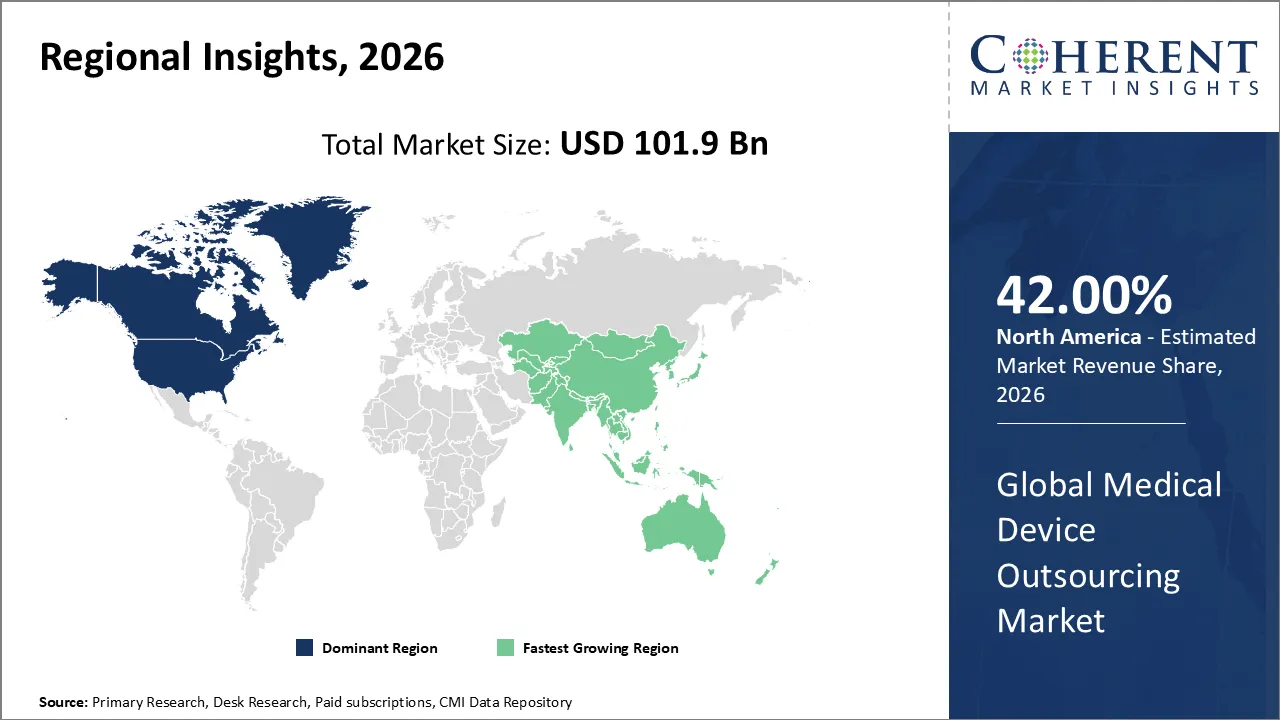
Medical device outsourcing market is estimated to be valued at USD 101.9 Bn in 2026 and is expected to reach USD 231.1 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 12.4% from 2026 to 2033.
The rising trend of employing outsourcing strategies by medical device manufacturing companies is another major driver of the growth of the Global Medical Devices Outsourcing Market. The need to reduce production costs and the emphasis on meeting stringent guidelines are prompting companies, including the original equipment manufacturing companies, to adopt the outsourcing strategy. By employing the strategy of outsourcing, companies are able to concentrate on their core activities such as research, innovation, and marketing, instead of worrying about the manufacturing, assembly, testing, and regulation of their products.
|
Current Events |
Description and its Impact |
|
US-China Technology Trade Relations |
|
|
FDA and Global Regulatory Harmonization Efforts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on product, the finished goods segment is estimated to contribute the highest market share of 52% in 2026 due to the increasing tendency of OEMs to outsource the complete device manufacturing rather than components. The outsourcing of finished goods benefits medical device companies in terms of lower operational cost, scale benefits, and improved speed-to-market.
For instance, in July 2025, Jabil Healthcare launched turnkey manufacturing solutions for medical devices, covering assembly, testing, and regulatory support services, to better serve OEM clients and accelerate commercialization.
By device type, the Class II devices segment is estimated to generate the highest market share, around 50%, in 2026. Class II devices include infusion pumps, surgical drapes, diagnostic imaging devices, and many other devices that incorporate a significant market share in the total medical device production worldwide. Moderate regulatory control is required for Class II devices, making these devices ideal for outsourcing.
For instance, in July 2025, Elevaris Medical Devices, which acts as a contract development and manufacturing organization (CDMO), introduced live prototyping for the development of medical devices, especially those classified under Class II.
Based on application, the cardiovascular segment is expected to hold a maximum share of 23% in 2026 due to rising global disease burdens and rapid advancements within cardiac device technologies. Increasing demand for stents, pacemakers, defibrillators, and catheter-based systems significantly boosts outsourced needs.
For instance, in January 2026, Boston Scientific announced a definitive agreement to acquire Penumbra, Inc. in a transaction valued at approximately $14.5 billion, after which its cardiovascular device portfolio was significantly enhanced, its products targeting conditions such as pulmonary embolism, stroke, deep vein thrombosis, heart attack, and aneurysms.
By services, the finished device manufacturing segment is estimated to contribute the highest market share of 35% in 2026. Medical device companies are increasingly outsourcing full-scale production with the objective of optimizing operational efficiency and concentrating resources on R&D and commercialization strategies. Contract manufacturers provide advanced production technologies, automated assembly lines, and global supply chain networks that ensure high-quality output with cost efficiency.
For instance, in May 2025, Quasar Medical announced the acquisition of Nordson’s contract manufacturing operations in Galway (Ireland) and Tecate (Mexico), significantly enhancing its global manufacturing footprint and production capabilities for complex medical devices.
To learn more about this report, Request Free Sample
North America is likely to continue leading the medical devices outsourcing market and account for the maximum share of 42% by 2026. The factors contributing to its leading position in the market include a well-established healthcare infrastructure, high investment of medical device firms in R&D, strong expertise in regulatory compliance, and wide adoption of outsourcing strategies by OEMs to gain operational efficiencies.
For instance, in March 2025, Medline Industries expanded its contract manufacturing and finished device services in the U.S., including new assembly lines for Class II cardiovascular and diagnostic devices.
The Asia Pacific market is expected to be the fastest-growing market for medical device outsourcing in 2026. The drivers of growth in this market include cost-effective production, availability of skilled labor, growth in healthcare infrastructure, and supportive government initiatives for medical device development in countries such as China, India, and Japan.
For instance, in November 2025, Jabil Healthcare announced the expansion of its manufacturing facility in Suzhou, China, adding advanced assembly and testing lines for Class II and Class III devices.
The U.S. medical devices outsourcing market is expanding at a rapid rate, considering the presence of key OEMs, high healthcare expenditure, and demands for advanced manufacturing technologies. With outsourcing, local manufacturers are focusing more on core product development activities and utilizing CMOs for other finishing activities.
For instance, in August 2025, Flex Ltd. expanded its medical device outsourcing for U.S. customers by adding more value to the service offerings in design, prototyping, and full-scale finished goods manufacturing to offer better support to OEMs in bringing complex devices to the market.
China’s medical devices outsourcing market is rapidly gaining ground as domestic and international OEMs seek cost‑effective manufacturing solutions with advanced capacity. Supportive industrial policies and a large skilled workforce, along with growing domestic demand for medical technologies, are driving investment in contract manufacturing facilities.
For instance, in November 2025, China’s National Medical Products Administration (NMPA) officially released a revised Good Manufacturing Practice (GMP) for Medical Devices, representing the first comprehensive update since 2014.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 101.9 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.4% | 2033 Value Projection: | USD 231.1 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
SGS SA, Intertek Group PLC, Wuxi Apptec, TüvSüd AG, Toxikon, INC., American Preclinical Services, Eurofins Scientific, Sterigenics International LLC, Pace Analytical Services LLC., Charles River Laboratories International, Inc., North American Science Associates, Inc., IQVIA, Accellent Inc., Celestica Inc., Benchmark Electronic Inc., Cadence Inc., Providien LLC, Thermo Fischer Scientific Inc., and West Pharmaceuticals Services Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising complexities associated with medical devices and enhanced regulatory requirements across various countries have prompted medical device OEMs to outsource various factors, including production, assembly, and other regulatory requirements. This is likely to help companies lower their operational costs and thereby enhance product development. The requirements hold greater significance for Class II and Class III devices that require moderate to high levels of technical skill and quality assurance.
The market offers scope for significant growth opportunities through the development of advanced manufacturing technology for sophisticated devices. Design, development, scale manufacturing, assembly, and/or packaging solutions for contract manufacturers can help them form partnerships. In addition, the development of advanced services for emerging markets or growing segments like cardiovascular, orthopedic, and diagnostics can increase revenue opportunities.
The medical devices outsourcing market is undergoing a fundamental transformation, driven by expertise, compliance, and innovation capacity. Outsourcing is increasingly viewed as a strategic extension of R&D and commercialization rather than a simple operational tool. OEMs are now relying on contract manufacturers not only for production but also for regulatory support, quality assurance, and complex assembly processes.
End-to-end manufacturing solutions are becoming the norm. Leading outsourcing partners provide integrated services including prototyping, full-scale production, assembly, testing, and regulatory documentation. Such capabilities allow OEMs to streamline development cycles and reduce time-to-market, particularly for sophisticated cardiovascular, orthopedic, and diagnostic devices. Regional hubs, especially in Asia Pacific, are emerging as centers of technical expertise and regulatory compliance, challenging the traditional perception of these regions as purely low-cost manufacturing destinations.
Regulatory developments, such as updates to China’s GMP and evolving FDA and EU MDR standards, are reshaping outsourcing dynamics. Manufacturers increasingly seek partners who can navigate these frameworks efficiently while maintaining production quality and scalability. This trend underscores that success in outsourcing now hinges on technical competency, regulatory insight, and the ability to deliver integrated solutions, rather than cost alone.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients