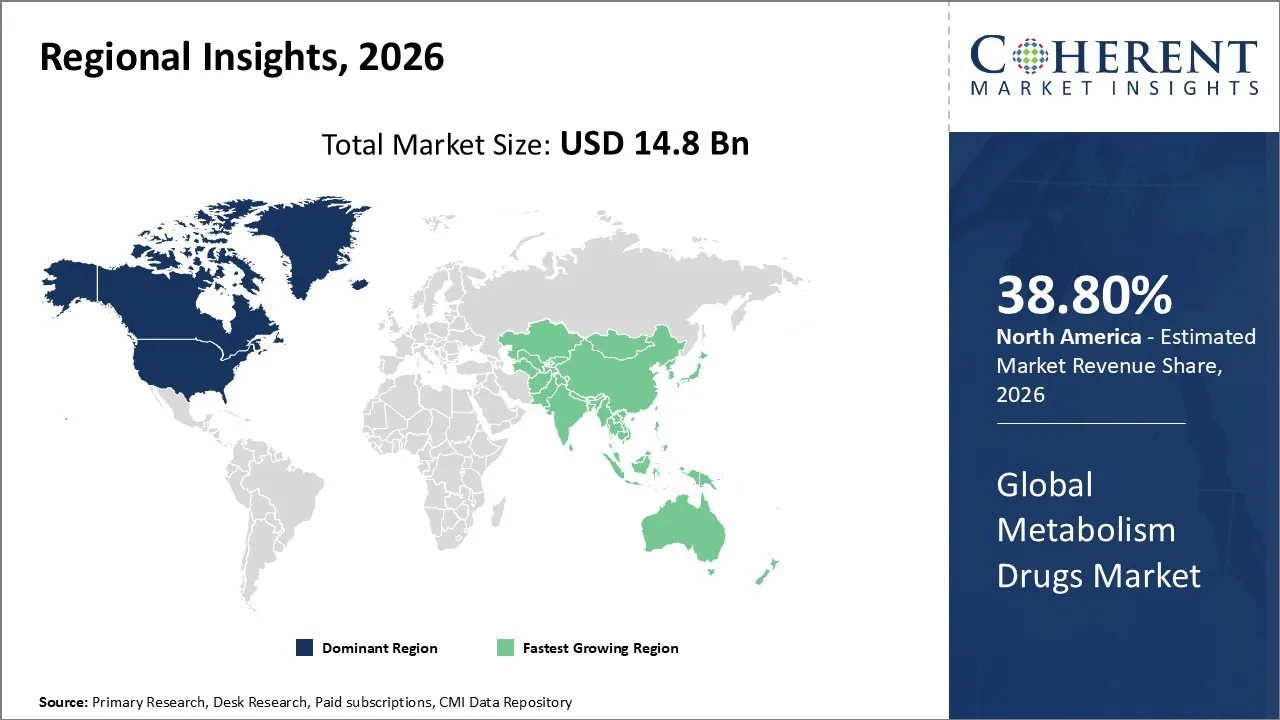
The Metabolism Drugs Market is estimated to be valued at USD 14.8 Bn in 2026 and is expected to reach USD 18.21 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.1% from 2026 to 2033.
The Metabolism Drugs Market delivers therapies that treat metabolic disorders such as diabetes, obesity, lipid abnormalities, and rare inherited metabolic diseases. Rising metabolic condition prevalence, biotechnology advancements, and increasing patient awareness drive the market, which offers insulin, enzyme replacement therapies, biologics, and targeted small-molecule drugs. Pharmaceutical companies actively advance R&D, while hospitals, specialty clinics, and retail pharmacies adopt these therapies supported by robust healthcare infrastructure and improved diagnostics. The growing emphasis on personalized medicine and digital health continues to expand market reach.
|
Current Events |
Description and its impact |
|
Geopolitical Developments Affecting Pharma Trade |
|
|
Technological Advancements in Drug Development |
|
|
Epidemiological Trends and Public Health Initiatives |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospital Pharmacies acquired the prominent market share of 45.8% in 2026. Hospital pharmacies drive the Metabolism Drugs Market by delivering complex therapies for metabolic disorders, such as insulin, biologics, and enzyme replacement treatments, safely and effectively. Increasing hospital admissions for chronic and acute metabolic conditions, the demand for clinical monitoring, and the management of high‑cost or specialized medications fuel their growth. Pharmacy teams use advanced infrastructure and integrate digital health systems to optimize therapy management, while healthcare providers and rising patient awareness expand access, further increasing hospital pharmacy utilization in this market.
Enzyme Replacement Therapy hold the largest market share of 34.6% in 2026. Enzyme replacement therapy (ERT) in the Metabolism Drugs Market grows as more patients are diagnosed with rare metabolic disorders such as Gaucher, Pompe, and Fabry diseases. Biotechnological innovations and recombinant enzyme production improve therapy safety and effectiveness, while pharmaceutical companies advance R&D and expand clinical pipelines with new treatment options. Regulators provide supportive frameworks and orphan drug incentives that promote adoption, and healthcare providers raise awareness to ensure timely treatment. Hospitals and specialized clinics increase access, further boosting ERT use globally. For instance, Takeda launched Adzynma (ADAMTS13, recombinant-krhn) to treat adults and children with congenital thrombotic thrombocytopenic purpura (cTTP) after receiving FDA approval for its use as a prophylactic or on-demand enzyme replacement therapy.
Glycogen Metabolism Disease expected to hold largest market share of 42.2% in 2026. The Metabolism Drugs Market for glycogen metabolism diseases expands as clinicians increasingly diagnose and recognize glycogen storage disorders, growing the number of patients receiving treatment. Pharmaceutical companies develop more effective and targeted therapies through advances in enzyme replacement, gene therapies, and precision medicine. Researchers and regulators drive innovation with strong R&D investment and supportive policies. Healthcare providers and patient advocacy groups raise awareness, while hospitals and specialty clinics broaden access, ensuring timely therapy initiation and wider adoption of treatments for these rare metabolic conditions.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. Rising cases of diabetes, obesity, and other metabolic disorders drive the North American Metabolism Drugs Market, increasing demand for advanced therapies like insulin, biologics, and enzyme replacement treatments. Pharmaceutical companies introduce novel drugs through strong innovation, robust R&D pipelines, and supportive regulatory frameworks. Healthcare providers expand access and improve adherence by leveraging widespread infrastructure, high patient awareness, and digital health tools. Hospitals, specialty clinics, and retail pharmacies actively deliver services, while personalized medicine and targeted therapies continue to shape market dynamics across the region.
Rising urbanization, lifestyle changes, and growing cases of diabetes, obesity, and other metabolic disorders drive the expansion of the Asia Pacific Metabolism Drugs Market, boosting demand for effective therapies. Pharmaceutical companies develop accessible and cost-effective treatments, including insulin, biologics, and enzyme replacement therapies, to address regional needs. Healthcare providers enhance diagnosis and treatment access through expanding infrastructure, hospital networks, and patient education. Governments promote initiatives, increase healthcare spending, and encourage digital health adoption, further supporting the distribution and management of metabolic drugs across the region. For instance, in November 2025, Precision Health Research, Singapore (PRECISE) launched Phase III of the National Precision Medicine Programme to study how genomic information can be effectively applied in healthcare, focusing on patients receiving medical treatments and clinical services.
The high prevalence of diabetes, obesity, and metabolic disorders drives growth in the United States Metabolism Drugs Market, increasing demand for innovative therapies such as insulin, biologics, and enzyme replacement treatments. Pharmaceutical companies launch advanced drugs backed by robust R&D pipelines and regulatory incentives, while healthcare providers improve patient care through extensive clinical infrastructure and digital health integration. Hospitals, specialty clinics, and retail pharmacies expand access actively, and rising patient awareness promotes early diagnosis and treatment adherence, establishing the U.S. as a key center for metabolic drug development and adoption. For instance, in March 2025, Duke-NUS Medical School and NUS Medicine launched the Systems Metabolomics Center (SysMeC), their first joint research center, to advance metabolomics research, enable earlier disease detection, and support precision medicine for improved patient outcomes.
Rising urbanization, changing diets, and increasing cases of diabetes, obesity, and other metabolic disorders drive the expansion of the China Metabolism Drugs Market, boosting demand for effective therapies. Pharmaceutical companies develop affordable and accessible treatments, including insulin, biologics, and enzyme replacement therapies, to serve the large patient population. Healthcare providers improve diagnosis and treatment by expanding hospital networks, specialized clinics, and telemedicine services. Government programs, increased healthcare investment, and adoption of digital health solutions further strengthen the distribution and management of metabolic drugs across the country.
The market is increasingly shaped by the global rise in diabetes, obesity, dyslipidemia, and rare inherited metabolic conditions. Lifestyle changes, urbanization, and aging populations contribute to higher disease incidence. This trend drives demand for a broad range of therapies, from insulin and biologics to enzyme replacement treatments. Healthcare providers and pharmaceutical companies are focusing on early diagnosis, prevention programs, and patient education to manage disease progression and improve long-term patient outcomes.
Pharmaceutical innovation continues to transform the market with next-generation biologics, recombinant enzymes, and gene-targeted therapies. These advances offer higher efficacy, fewer side effects, and personalized treatment approaches. Research is expanding pipelines for rare metabolic disorders, while improved delivery methods, including subcutaneous injections and longer-acting formulations, enhance patient convenience and adherence. Manufacturers are increasingly integrating biotechnology and precision medicine to address unmet therapeutic needs across both common and rare metabolic diseases.
Rare inherited metabolic disorders, such as glycogen storage diseases and lysosomal storage disorders, remain under-treated, offering untapped market potential. Advances in gene therapy, enzyme replacement, and personalized treatments provide pharmaceutical companies the opportunity to introduce novel, life-changing therapies. Collaborations with research institutions, patient advocacy groups, and regulatory agencies can accelerate clinical development and market access, addressing unmet needs and establishing leadership in specialized segments of the metabolism drugs market.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 14.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.1% | 2033 Value Projection: | USD 18.21 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., Novartis AG, Amicus Therapeutics, AstraZeneca Plc, Boehringer Ingelheim, Shire Plc, Sanofi Genzyme, BioMarin Pharmaceutical, Inc., Horizon Pharma Plc, and GlaxoSmithKline Plc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients