The naloxone market is estimated to be valued at USD 1,644.1 Mn in 2026 and is expected to reach USD 3,370.5 Mn by 2033, growing at a compound annual growth rate (CAGR) of 10.8% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
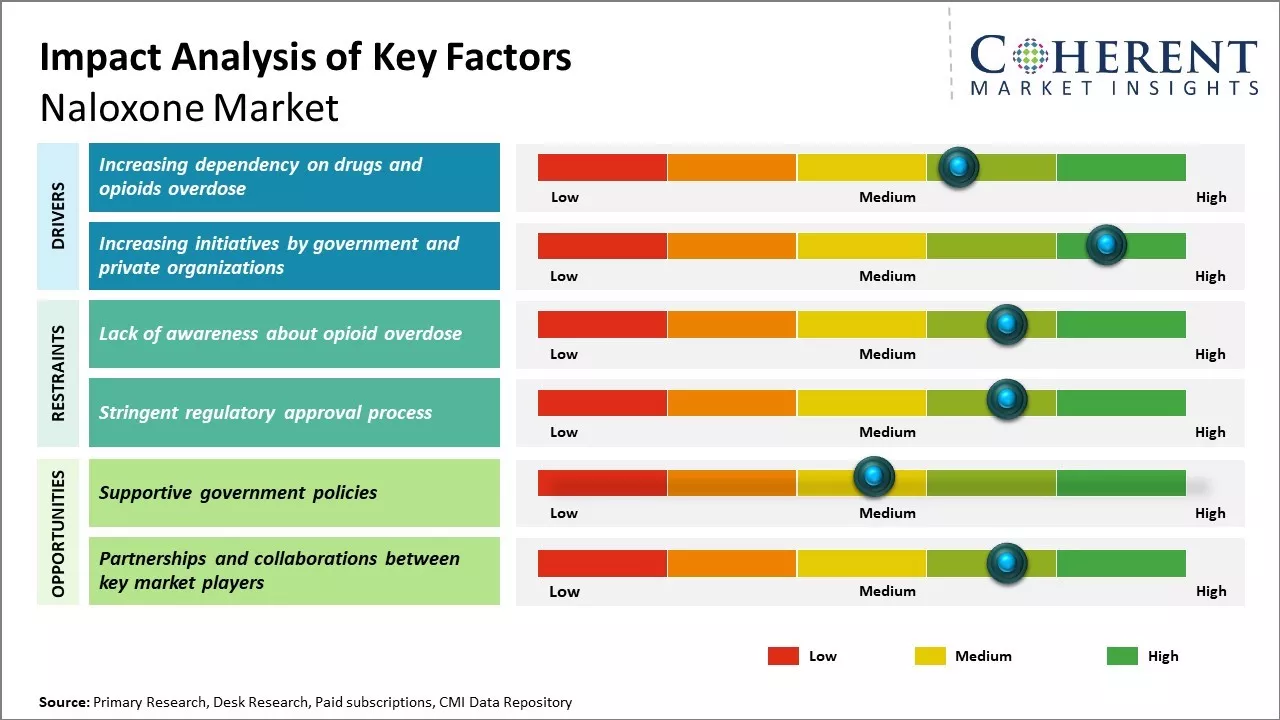
The demand for naloxone is expected to rise significantly during the forecast period owing to the increasing prevalence of opioid overdose cases across the world. With rising opioid misuse worldwide due to increased opioid prescriptions for chronic pain management, the cases of opioid overdoses have surged. Naloxone acts as an antidote to reverse opioid overdose and save lives. Favorable government policies and programs aimed at increasing public awareness and availability of naloxone are also expected to boost the market growth during the analysis period.
Increasing dependency on drugs and opioids overdose
For instance, according to the data published by the World Health Organization (WHO) in August 2021, around 0.5 million deaths are caused by drug use, with more than 30% of these deaths attributed to overdose and more than 70% of these deaths caused due to opioids worldwide. Moreover, according to the data published by the Centers for Disease Control and Prevention, in 2021, around 100,306 drug overdose deaths were reported in U.S. during 12-month period ending in April 2021.
Get actionable strategies to beat competition: Request Free Sample
Increasing Initiatives by Government and Private OrganizationsVarious naloxone manufacturers, private organizations, and government organizations are focusing on initiatives to reduce the death rate related to opioids overdose by increasing access to products such as naloxone for the population. For instance, on March 3, 2022, the Delaware Department of Health and Social Services’ Division of Substance Abuse and Mental Health (DSAMH) announced about hosting free monthly naloxone access training for all members of the Delaware state, U.S. This training will include learning about recognizing and responding to an opioid overdose.

To learn more about this report, Request Free Sample
Market Challenges: Lack of awareness about opioid overdoseOne of the key challenges for the naloxone market is the lack of awareness about opioid overdose and the importance of naloxone use. Many people still view addiction as a moral failing rather than a public health issue. This stigma prevents discussions around naloxone and limits its accessibility. Regaining community trust and changing public perception will take significant educational efforts.
Market Opportunities: Supportive government policies
The ongoing opioid epidemic has shone a light on this issue and helped shift discussions into a healthcare framework. Government policies now encourage naloxone distribution and Good Samaritan laws. This policy support can help the market expand distribution beyond first responders. Rising overdose rates also mean more people may witness an event where naloxone could save a life, increasing willingness to carry it.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
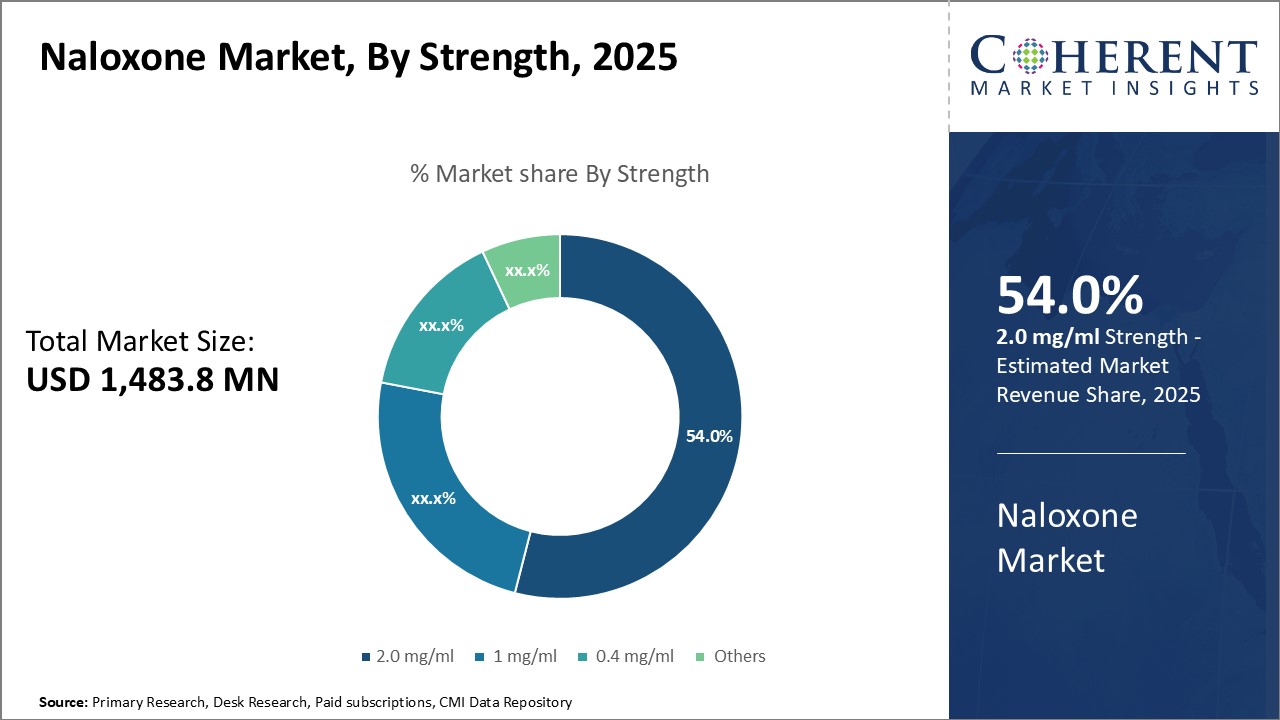
Insights, By Strength: Convenience and precision drive the adoption of higher strength formulationsThe strength segment includes 2.0 mg/ml, 1 mg/ml, 0.4 mg/ml, and others. The 2.0 mg/ml segment is anticipated to hold 55.0% of the market share in 2026. Many patients experiencing opioid overdose require immediate emergency treatment, making every second critical. The higher concentration of 2.0 mg/ml allows for a smaller volume of drug to be administered through alternate routes like intranasal or intramuscular injections. This is particularly useful when intravenous access may be difficult or delayed. It also reduces the risk of dosing errors as lesser volume needs to be measured accurately. The more concentrated formulation effectively balances the need for swift reversal of overdose against ensuring the right dose is delivered. Additional training for medical professionals and awareness programs on its safe use have further boosted preference and market position of the 2.0 mg/ml variant.
To learn more about this report, Request Free Sample
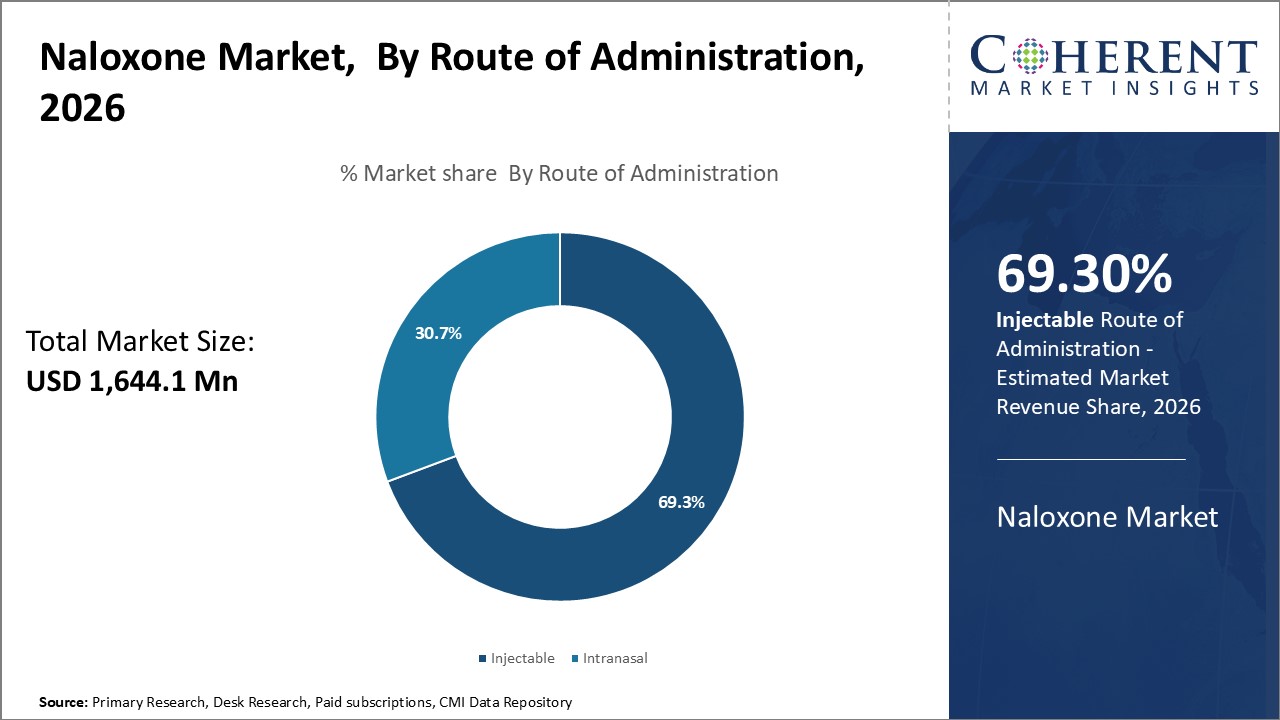
Insights, By Route of Administration: Injectable route drives adoption in emergency response
The route of administration segment includes intranasal and injectable. Injectable contributes the highest share of the naloxone market and is projected to hold 69.3% of the market share in 2026. The injectable segment holds a dominant share in the naloxone market due to its effectiveness in emergency situations like an opioid overdose. Administering naloxone through an injection allows for higher bioavailability and a quicker onset of action compared to other potential routes of administration. Injectable naloxone provides almost instant drug release into the bloodstream, generally bringing someone back from an overdose within 2 to 8 minutes. With every second being critical during an overdose emergency, injectable drugs that get absorbed and circulated immediately are preferred by medical professionals. The ease of use of intramuscular or subcutaneous injections allows bystanders or first responders to rapidly counteract the effects of opioids before it's too late.
Insights, By Distribution Channel: Accessibility and familiarity boost retail pharmacy dominance
The distribution channel segment includes hospital pharmacies, retail pharmacies, and online pharmacies. Retail pharmacies contribute the highest share of the naloxone market and is projected to hold 40.7% of the market share in 2026. Patients and caregivers frequently choose to fill naloxone prescriptions from retail stores which are widely spread across most communities. This provides them with round-the-clock availability without requiring an appointment or wait time. Retail pharmacies are also where individuals go regularly for other medication needs. Their experience and knowledge of retail outlets instills confidence among consumers for procuring naloxone conveniently. With rising rates of opioid abuse developing into a public health crisis, retail distribution further aids over-the-counter dispensing policies and take-home naloxone programs and training. Wide reimbursement coverage has further cemented retail pharmacies as the most approachable access point for this important rescue medication.
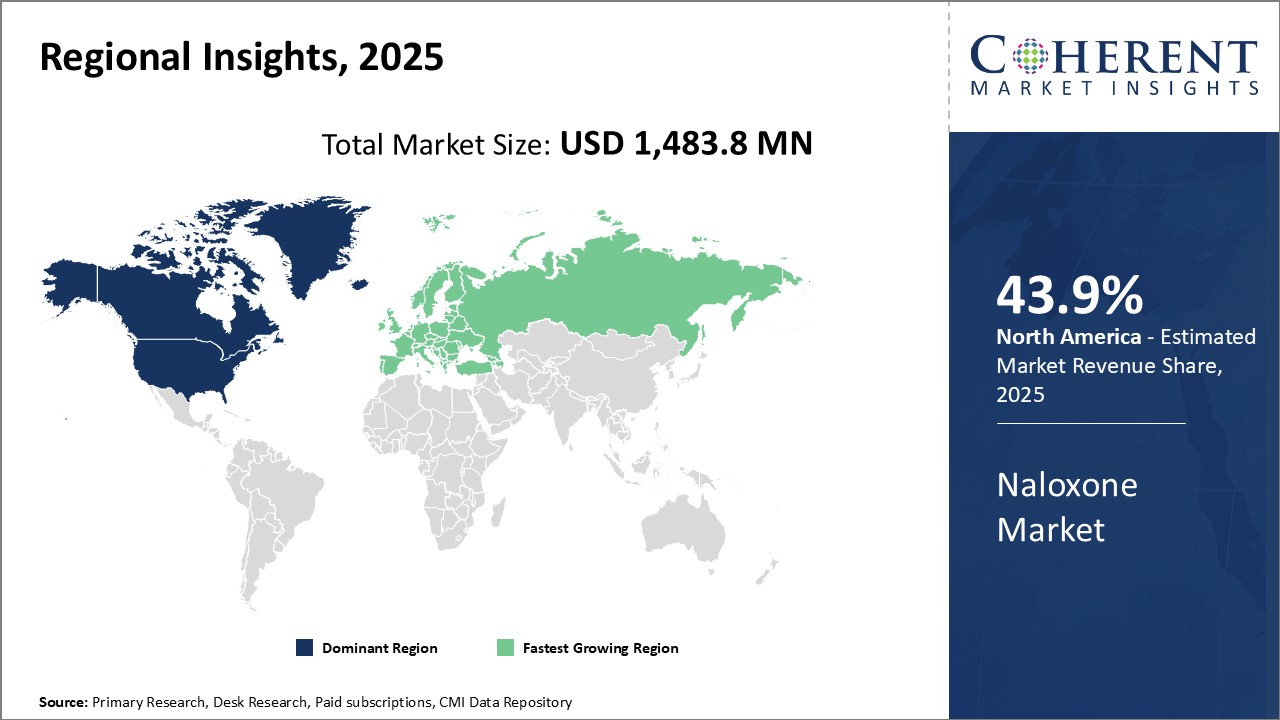
Need a Different Region or Segment? Request Free Sample
North America represents the largest market for naloxone globally and is expected to hold 43.9% of the market share in 2026. The growing epidemic of opioid abuse and rising mortality rates has led to increased government focus and policy changes to expand access to naloxone. Several states and municipalities now recommend or require naloxone to be co-prescribed with opioid prescriptions, thus driving up demand. Moreover, most private and public healthcare plans provide coverage for naloxone, making it affordable for users. Leading pharmaceutical manufacturers also have a well-established presence in the U.S. with distribution networks across the supply chain, ensuring wide availability of branded and generic naloxone products.
Europe has emerged as the second major region for naloxone, led by the markets in Germany, France, and the U.K. While the crisis of opioid abuse is not as severe as in North America, many European countries are gradually realizing the therapeutic potential of naloxone in addressing increasing drug overdose cases. Naloxone prescription trends in these countries show faster growth compared to other European markets. Manufacturers have also prioritized these countries and launched localized educational programs to promote off-label use of naloxone by potential first responders like family members. This is positively impacting the accessibility of naloxone nasal sprays and auto-injectors, particularly in community settings. The supportive regulatory environment and nationwide healthcare coverage further support the growth of the naloxone market across major Western European markets.
Naloxone Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,644.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.8% | 2033 Value Projection: | USD 3,370.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
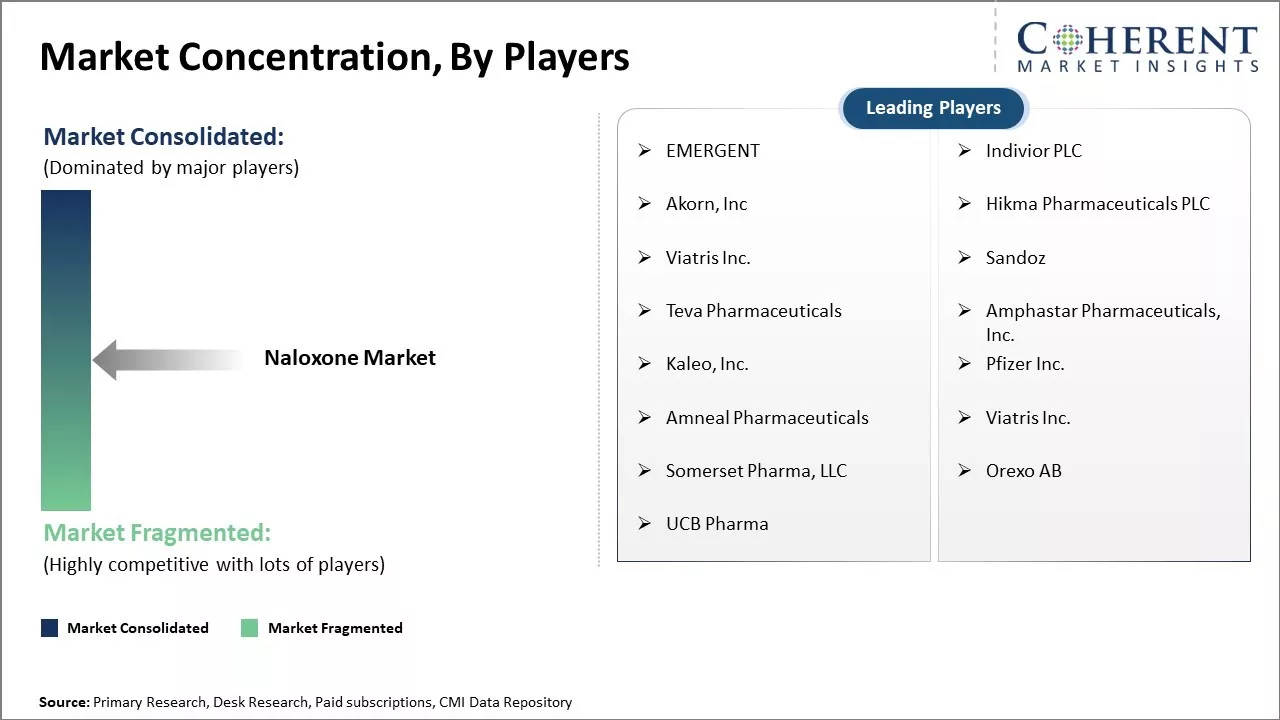
| Companies covered: |
EMERGENT, Indivior PLC, Akorn, Inc, Hikma Pharmaceuticals PLC, Viatris Inc., Sandoz, Teva Pharmaceuticals, Amphastar Pharmaceuticals, Inc., Kaleo, Inc., Pfizer Inc., Amneal Pharmaceuticals, Viatris Inc., Somerset Pharma, LLC, Orexo AB, and UCB Pharma |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients