandoz, Horizon Therapeutics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Inflammatory Bowel Disease Market covers treatments for chronic inflammatory conditions of the gastrointestinal tract such as Crohn's disease and ulcerative colitis. The market includes various drug classes such as amino salicylates, corticosteroids, immunomodulators, antibiotics and biologics that help manage symptoms and induce and maintain remission. It also consists of diagnostic and monitoring tests for inflammatory bowel disease. The key players are focusing on developing novel biologics and biosimilars to address the high unmet needs and improve outcomes for people suffering from these chronic gastrointestinal disorders.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients