Myeloproliferative Neoplasms Treatment Market is estimated to be valued at USD 10.11 Bn in 2026 and is expected to reach USD 13.39 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.1% from 2026 to 2033.
Analysts’ Views on Global Myeloproliferative Neoplasms Treatment Market:
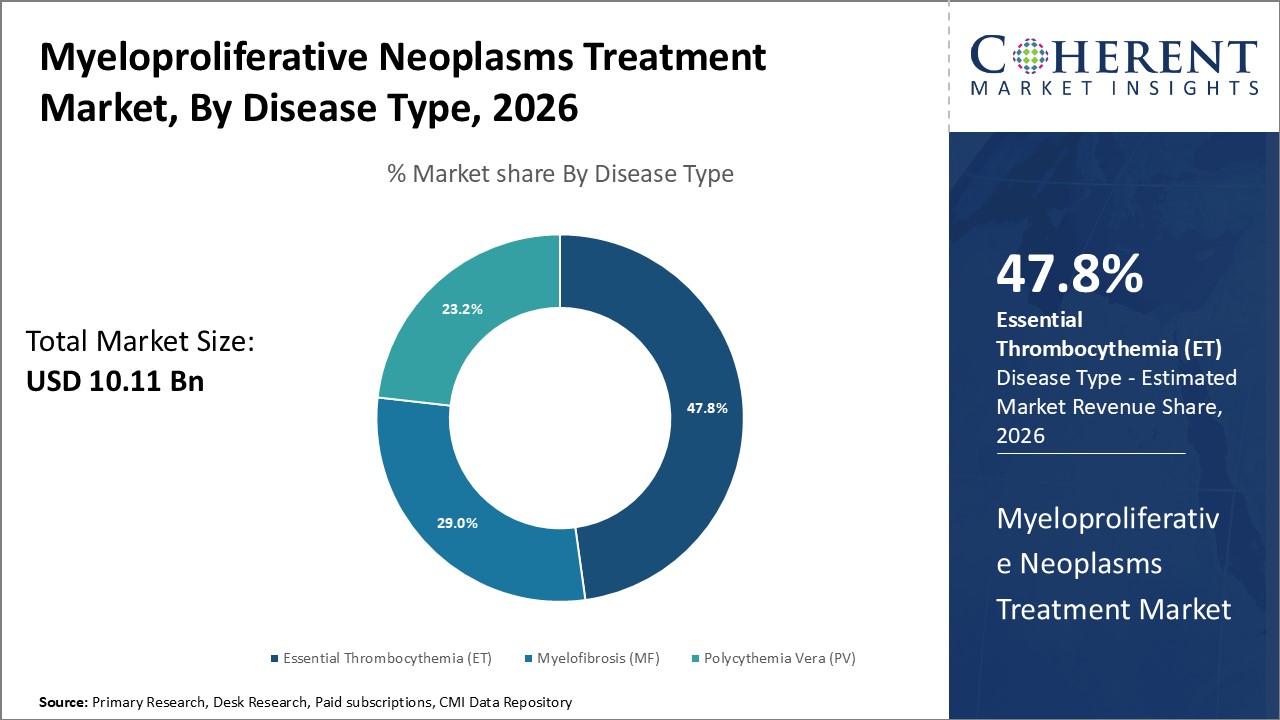
Myeloproliferative neoplasms (MPNs) are types of blood cancer that begin with an abnormal mutation (change) in a stem cell in the bone marrow. The change leads to an overproduction of any combination of white cells, red cells and platelets. Type of Myeloproliferative neoplasms (MPNs) are Essential Thrombocythemia - Essential Thrombocythemia (ET) Is a rare blood disease in which the bone marrow produces too many platelets. High numbers of platelets may lead to a thrombus, a blood clot that forms in a blood vessel. Myelofibrosis - Myelofibrosis (MF) is a rare disorder in which abnormal blood cells and fibers build up in the bone marrow. Polycythemia Vera - Polycythemia Vera (PV) Is one of a related group of blood cancers known as “myeloproliferative neoplasms” (MPNs). Too many red blood cells are made in the bone marrow and, in many cases, the numbers of white blood cells and platelets are also elevated.
Figure 1. Global Myeloproliferative Neoplasms Treatment Market Share (%), By Disease Type, 2026
To learn more about this report, Request Free Sample
Global Myeloproliferative Neoplasms Treatment Market – Drivers
Increasing research and development activities by the market players: Increasing research and development activities by the market players is expected to prople the market growth over the forecast period. For instance, in May 4, 2023, PharmaEssentia, a pharmaceutical company, announced the initiation of its ECLIPSE PV Phase IIIb clinical trial of ropeginterferon alfa-2b-njft (BESREMi) for the treatment of polycythemia vera (PV), a rare, chronic and life-threatening blood cancer. The single-arm study will evaluate an accelerated dosing schedule for ropeginterferon alfa-2b-njft against the currently labelled dosing in 100 adults from U.S. and Canada. Moreover, the subjects will receive starting dosage of 250mcg, 350mcg at week two, with a target optimal dose of 500mcg at week four. In September 8, 2022, Silence Therapeutics, a pharmaceutical company, was granted fast track designation by the U.S. Food and Drug Administration (FDA) for the treatment of polycythemia vera.
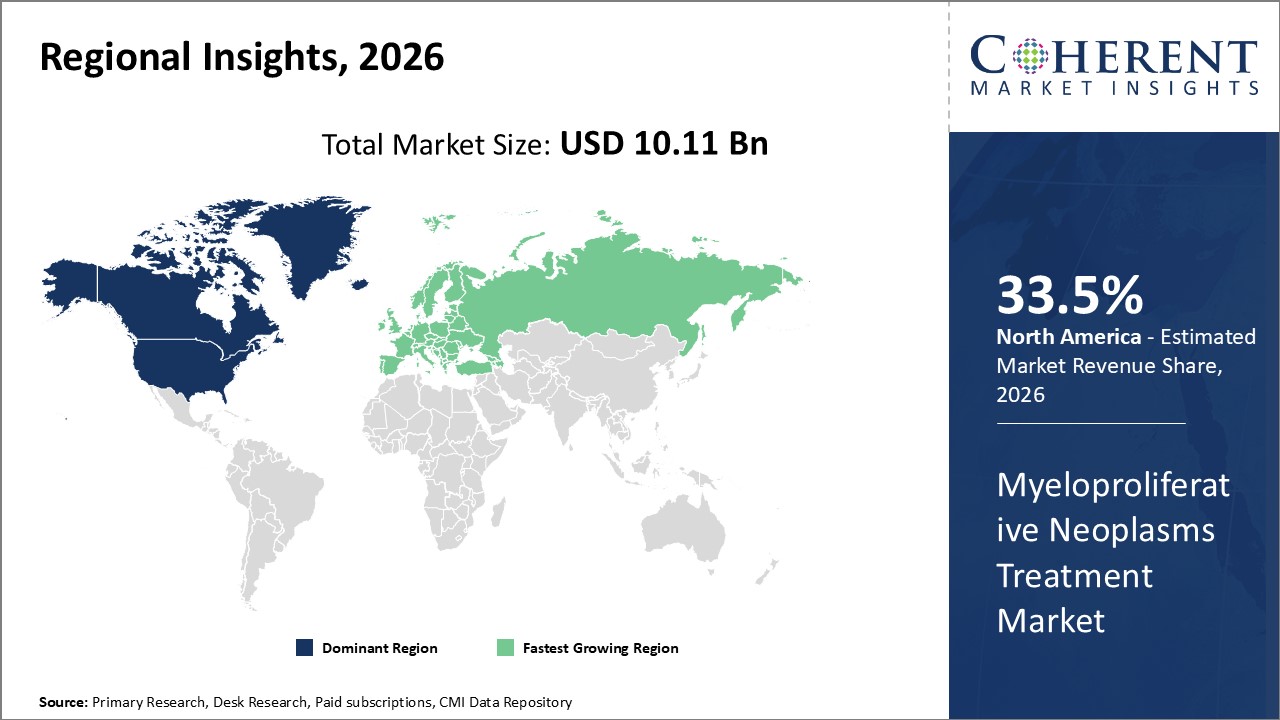
Figure 2. Global Myeloproliferative Neoplasms Treatment Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global myeloproliferative neoplasms treatment market - Regional Analysis
Among regions, North America is expected to hold a dominant position in the global myeloproliferative neoplasms treatment market over the forecast period. North America is estimated to hold 33.5% of the market share in 2026. The global myeloproliferative neoplasms treatment market is expected to witness significant growth in the coming years, driven by the high prevalence of myeloproliferative neoplasms. Increasing prevalence of diseases like polycythemia vera in the region is expected to aid in the market growth over the forecast period. For instance, according to the data published by the National Center for Biotechnology Information (NCBI) in May 20, 2023, the prevalence of polycythemia vera has been estimated to be approximately 22 cases per 100,000 population globally.
Myeloproliferative Neoplasms Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 10.11 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.1% | 2033 Value Projection: | USD 13.39 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, PharmaEssentia Corporation, Bristol Myers Squibb, Pfizer, Inc., Takeda Pharmaceutical Company Limited, Incyte, Johnson & Johnson, Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche AG, Gamida Cell, Sierra Oncology, Inc, CTI BioPharma Corp, Imago BioSciences, AbbVie Inc, and Constellation Pharmaceuticals GMBH |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Moreover, the according to the data published by the World Health Orgnisation in June 1, 2020, the healthcare services have been partially or completely disrupted in many countries. More than half (53%) of the countries surveyed have partially or completely disrupted services for the treatment of non communicable diseases.
Global Myeloproliferative Neoplasms Treatment Market Segmentation:
The global myeloproliferative neoplasms treatment market report is segmented by disease type, drug class, distribution channel, and region.
Among all the segmentation, the Essential Thrombocythemia (ET) segment is expected to dominate the market over the forecast period.
Global Myeloproliferative Neoplasms Treatment Market - Cross Sectional Analysis:
Key players are focusing on inorganic strategies such as merger and acquisition is also expected to boost myeloproliferative neoplasms treatment market. For instance, in June 10, 2020, Menarini Group, a privately held Italy-based pharmaceutical and diagnostics company, announced the acquisition of Stemline Therapeutics Inc., a commercial-stage biopharmaceutical company focused on the development and commercialization of novel oncology therapeutics, for US$ 677 million.
Global Myeloproliferative Neoplasms Treatment Market: Key Developments
Global Myeloproliferative Neoplasms Treatment Market: Key Trends
Global Myeloproliferative Neoplasms Treatment Market: Restraint
Global Myeloproliferative Neoplasms Treatment Market - Key Players
Major players operating in the global myeloproliferative neoplasms treatment market include Novartis AG, PharmaEssentia Corporation, Bristol Myers Squibb, Pfizer, Inc., Takeda Pharmaceutical Company Limited, Incyte, Johnson & Johnson, Teva Pharmaceutical Industries Ltd, F. Hoffmann-La Roche AG, Gamida Cell, Sierra Oncology, Inc, CTI BioPharma Corp, Imago BioSciences, AbbVie Inc, and Constellation Pharmaceuticals.
Definition: Myeloproliferative neoplasms (MPNs) are types of blood cancer that begin with an abnormal mutation in a stem cell in the bone marrow. The change leads to an overproduction of any combination of white cells, red cells and platelets.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients