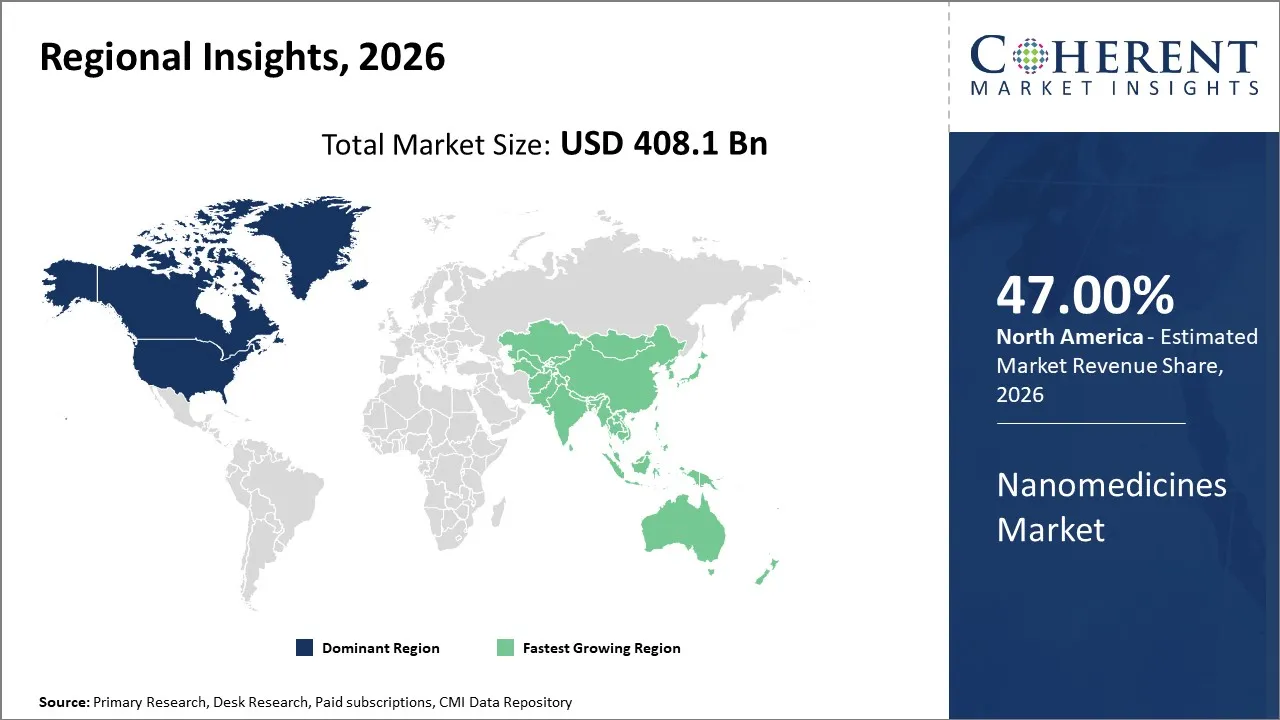
The global nanomedicines market size is expected to reach approximately USD 408.1 Bn in 2026 and USD 925.5 Bn by 2033, growing at a CAGR of 12.9% throughout the forecast period (2026-2033). The market is driven by the increasing regulatory approval for complex nano-formulations, stronger public and private investment in nanomedicine R&D, and expanding use of nanocarriers in oncology and RNA therapeutics.
Nanomedicines use nanotechnology (particles 1–100 nanometers) to change healthcare through focused drug delivery, enhanced diagnostics, and regenerative medicine. Improved drug stability and solubility, less systemic toxicity, the capacity to pass through biological barriers (such as the blood-brain barrier), and earlier, more accurate illness diagnosis are some of the main advantages. The application of nanotechnology principles to medicine is known as nanomedicine. This strategy encourages the growth of personalized medicine, which has the potential to completely transform healthcare. Nanomedicine has some drawbacks as well as intrinsic benefits including precision and biocompatibility when compared to traditional and molecular medicine. The nanomedicines market is gaining strategic importance due to improve drug delivery, increase efficacy, and reduce side effects compared with conventional formulations.
|
Current Event |
Description and its Impact |
|
Increasing adoption of inorganic growth Strategies |
|
|
Public-sector nanotechnology funding remains strong, supporting future nanomedicine pipelines |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on application, oncology is expected to lead the market, accounting for a revenue share of 45% in 2026. High global cancer burden and strong need for target drug delivery supports the dominance of the segment in the market. Cancer treatment requires precise targeting of tumor cells, which nanomedicines effectively provide through nanoparticle-based delivery systems. Many approved nanomedicines drugs (e.g., liposomal formulations) are extensively used in cancer therapy, contributing to higher market share. Pharmaceutical companies heavily in oncology-focused nanotechnology research, leading to continuous innovation and product development. Moreover, increasing prevalence of oncology has driven the growth of the market over the forecast period. For instance, in February 2026, the American Cancer Society (ACS) estimates that there will be more than 2.11 million new cases of cancer and roughly 626,140 cancer-related deaths in the U.S. in 2026, with an average of 5,800 new diagnoses every day. Lung cancer continues to be the most common cause of death, surpassing the total deaths from pancreatic and colorectal malignancies.
To learn more about this report, Request Free Sample
North America is expected to dominate the global nanomedicines market, accounting for a share of 47% in 2026. North America region holds largest revenue share in the market due to its high healthcare expenditure, favourable regulations, and strong R&D capabilities, and advanced healthcare facilities. North America leads the global market due to the presence of a highly developed research ecosystem with leading research institutes, universities, and biotech hubs driving continuous innovation in nanomedicines. A large number of patients with cardiovascular diseases, neurological disorders, and cancer drives strong demand for effective and targeted treatments. North America is quick to adopt innovations such as personalized medicine and nanotechnology-based drug delivery, giving it a competitive edge over other region. Furthermore, many global leaders in nanomedicines and drug development are based in North America, contributing to large-scale production and commercialization.
Asia Pacific is expected to grow at the fastest CAGR in the nanomedicines market during the forecast period. Asia Pacific has huge population base with rising cases of cardiovascular diseases, diabetes, and cancer which creates strong demand for targeted and effective treatments. Rapid urbanization, changing lifestyle and aging are increasing chronic disease burden, driving the need for innovative drug delivery systems. Increasing government support and funding in the Asia Pacific region has significantly driven the growth of the market. Countries like China, India, and Japan are significantly increasing healthcare spending. Allowing greater adoption of advanced therapies like nanomedicines. Moreover, favorable regulatory developments and improving healthcare infrastructure has significantly driven the growth of the Asia Pacific market.
The U.S. country holds the dominant share in the North America nanomedicines market due to the presence of strong research & development ecosystem, high healthcare expenditure, and presence of major pharmaceutical & biotech companies in the U.S. The U.S. Food and Drug Administration provide a structured and supportive approval pathway for advanced therapies, encouraging frequent drug approvals and launches.
The China country dominates the Asia Pacific nanomedicines market due to presence of world’s largest populations, with rising cases of chronic diseases which creates strong demand for advanced treatment solutions like nanomedicines. Increasing government initiative and funding programs in China has improved access to advanced therapies and medicines. China promotes nanotechnology through national research programs, funding initiatives and strategic healthcare policies. China offers skilled workforce and lower production costs making it global hub for drug manufacturing and exports. Moreover, China is focused on improving its regulatory framework to encourage innovation and speed up drug approvals which supports faster commercialization.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 408.1 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.9% | 2033 Value Projection: | USD 925.5 Bn |
| Geographies covered: |
|
||
| Segments covered: | By Application: Cardiovascular, Anti-inflammatories, Anti-infective, Neurology, Oncology, Others (dental, orthopedic, urology, opthalmic) | ||
| Companies covered: |
Abbott Laboratories, CombiMatrix Corporation, Clene Nanomedicine, Inc., Cellics Therapeutics, Inc., Nanobiotix S.A, Celgene Corporation, GE Healthcare, NanoViricides, Inc., Johnson & Johnson, Mallinckrodt plc., Sirnaomics Inc., Precision NanoSystems Inc., Merck & Company Inc., Nanosphere, Inc., Pfizer Inc., and Sigma-Tau Pharmaceuticals Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence of chronic diseases has significantly driven the growth of the market over the forecast period. Aging and unhealthy lifestyles are increasing chronic disease incidence globally. Due to increasing disease burden, there is rise in demand for advanced therapies. Chronic diseases like cardiovascular diseases, cancer, and neurological disorders require effective and long-term treatment solutions. Nanomedicines offer efficient targeted and efficient drug delivery, increasing adoption.
Furthermore, according to World Health Organization, with an estimated 17.9 million deaths annually, cardiovascular diseases (CVDs) are the world's leading cause of mortality.
Increasing approval and launch of new products has expanded available treatment options. Approved products gain regulatory validation, which increases trust among physician and patients, leading to quicker adoption. Each new product launch generates additional sales and revenues streams, directly increasing overall market size. Innovation and launch of new product increase competition, pushes market players to improve drug delivery systems and develop more advanced formulations, which enhances overall market development.
Personalised medicines provide higher treatment effectiveness as they are manufactured based on molecular and genetic profile. The global shift toward precision healthcare is boosting the adoption of personalized nanomedicines, thereby expanding the overall market. Personalised nanomedicines are highly effective in cancer treatment, where therapies are designed based on tumor biology, driving demand in the dominant oncology segment. In addition, personalised customized therapies have higher success rates in clinical trials, encouraging pharmaceutical companies and biopharmaceutical companies to invest more in nanomedicine research & development. Moreover, increasing government funding for the development of the personalised nanomedicine and nanotechnology-based research, fuels product development and commercialization.
Smart Nanocarriers provide controlled and targeted drug release, which has increased the demand for the smart nanocarriers. Smart nanocarriers can release drugs in response to specific conditions such as temperature, enzymes, and pH, ensuring precise delivery to diseases sites and improving treatment efficiency. These advanced carriers reduce exposure of healthy tissues to drugs, thereby cutting-off toxicity and adverse reactions, increasing patient acceptance. Moreover, use of smart nanocarriers has many other benefits such as enhanced solubility and drug stability, improved therapeutic outcomes, and expansion into multiple therapeutic areas.
Growing demand for targeted drug delivery has estimated to create significant opportunity for the growth of the nanomedicines market. Targeted drug delivery ensures that drugs reach the specific diseased sites (e.g., tumor cells), better patient outcomes and improves therapeutic efficiency. Nanomedicines reduce exposure of healthy tissues to drugs, thereby reducing adverse effects and toxicity, which increases their adoption over conventional therapies. Moreover, targeted drug delivery is highly effective in oncology and widely used in treating chronic diseases such as neurological and cardiovascular disorders. Nanotechnology enhances the stability of drugs and solubility, allowing better absorption and effectiveness, especially for poorly soluble drugs. Hence, rising demand for targeted drug delivery drives the nanomedicines market by improving treatment efficiency, safety and patient outcomes, while supporting advancements in chronic and personalized disease therapies.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients